Water Treatment Industrial Water Supplies Two Basic sources

Water Treatment

Industrial Water Supplies • Two Basic sources – Municipality – Private – Hybrid • No source provide pure water • Impurities Variation – Character – Amount

General Impurities • • • Dissolved Mineral Matter Dissolved Gases Turbidity and Sediment Color and Organic Matter Tastes and Odour Microorganisms

Safe drinking water Free from pathogenic organisms Clear Not saline Free from offensive taste or smell Free from compounds that may have adverse effect on human health • Free from chemicals that cause corrosion of water supply systems • • •

WATER QUALITY PARAMETERS • Physical parameters • Chemical • Bacteriological

BUREAU OFINDIAN STANDARDS IS 10500 -1991 Parameters Disirable limit Permissible limit Colour Hazen unit 5 25 Turbidity-NTU 5 10 p. H 6. 5 -8. 5 Hardness (as Ca. CO 3)mg/l 0. 3 1 TDS 500 2000
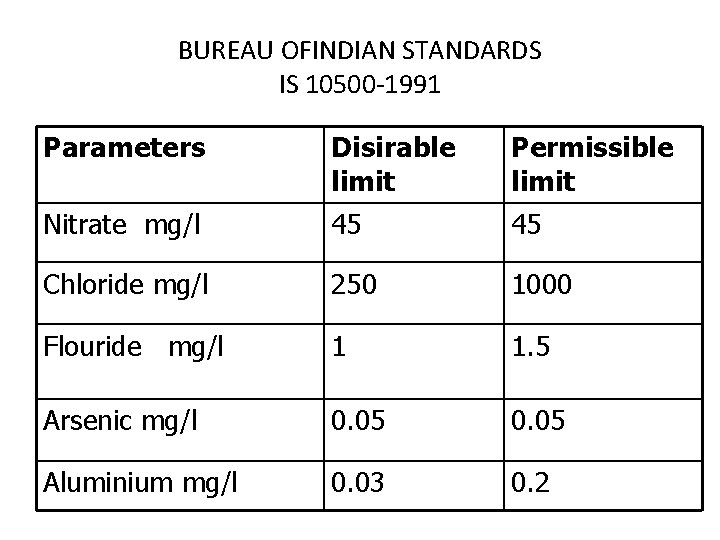
BUREAU OFINDIAN STANDARDS IS 10500 -1991 Parameters Disirable limit Permissible limit Nitrate mg/l 45 45 Chloride mg/l 250 1000 Flouride mg/l 1 1. 5 Arsenic mg/l 0. 05 Aluminium mg/l 0. 03 0. 2

Colour • May be due to the Presence of organic matter, metals(iron, manganese) or highly coloured industrial waste • Aesthetically displeasing • Disirable that drinking water be colourless • Disirable limit, 5 Hazen unit • Permissible limit 25 Hazen Unit

Taste and Odour • Mainly due to organic substances, , Biological activity, industrial pollution • Taste buds in the oral cavity specially detect inorganic compounds of metals like magnesium, calcium, sodium, copper, iron and zinc • Water should be free from objectionable taste and odour.

Turbidity • Caused by suspended matter • High level turbidity shield and protect bacteria from the action of disinfecting agents • Disirable limit-5 NTU should be below 1 NTU when disinfection is practiced Permissible limit-10 NTU

p. H • It is the measure of hydrogen ion concentration • Neutral water p. H-7 • Acidic water has p. H below 7 • Basic water has p. H above 7 • Disirable limit 6. 5 -8. 5 Beyond this limit the water will affect the mucous membrane and water supply system

Substances that change p. H of water Acidic Industries Sugar Distillery Electro. Plating unit • Pickle • • • Basic 5– 6 3 - 4 2. 5 -4 2 - 3 • • Paper 8 – 10 Textile 8. 5 -11 Fertiliser 6. 5 - 9 Oil Refineries 6. 5 -9. 5
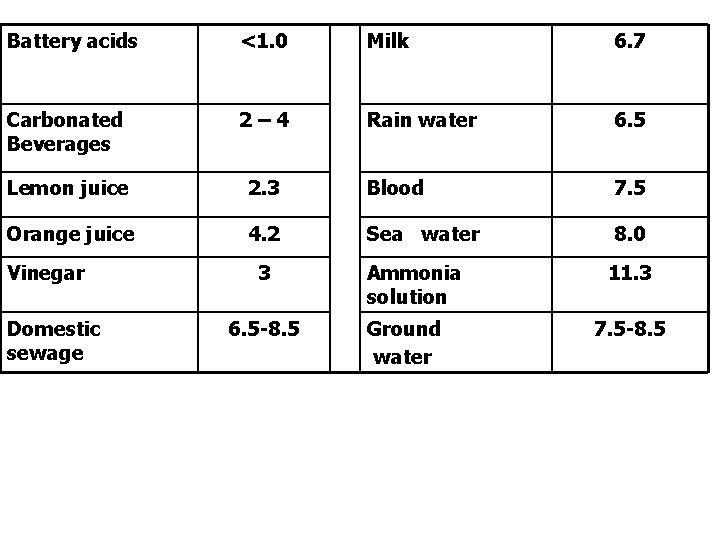
Battery acids <1. 0 Milk 6. 7 Carbonated Beverages 2– 4 Rain water 6. 5 Lemon juice 2. 3 Blood 7. 5 Orange juice 4. 2 Sea water 8. 0 Vinegar Domestic sewage 3 6. 5 -8. 5 Ammonia solution Ground water 11. 3 7. 5 -8. 5

Alkalinity • Quantitative Measure of Acid Neutralization – Acid Neutralizing Capacity – Indicates Basic p. H – Anions • Hydroxide • Carbonates • Bicarbonates • Titration – Different Indicators for Different Alkalinity • Phenolphthalein Vs Methyl Orange • Assignment# • “Methyl Organe Alkalinity is Total alkalinity”. This is not true for Phenolphthalein. • Assignment# ? ? ? – Applications of Alkalinity in commercial and domestic applications

