Wave Particles Physics 100 Chapt 22 Maxwell E











- Slides: 22

Wave? Particles? ? Physics 100 Chapt 22

Maxwell E B James Clerk Maxwell Light is a wave of oscillating E- and B-fields

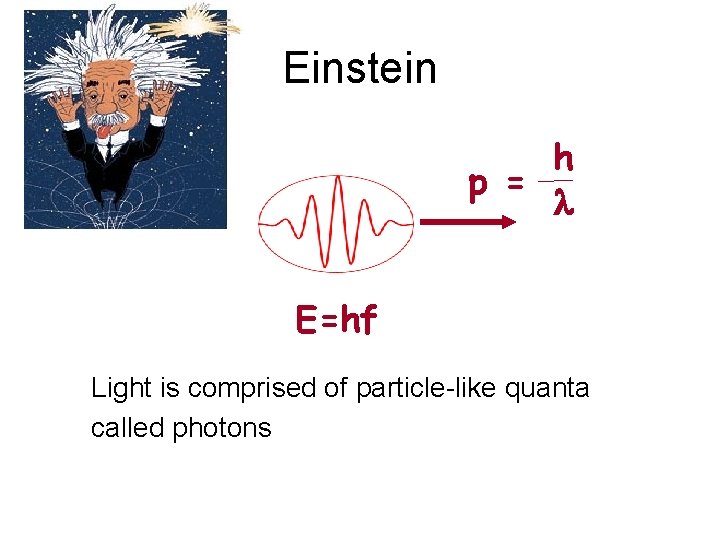
Einstein h p = l E=hf Light is comprised of particle-like quanta called photons

Who’s right? ? Waves explain diffraction & interference Photons explain photoelectric effect & Compton scattering

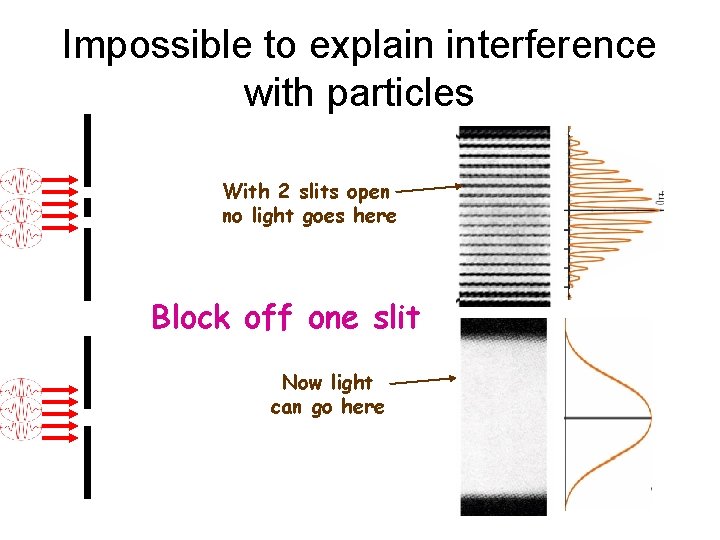
Impossible to explain interference with particles With 2 slits open no light goes here Block off one slit Now light can go here

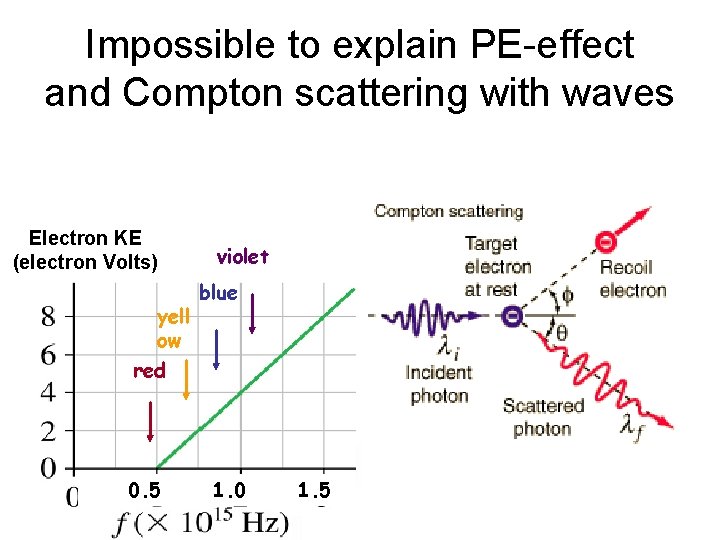
Impossible to explain PE-effect and Compton scattering with waves Electron KE (electron Volts) yell ow violet blue red 0. 5 1. 0 1. 5

