Water Science Electroneutrality p H Alkalinity Acidity Reversible

Water Science • Electroneutrality, p. H, Alkalinity, Acidity • Reversible Equations • Carbonate Equilibrium System • Examples • Rain • Ocean p. H • Acid Rain • Acid Mine Drainage naturescrusaders. files. wordpress. com

Water Electroneutrality • Balanced: ∑ Cations = ∑ Anions • ∑ CC x e C = ∑ CA x e A • CC = moles / liter of cation c (one mole = 6. 02 x 1023) • e. C = number of charges per molecule of cation C • A subscript is for anions • Pure water: [H+] = [OH-] • Natural water: [H+] = [OH-] + [HCO 3 -] + 2[CO 3 -2] blog. ssis. edu. vn/

p. H - reporting H+ concentration • p. H = - log {H+} • {} = activity, the effective concentration • p. H = - log [H+] for dilute solutions • [] = Molar concentration (moles/Liter) • [H+] = 10 -p. H www. craterlakeoregon. org/

Why do we care about p. H? • Biological systems • Extremes are disruptive • Corrosivity • Equilibrium relationships involving H+ www. safetrack. se/

Good p. H ranges • Freshwater. . . • Marine. . . • Drinking. . . • Soda?

Alkalinity • Capacity of water sample to neutralize an. . . • Capacity of a water sample to take H+ without significant change in. . . • Common ions that give water alkalinity • HCO 3 -, CO 32 - & OH-

Acidity • Capacity of water to take OH- without significant p. H change • Opposite of Alkalinity • Capacity of water sample to neutralize a base • Common ion: H+ • For Acid Mine Drainage • Acidity =~f(Fe 2+, Fe 3+, Al, Mn, and H+) csmphotos. files. wordpress. com

p. H, Alkalinity & Acidity • Waters with same p. H can have different amounts of Alkalinity or Acidity • Chemicals may be present that can react with or release H+ or OH- • The lower the alkalinity (or acidity), the easier it is to change p. H

Reversible Reactions • a. A + b. B ↔ c. C + d. D • A & B can react to form C and D • C & D can react to form A and B • After sufficient time has passed, equilibrium is reached • Equilibrium can be perturbed by adding more reactant or product
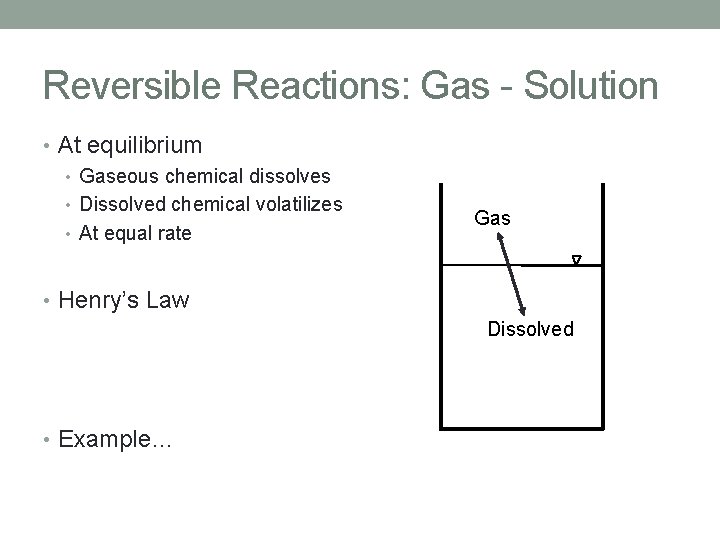
Reversible Reactions: Gas - Solution • At equilibrium • Gaseous chemical dissolves • Dissolved chemical volatilizes • At equal rate Gas • Henry’s Law Dissolved • Example…

Reversible Reactions In Solution • At equilibrium • Reactants become products • Products become reactants • At equal rate • Example…

Reversible Reactions: Solution - Precipitate • At equilibrium • Precipitate is formed • Precipitate is dissolved • At equal rates www. struvite. info/ • Example…

Equilibrium Equation a. A + b. B ↔ c. C + d. D K is constant at given temperature & relatively low concentration

Example - Pure Water • What is water? . . . • Reversible equation? . . . • Equilibrium equation? . . . • At 25 C, Kw =. . . • Even if other sources of H+ (acids) or OH- (bases) are present • Which substance predominates? . . .

p. H of Pure Water @ 25 C? • Follow these steps • Write down species. . . • Write electroneutrality eq. . . • Write equilibrium equation. . • Solve equations. . .