HARDNESS • It is total concentration of calcium and magnesium ions • Temporary hardness – Bicarbonates of Calcium and Magnesium • Permanent hardness – Sulphates, chlorides and nitrates of calcium and magnesium

Hardness – contd… • • • 0 – 50 mg/l - soft 50 – 150 mg/l - moderately hard 150 – 300 mg/l - hard 300 above - very hard Surface water is softer than ground water Causes encrustations in water supply structures

Hardness • Cations – Sodium – Calcium – Magnesium – Silicon – Iron – Manganese – Aluminium
Hardness • Temporary Vs Permanent – Hydrogencarbonate – Le. Chatelier’s Principle – Thermally unstable • Formation of Precipitate – Solubility Curve • Clark’s Method • Your Home Kettle
Hardness. . • Temporary Vs Permanent – Other than bicarbonate • Chloride, • Nitrate • Sulphate – Thermally stable • Ion Exchange

Hard Water Softening Methods • Three Basic Methods – Membrane Filtration – Chemical Precipitation • Soda Process • Lime Soda Process – Cold Lime Soda – Hot Lime Soda – Ion Exchange • Units (Ca. CO 3)
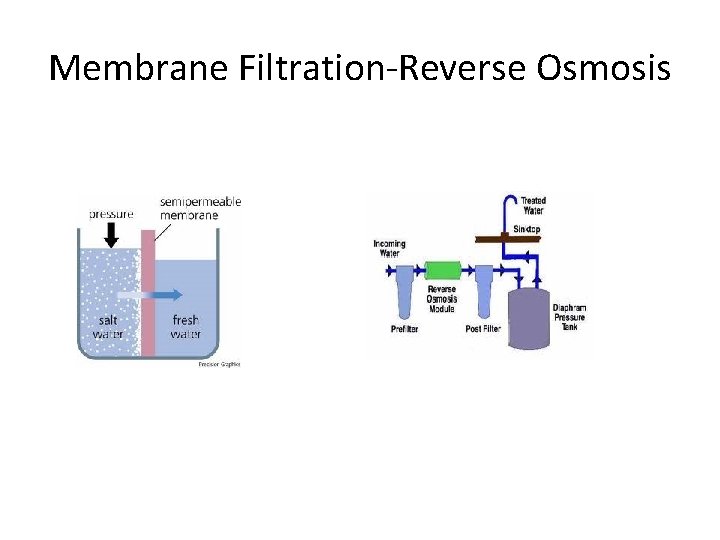
Membrane Filtration-Reverse Osmosis


Advantages • • Addition of No Chemicals Simple in Operation Safe (involves no hazardous chemicals) About 100% removal of hardness

Disadvantage • High Membrane Cost • Proper rejection of concentrated water • Require pretreatment

Chemical Precipitation Method • Chemical and Physical in Nature – Addition of chemicals to produce precipitates – Settling of sludge • Doesnot removal all the hardness • Reduces hardness 45 to 90 ppm of Ca. CO 3 • Provides Adequate corrosion protection to the distribution system

Permanent and Temporary Hardness • A sample of water has the following impurities in mg/liter. Find the temporary and permanent hardness in ppm. • Ca (HCO 3)2 = 10. 0 ppm • Mg (HCO 3)2 = 8 ppm • Ca. SO 4 = 12. 00 ppm • Mol. Wt. Ca (HCO 3)2 =162 • Mg (HCO 3)2 =146 • Ca. SO 4=136

Types • Soda Process (Caustic Soda) • Lime Process ( Ca. O or Ca(OH)2) • Lime-Soda Process – Cold Lime Process – Hot Lime Process

Soda Process • Involves the addition of Na. OH – Double Displacement Reaction • Production of Less Sludge • Costly

Lime Process • • Involves the addition of Ca. O Reaction with Ca(OH)2 Relatively Cheaper Produces extensive Sludge – Not preffered

Lime-Soda • Lime – Ca. O – Ca(OH)2 • Soda – Na 2 CO 3 • Optimization • Guess the Reactions? ? ?