Make an intereference pattern with low intensity light One photon at a time goes through the two-slit apparatus The interference pattern emerges one dot at a time

Wave-Particle “duality -Light behaves like a wave when it propagates through space -And as a particle when it interacts with matter

Photon photography

Louis de Broglie Wave-particle duality is a universal phenomenon If light behaves as particles, maybe other particles (such as electrons) behave as waves Photons: particles: h p = l h l = p h l = p

Ordinary-sized objects have tiny wavelengths g k 2 0. 30 m/s -34 Js 6. 6 x 10 h = l = p mv 0. 2 kg x 30 m/s = 6. 6 x 10 -34 Js = 1. 1 x 10 -34 m 6. 0 kg m/s Incredibly small

the wavelength of an electron is not so small 9 x 10 -31 kg - 6 x 106 m/s h = l = p mv = 6. 6 x 10 -34 Js 9 x 10 -31 kg x 6 x 106 m/s 6. 6 x 10 -34 Js 5. 4 x 10 -24 kg m/s = 1. 2 x 10 -10 m About the size of an atom

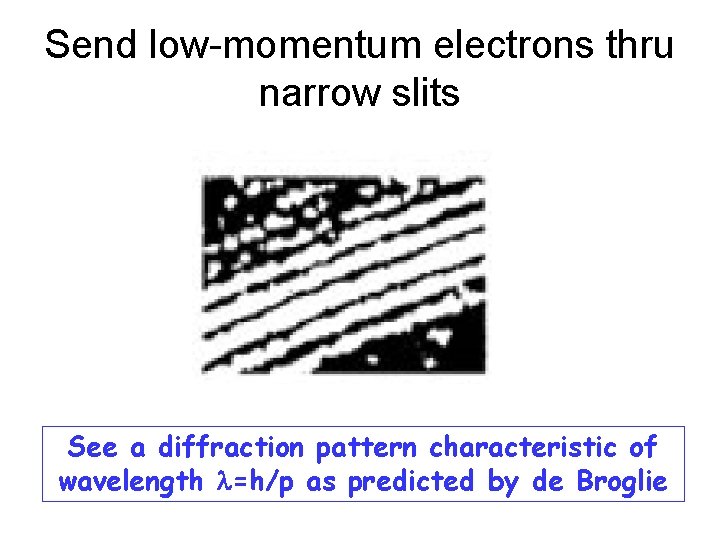
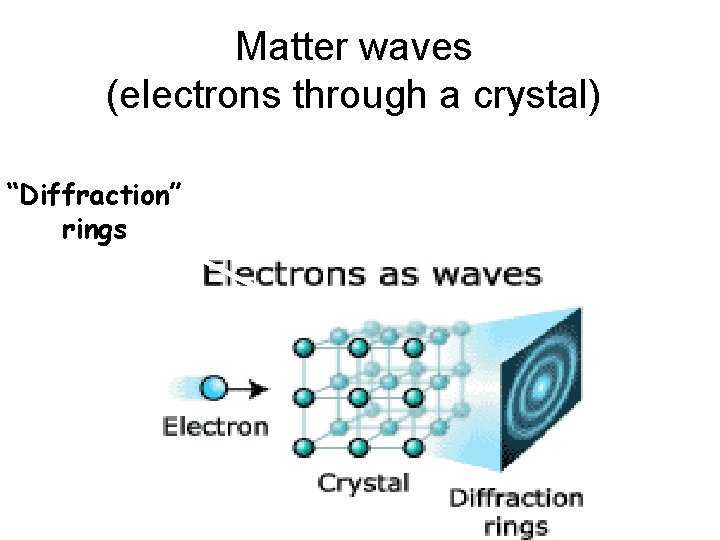
Send low-momentum electrons thru narrow slits See a diffraction pattern characteristic of wavelength l=h/p as predicted by de Broglie