Now, add… • OH- ? (add a strong base) • Systems finds new equilbrium, with less H+ and more OH • p. H goes up • H+ ? (add a strong acid, precipitate iron, …) • Systems finds new equilbrium, with more H+ and less OH • p. H goes down. • Either way, [H+] [OH-] = Kw still holds

Precipitation of Limestone • Ca. CO 3 -2 ↔ Ca+2 + CO 3 -2 • [Ca+2] [CO 3 -2] = Ksp • Ksp = Solubility product • Do not include precipitate in equilibrium equation • [Ca+2] [CO 3 -2] < Ksp? . . . • [Ca+2] [CO 3 -2] > Ksp? . . .
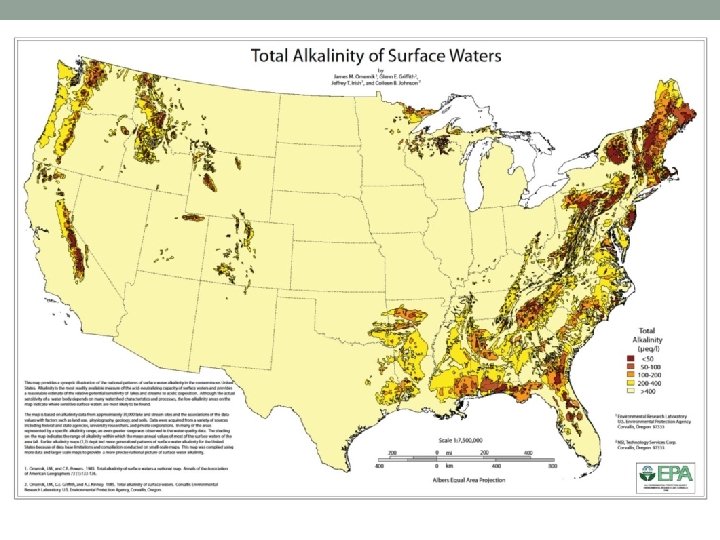

Carbonate Equilibrium System (CES) • Keeps natural waters in good p. H range • Common Chemicals • Carbon Dioxide, CO 2 • Carbonic Acid, H 2 CO 3 • Bicarbonate, HCO 3 • Carbonate, CO 3 -2 • Solids containing Bicarbonate, Carbonate, Hydroxide, and (though rarely) certain Oxides (Ca. O), …
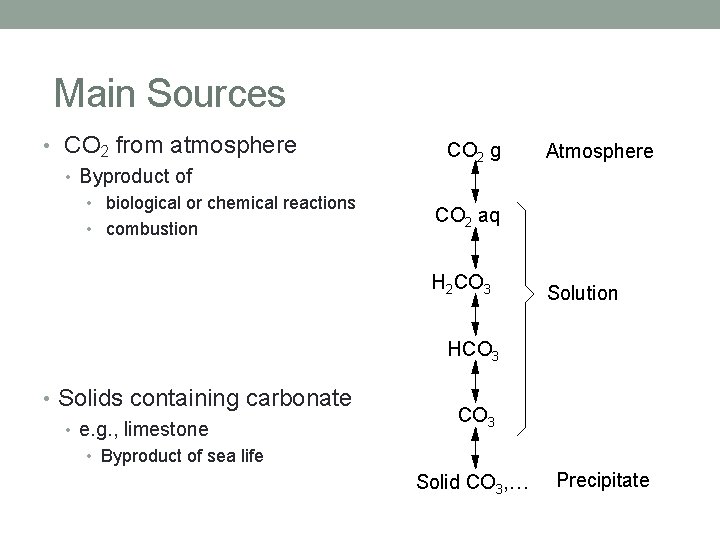
Main Sources • CO 2 from atmosphere • Byproduct of • biological or chemical reactions • combustion CO 2 g Atmosphere CO 2 aq H 2 CO 3 Solution HCO 3 • Solids containing carbonate • e. g. , limestone CO 3 • Byproduct of sea life Solid CO 3, … Precipitate