Fundamental Process
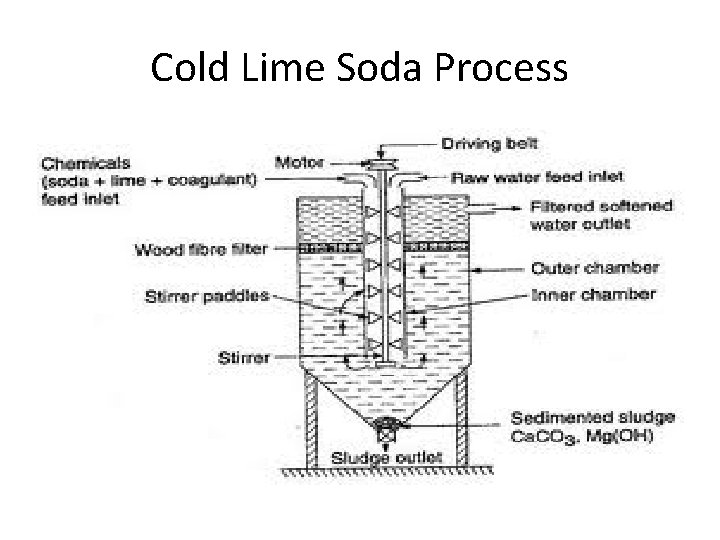
Cold Lime Soda Process
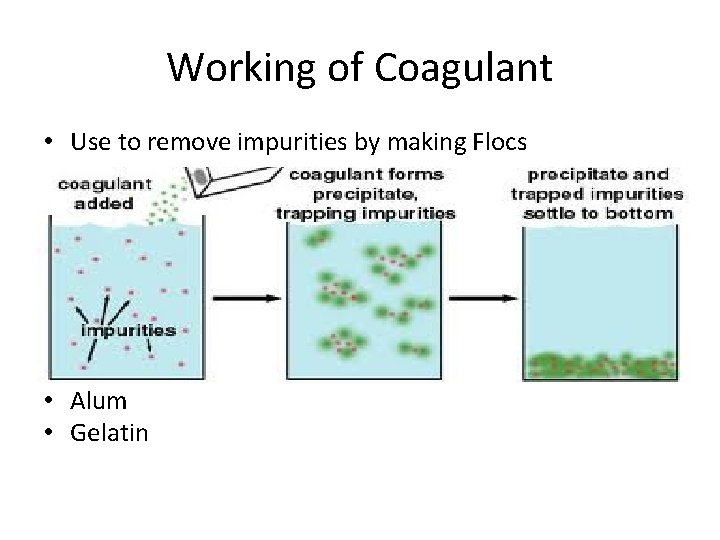
Working of Coagulant • Use to remove impurities by making Flocs • Alum • Gelatin
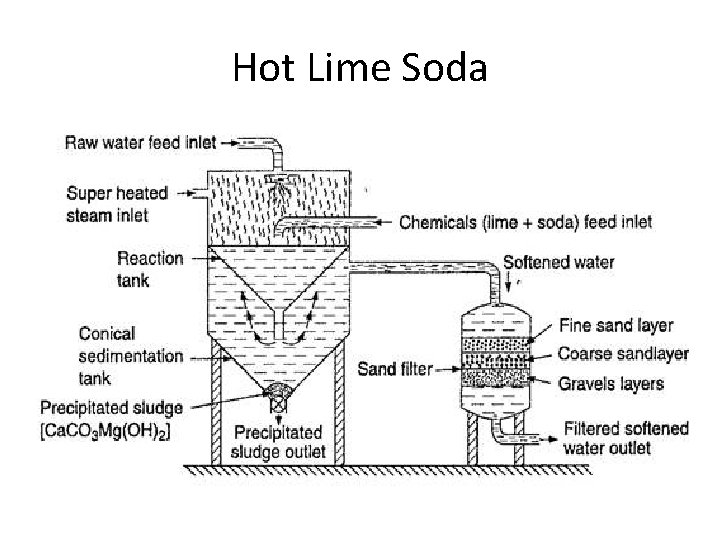
Hot Lime Soda

Advantages of Hot Lime-Soda • Precipitation Reaction Completion • Faster Reaction • Rapid Sludge Settling – Viscosity • Ease in Filtration • No Coagulant Needed • Degasification Better than Cold Lime
ION EXCHANGE

Basics • Involves Exchange of ions between an electrolyte and a complex • Solutions Vs mineral or polymeric ion exchangers • Typical Examples – Resin ( Functionalized Porous or gel polymer) – Zeolites – Clay

• Ion Exchange Resin – Insoluble – High molecular weight – Polymers – Porous Structure • Three Types – Cation – Anion – Amphoteric AMP 03
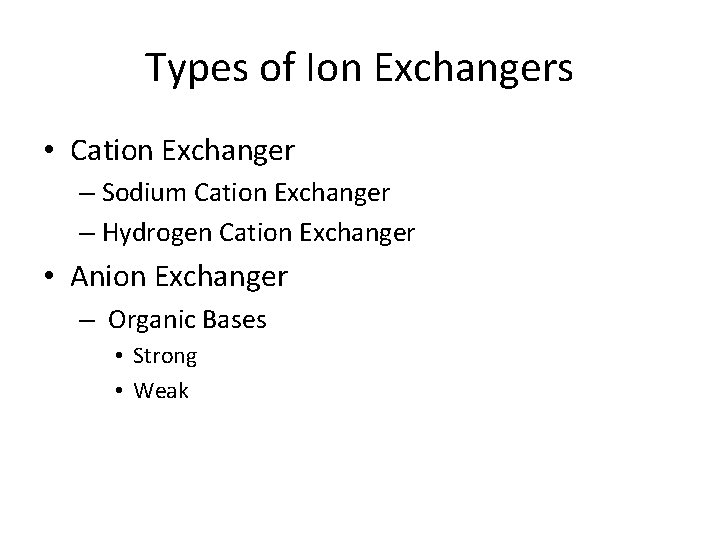
Types of Ion Exchangers • Cation Exchanger – Sodium Cation Exchanger – Hydrogen Cation Exchanger • Anion Exchanger – Organic Bases • Strong • Weak

Cation Exchanger • Removes Ca 2+ and Mg 2+ • Two Famous Types – Sodium Cation Exchanger – Hydrogen Cation Exchanger • Acidic Functional Groups – -COOH – -SO 3 H » Exchanges H+ – Sodium Cation Exchanger • Sodium Zeolites

Anion Exchanger • Removes Cl-, SO 42 -, NO 3 • Contains Basic Groups – Ammonium ions – Quaternary phosphorium ion • Capable of exchanging OH- ions

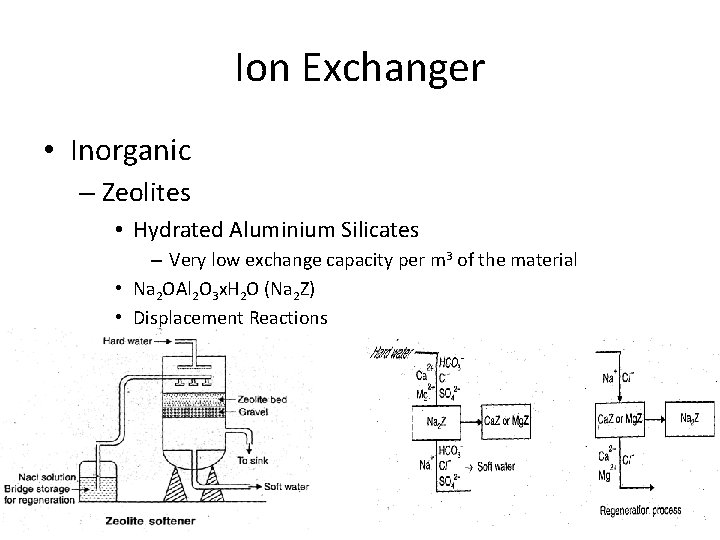
Ion Exchanger • Inorganic – Zeolites • Hydrated Aluminium Silicates – Very low exchange capacity per m 3 of the material • Na 2 OAl 2 O 3 x. H 2 O (Na 2 Z) • Displacement Reactions