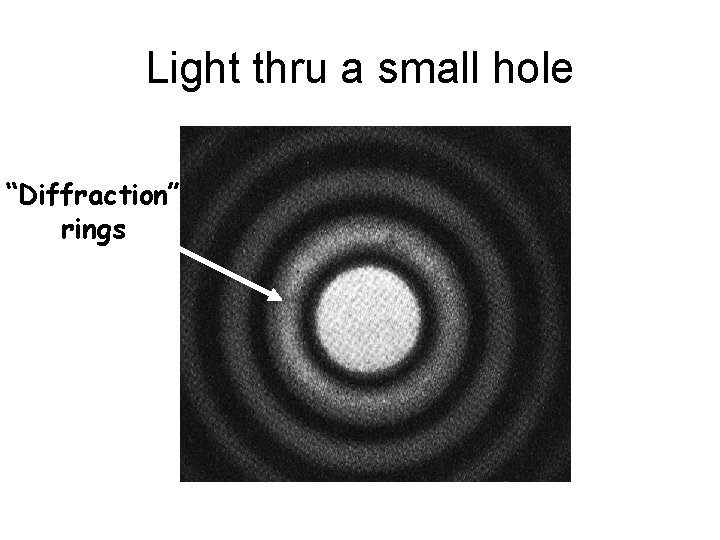
Light thru a small hole “Diffraction” rings

Matter waves (electrons through a crystal) “Diffraction” rings

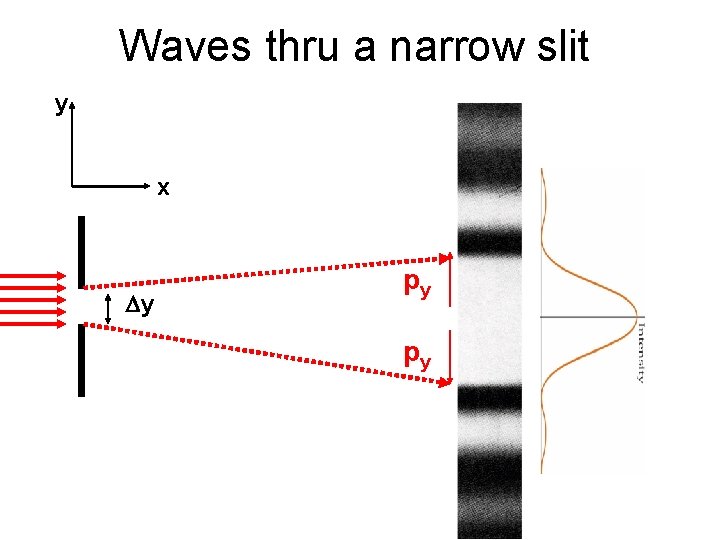
Waves thru a narrow slit y x Dy py py

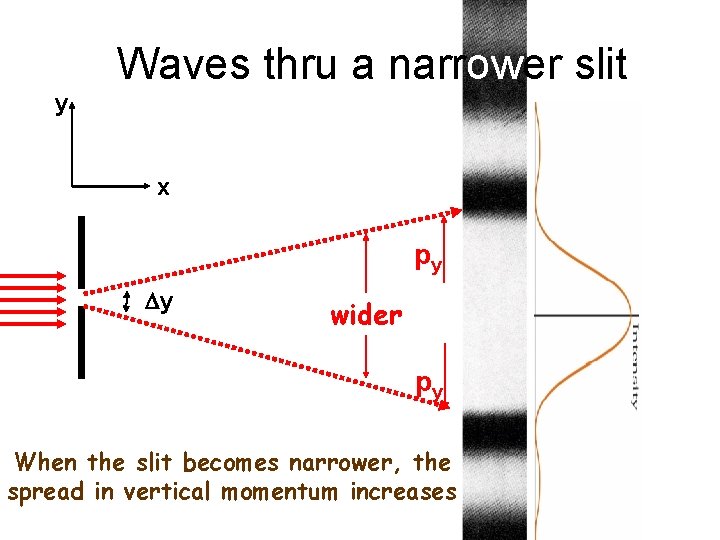
Waves thru a narrower slit y x py Dy wider py When the slit becomes narrower, the spread in vertical momentum increases

Heisenberg Uncertainty Principle Dy Dpy > h Uncertainty in location Uncertainty in momentum in that direction If you make one of these smaller, the other has to become bigger