CES Reactions Reversible Equilibrium (at 25 C) • CO 2 g ↔ CO 2 aq • CO 2 aq = CO 2 g / 1637 atm • CO 2 aq + H 2 O ↔ H 2 CO 3 • [H 2 CO 3] / [CO 2 aq] = 1. 58 x 10 -3 • H 2 CO 3 ↔ H+ + HCO 3 - • [H+] [HCO 3 -] / [H 2 CO 3*] = 4. 47 x 10 -7 M • HCO 3 - ↔ H+ + CO 3 -2 • [H+] [CO 3 -2] / [HCO 3 -] = 4. 68 x 10 -11 M • Ca+2 + CO 3 -2 ↔ Ca. CO 3 • [Ca+2] [CO 3 -2] = 3. 80 x 10 -9 M 2 Where CO 2 g is in atm & CO 2 aq is in mole fraction
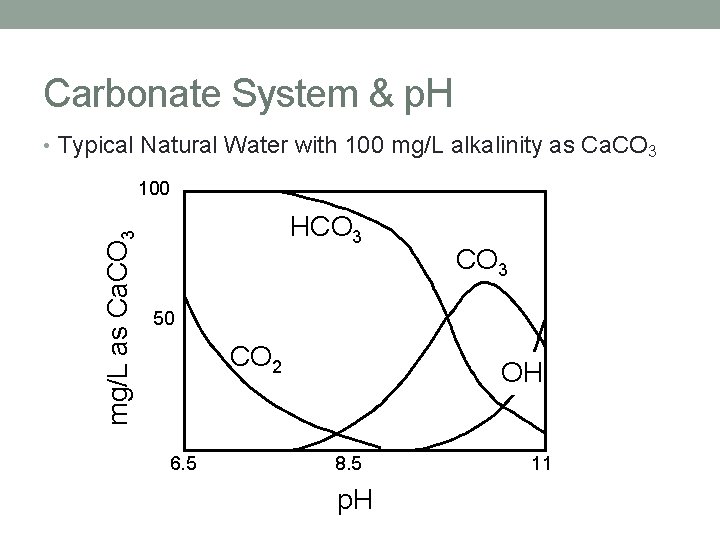
Carbonate System & p. H • Typical Natural Water with 100 mg/L alkalinity as Ca. CO 3 mg/L as Ca. CO 3 100 HCO 3 50 CO 2 6. 5 OH 8. 5 p. H 11

Carbonate System Scenarios • Open / Closed • Open - in equilibrium with atmosphere • open container, streams & shallow lakes, upper ocean • Closed - not in equilibrium • Closed tank, quick reactions, deep regions of water bodies, soil gases • Homogeneous / Heterogeneous • Homogeneous – in equilibrium with CO 3 solids • Heterogeneous– not in equilibrium with CO 3 solids • Closed & heterogeneous • bottom of stratified lake with floor of limestone • water treatment unit with soda ash / acid or base / CO 2 & some Ca. CO 3 precipitates
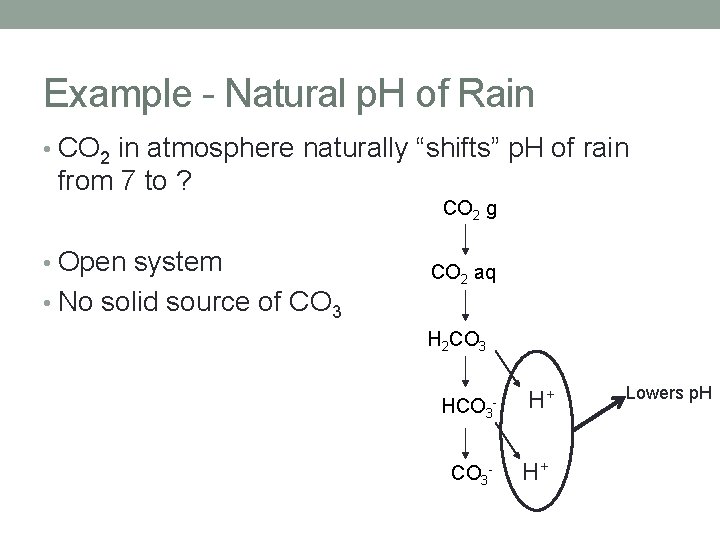
Example - Natural p. H of Rain • CO 2 in atmosphere naturally “shifts” p. H of rain from 7 to ? CO 2 g • Open system • No solid source of CO 3 CO 2 aq H 2 CO 3 HCO 3 - - H+ H+ Lowers p. H
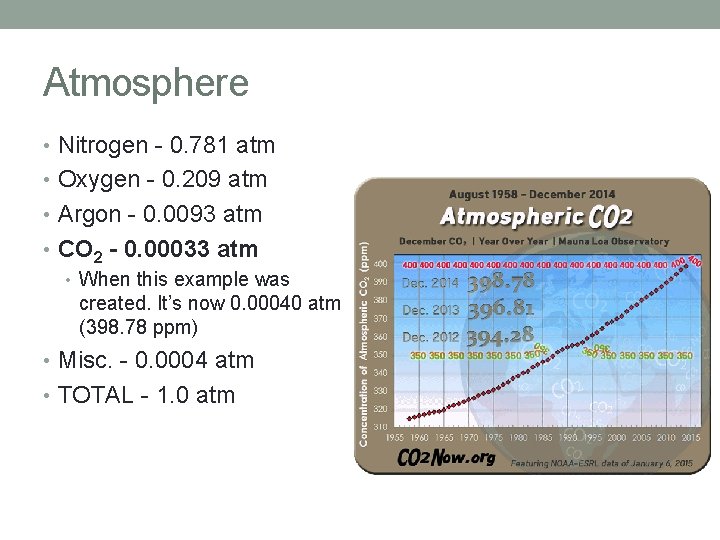
Atmosphere • Nitrogen - 0. 781 atm • Oxygen - 0. 209 atm • Argon - 0. 0093 atm • CO 2 - 0. 00033 atm • When this example was created. It’s now 0. 00040 atm (398. 78 ppm) • Misc. - 0. 0004 atm • TOTAL - 1. 0 atm