• Organic – Very high exchange capacity – Sulphonated natural products like coal, lignite – Polystyrene-Divinylbenzene
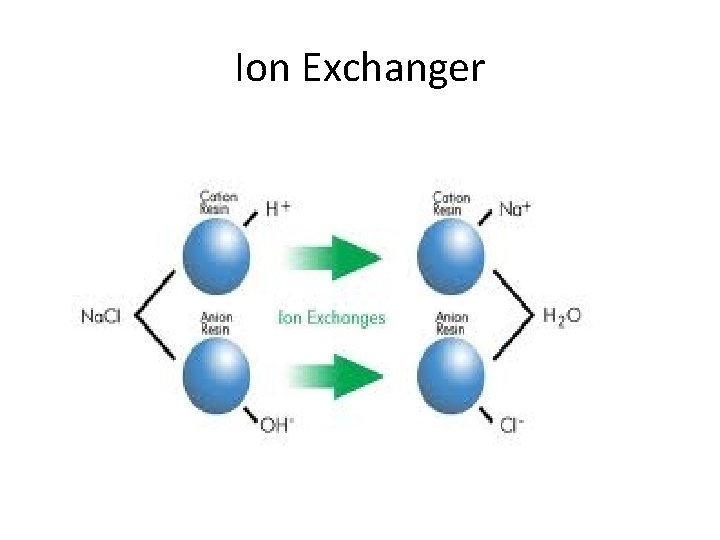
Ion Exchanger


