Water The Facts Water is the most important














- Slides: 16


Water: The Facts ©Water is the most important molecule in living organisms. ©Water consists of 1 atom of oxygen combined with 2 atoms of hydrogen. ©Water makes up 70 to 95 percent of most organisms. H H

What are the characteristics of water? 1. Water is polar. 2. Water molecules attract other water molecules. This property is cohesion. 3. Water displays adhesion. 4. Water has a high specific heat. 5. Water expands when it freezes.

1. Water is Polar © A polar molecule has an unequal distribution of charge -each molecule has a positive end a negative end. © All polar substances are attracted to other polar substances.

Why is this polarity important? © Polar water molecules attract other water molecules as well as ions and other polar molecules. © Because of this attraction, water can dissolve many ionic compounds, such as salt and polar molecules like sugar. © Water is known as the universal solvent (almost anything can be dissolved by water)

©Solvent= what does the dissolving (water) ©Solute= what is being dissolved (sugar, salt) ©Solvent +solute=solution (sugar water) or (salt water)

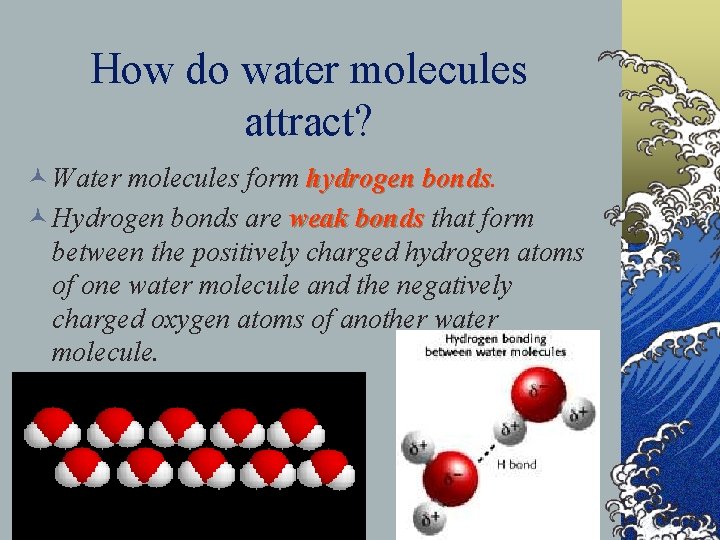
How do water molecules attract? © Water molecules form hydrogen bonds © Hydrogen bonds are weak bonds that form between the positively charged hydrogen atoms of one water molecule and the negatively charged oxygen atoms of another water molecule.

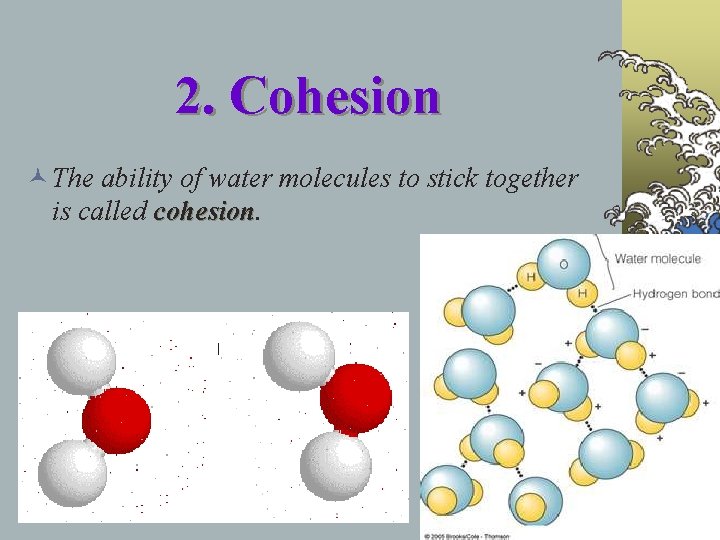
2. Cohesion © The ability of water molecules to stick together is called cohesion

Cohesion © Results in Surface tension (a measure of the strength of water’s surface) © Produces a surface film on water that allows insects to walk on the surface of water

3. Adhesion © Water displays the property of adhesion which is the ability of one substance to cling to another. © Examples of adhesion include water’s ability to creep up thin tubes. Plants show this property when they send water from the soil up through their tissues.

Adhesion © Water will make hydrogen bonds with other surfaces such as glass, soil, plant tissues, and cotton. © Capillary action-water molecules will “tow” each other along when in a thin glass tube. © Example: transpiration process which plants and trees remove water from the soil, and paper towels soak up water.

4. High Specific Heat © Water resists temperature change, both for heating and cooling. © In order for water to evaporate, hydrogen bonds must be broken. © As water evaporates, it removes a lot of heat with it. © Water vapor forms a kind of global ‘‘blanket” which helps to keep the Earth warm.

5. Water expands when it freezes. © Because of this property, ice is less dense than liquid water. © This property can be seen when ice forms and floats on the surface of a pond.

Water expands when it freezes. ©Liquid water has hydrogen bonds that are constantly being broken and reformed. ©Frozen water forms a crystal-like lattice whereby molecules are set at fixed distances.

Water is Less Dense as a Solid! • Which is ice and which is water?

Water is Less Dense as a Solid! Water Ice