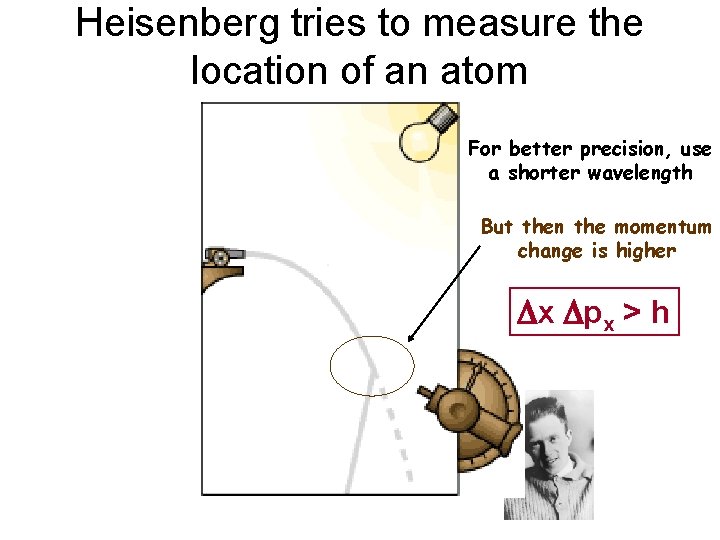
Heisenberg tries to measure the location of an atom For better precision, use a shorter wavelength But then the momentum change is higher Dx Dpx > h

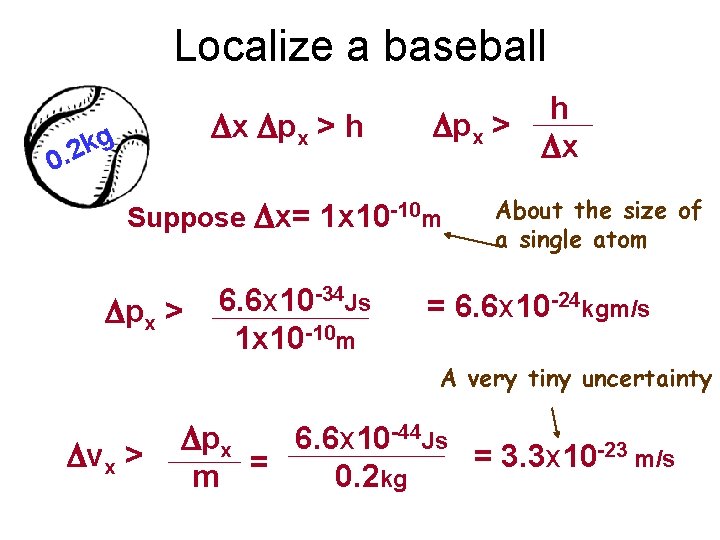
Localize a baseball Dx Dpx > h g k 0. 2 h Dpx > Dx Suppose Dx= 1 x 10 -10 m Dpx > 6. 6 x 10 -34 Js 1 x 10 -10 m About the size of a single atom = 6. 6 x 10 -24 kgm/s A very tiny uncertainty Dvx > Dpx 6. 6 x 10 -44 Js -23 m/s = 3. 3 x 10 m = 0. 2 kg

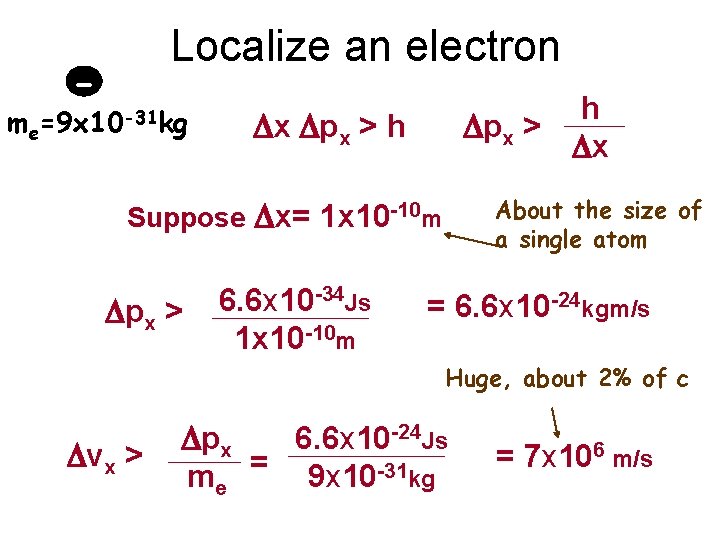
me Localize an electron - =9 x 10 -31 kg h Dpx > Dx Dx Dpx > h About the size of a single atom Suppose Dx= 1 x 10 -10 m Dpx > 6. 6 x 10 -34 Js 1 x 10 -10 m = 6. 6 x 10 -24 kgm/s Huge, about 2% of c Dvx > Dpx 6. 6 x 10 -24 Js me = 9 x 10 -31 kg = 7 x 106 m/s

uncertainty is inherent in the quantum world