IMPORTANT PARTICLE TECHNOLOGY
Crusher

Ball Mill
Filtration
Sedimentation
Centrifugal Separation

Magnetic Separation
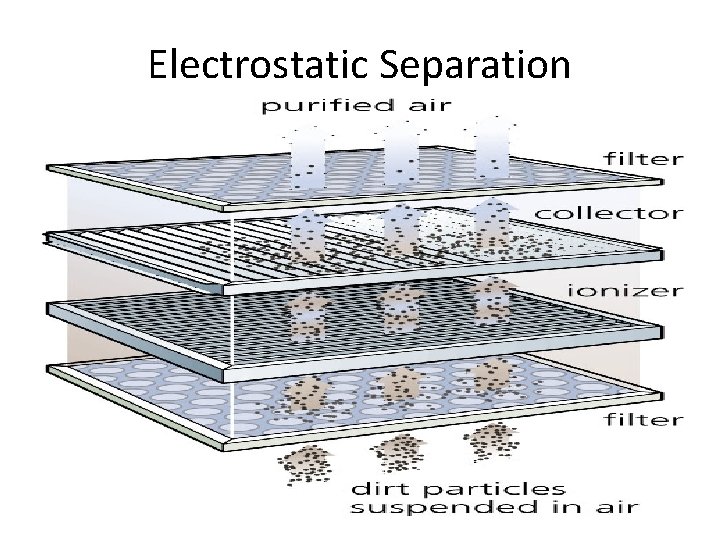
Electrostatic Separation
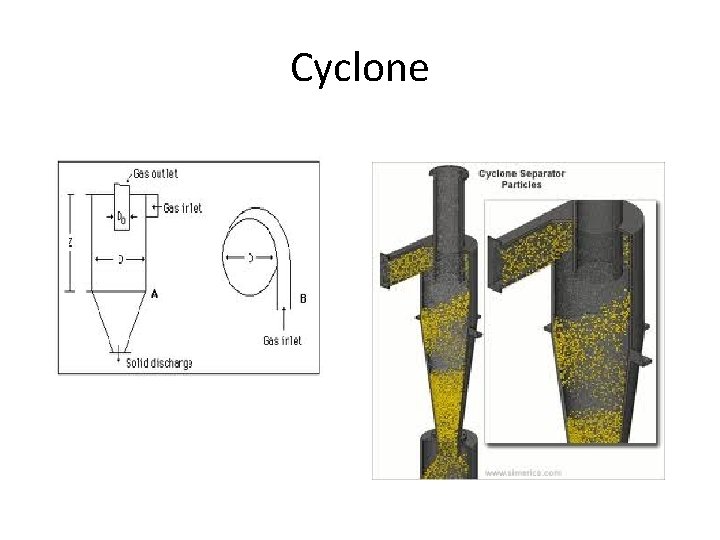
Cyclone
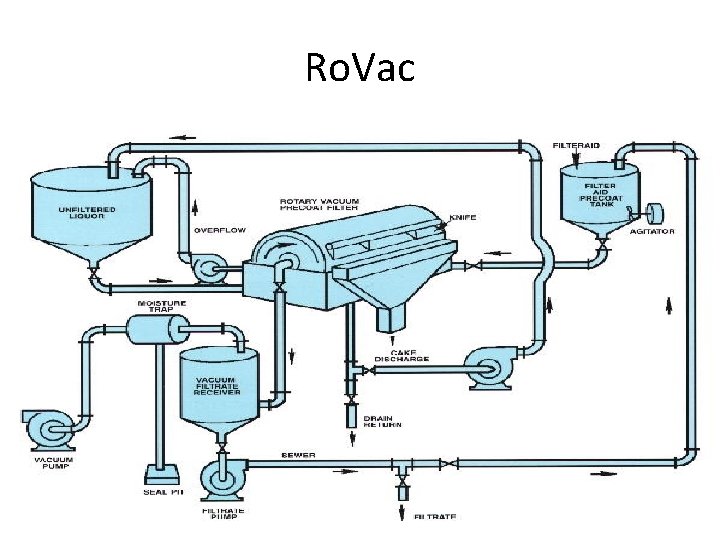
Ro. Vac
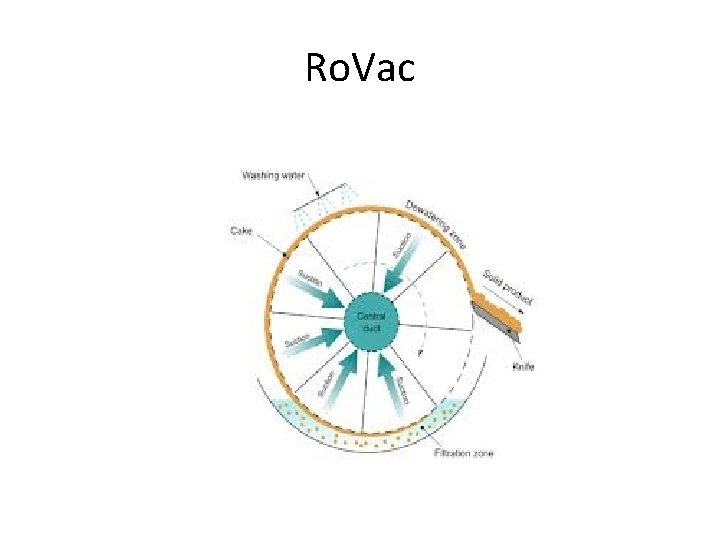
Ro. Vac
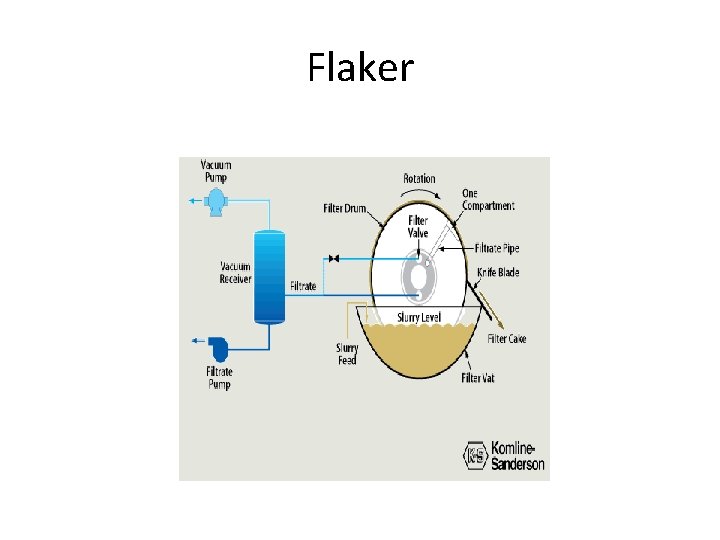
Flaker
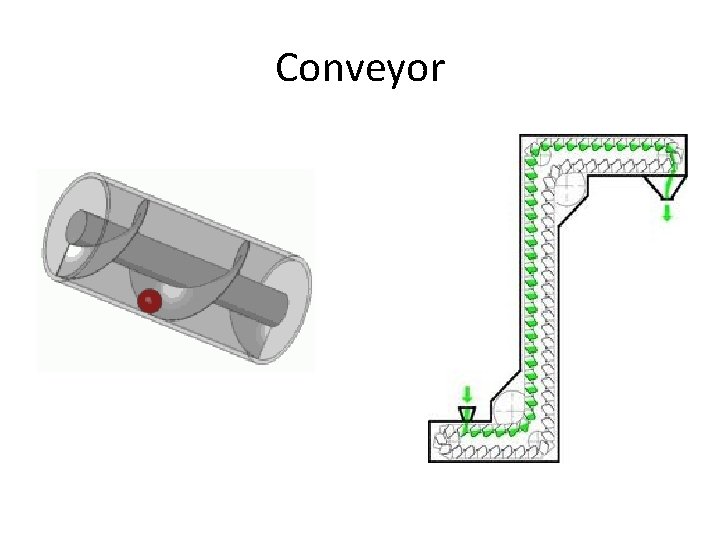
Conveyor

Crystallizer

HEAT TRANSFER
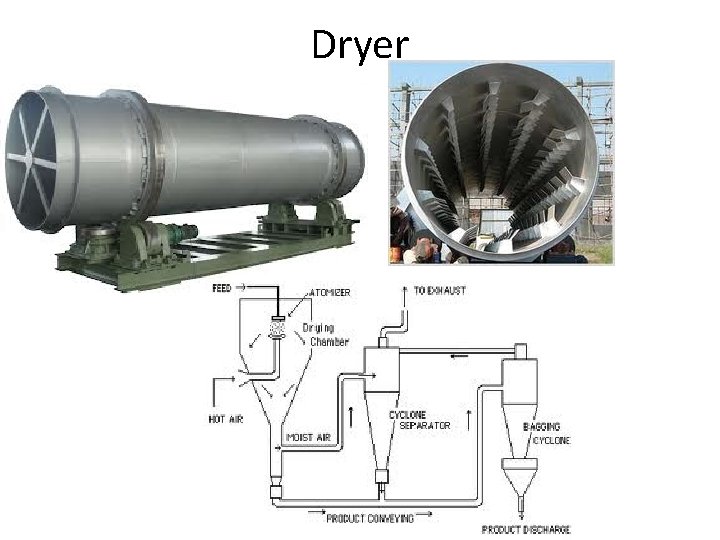
Dryer
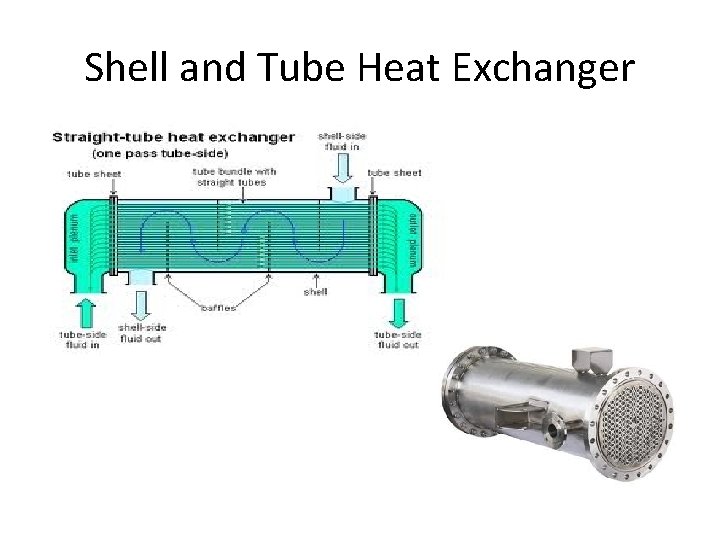
Shell and Tube Heat Exchanger

Spiral Tube H. E. Plate and Frame Heat Exchanger
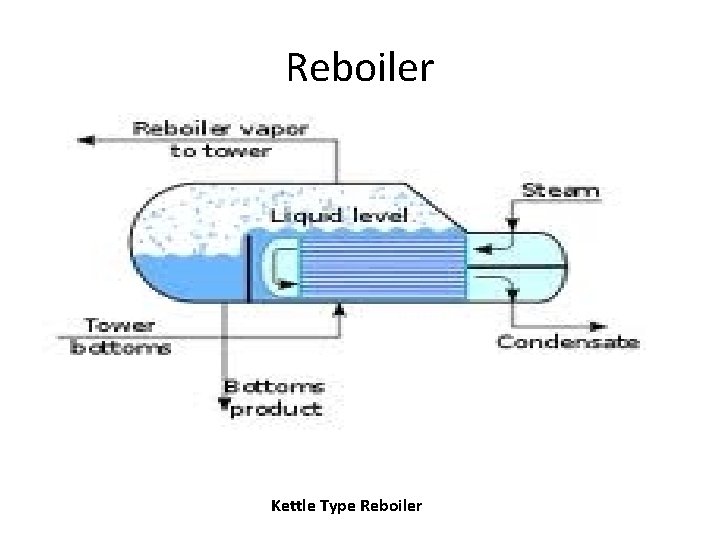
Reboiler Kettle Type Reboiler
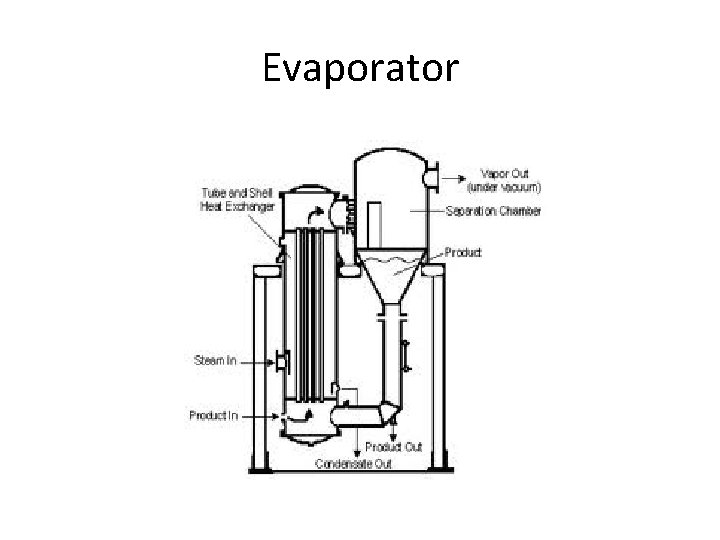
Evaporator
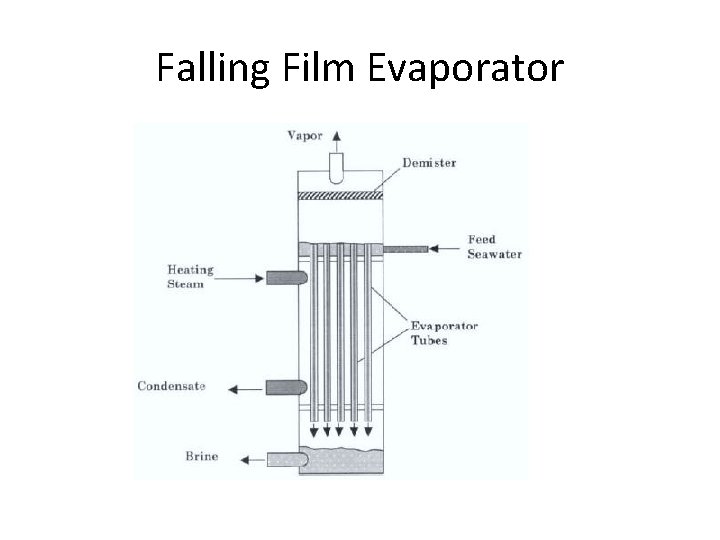
Falling Film Evaporator
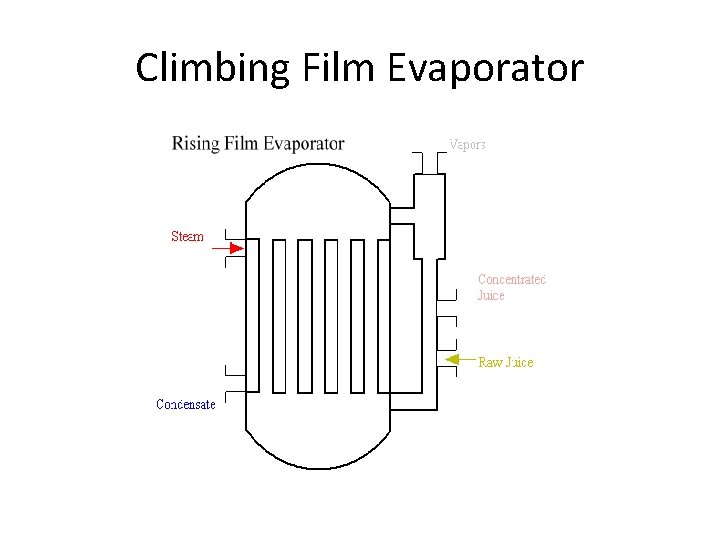
Climbing Film Evaporator

Multiple Effect Evaporators

Mass Transfer
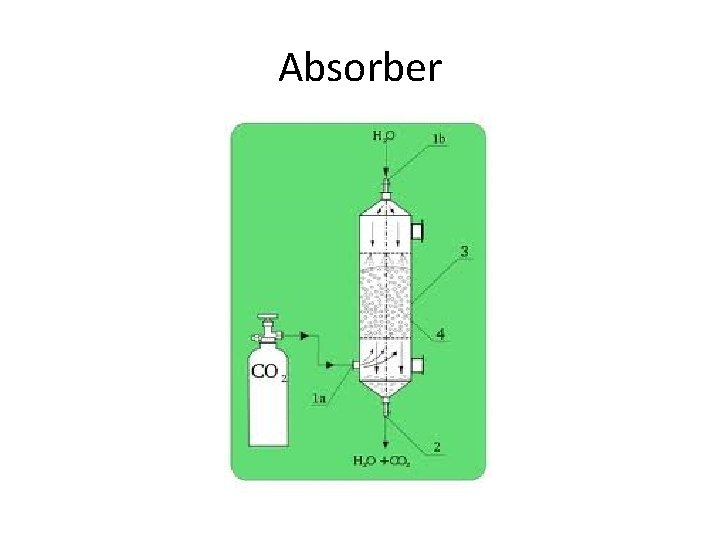
Absorber
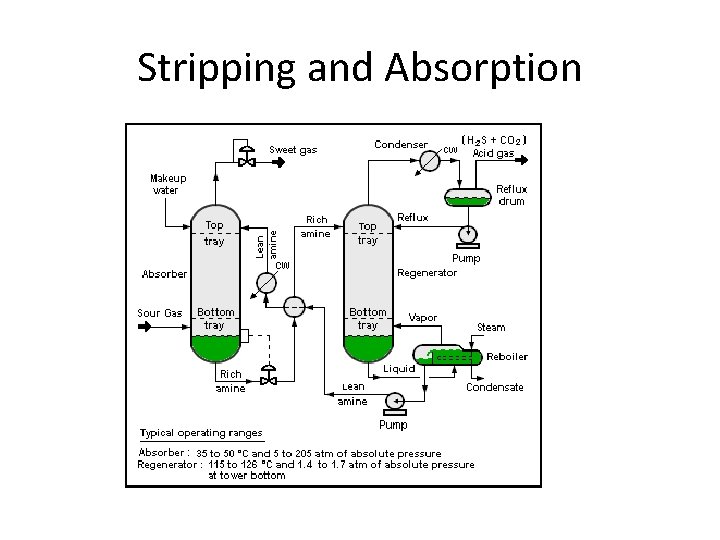
Stripping and Absorption
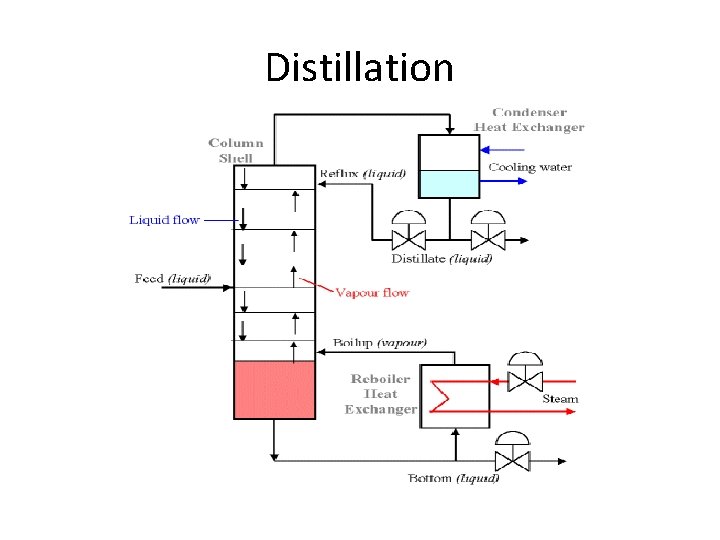
Distillation

Column Internals (Plates and Packing)

Bubble Cap
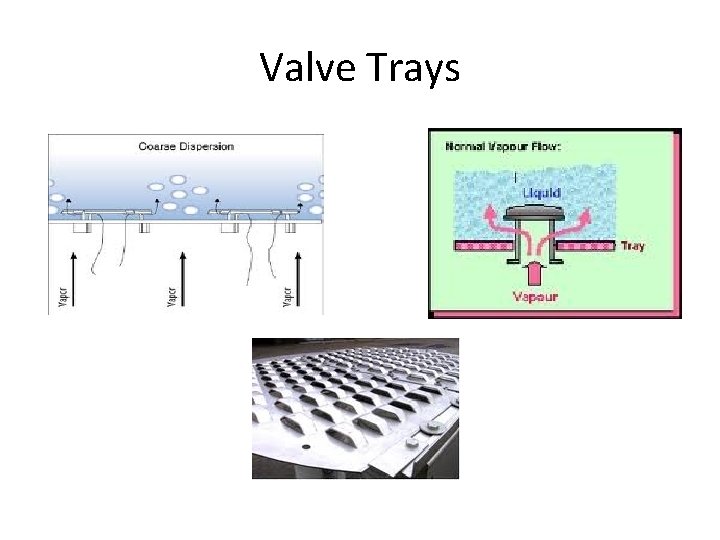
Valve Trays

Sieve Trays

Packing

Reactors

Reactors • Two basic types – Countinuous Stirred Tank Reactor (CSTR) • MFR – Plug Flow Reactor (PFR)
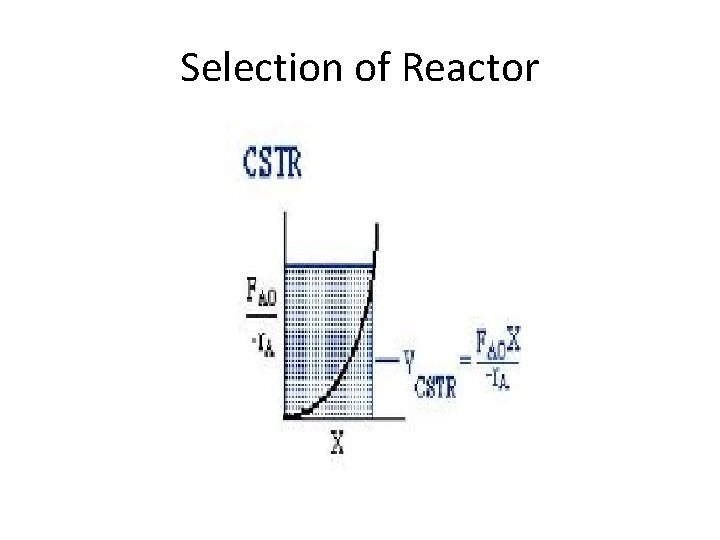
Selection of Reactor
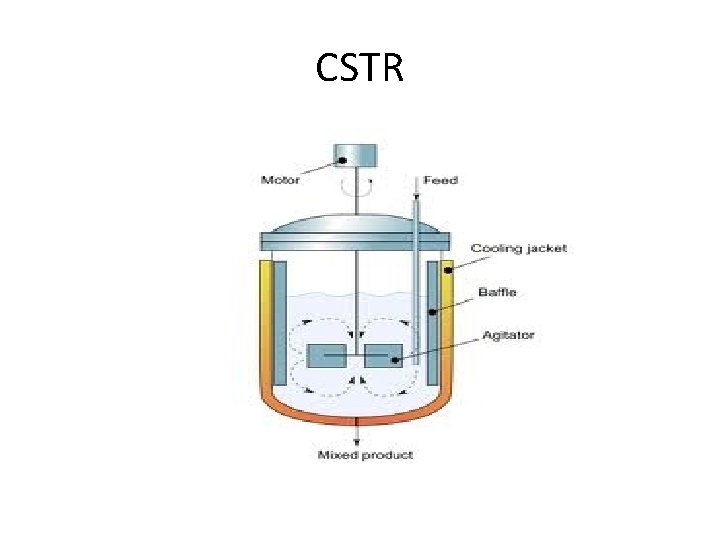
CSTR
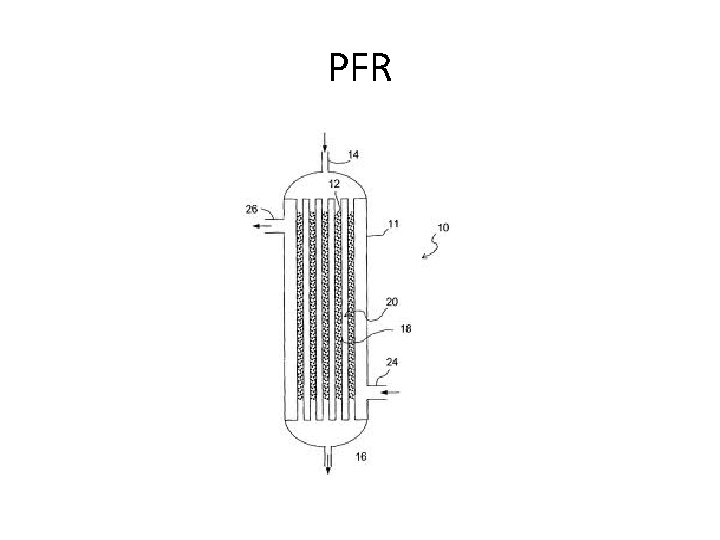
PFR
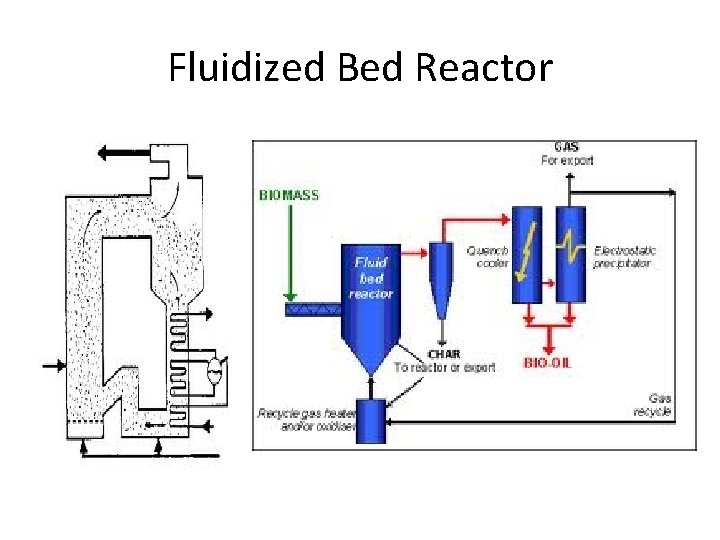
Fluidized Bed Reactor
- Slides: 84