Solution: p. H of Rain (1) • Atmosphere to raindrop - Henry’s Law: CO 2 aq = CO 2 g / 1637 atm • • Convert to M: [CO 2 aq] = CO 2 aq x Mw • Mw = molar density of water = 55. 56 mol/l •

Solution: p. H of Rain (2) • Reaction with water forms carbonic acid - Equilibrium Equation: [H 2 CO 3 aq] = 1. 58 x 10 -3 M • [CO 2 aq] • • [H 2 CO 3* aq] = [CO 2 aq] + [H 2 CO 2 aq] •

Solution: p. H of Rain (3) • Dissociation of carbonic acid - Equilibrium Equation: [H+] [HCO 3 -] = 4. 47 x 10 -7 M • [H 2 CO 3*] • Two unknowns? Use electroneutrality: [H+] = [OH-] + [HCO 3 -] + 2[CO 3 -2] • Rain water is acidic, so [OH-] & [CO 3 -2] will be small giving [H+] ≈ [HCO 3 -], leading to… • •
![Solution: p. H of Rain (4) • p. H = - log [H+] = Solution: p. H of Rain (4) • p. H = - log [H+] =](http://slidetodoc.com/presentation_image_h2/47e080d5a9a1b8dd20be569a698839be/image-29.jpg)
Solution: p. H of Rain (4) • p. H = - log [H+] = -log (2. 13 x 10 -6) =… • Check assumptions? Use equilibrium equations: • [OH-] = 10 -14 M 2 / [H+] = 10 -14 M 2 / 2. 13 x 10 -6 M = 4. 68 x 10 -9 M (small, OK)) • [CO 3 -2] = 4. 68 x 10 -11 M • [H+] / [HCO 3 -] = 4. 68 x 10 -11 M • 2. 13 x 10 -6 M / 2. 13 x 10 -6 M = 4. 68 x 10 -11 M (small, OK)

p. H of Rain • CO 2 in atmosphere goes up? . . . • • Use this method for different conditions? • Need to check assumptions about relative concentration of [OH-] & [CO 3 -2]

Atmospheric CO 2 & Ocean p. H • Oceans can be CO 2 sink or source • Currently: sink, taking ~ 30% of anthropocentric CO 2 • Primary mechanisms • Carbonate equilibrium system (Henry’s Law, …) • Biological Pump • Living creatures take up carbon, some are trapped in sea bed • Too much CO 2? • p. H drop of oceans could effect sea organisms • From 1751 to 1994 surface ocean p. H estimated to have dropped from ~ 8. 18 to 8. 10

Ocean p. H Change • ~half Anthropogenic CO 2 has been absorbed by ocean so far • Currently absorbing ~1/3 • 22 M tons / day • Could drop to 7. 6 • Drastic effect on shell-forming organisms National Geographic (2014) “Ocean Acidification”, ocean. nationalgeographic. com.

Acid Rain • Rain with p. H below 5. 7 • Most prevalent cause: • SOx produced from burning coal • SOx reacts with water to form sulfuric acid • Sulfuric acid dissolves into atmospheric water and dissociates into H- and SO 4 -2
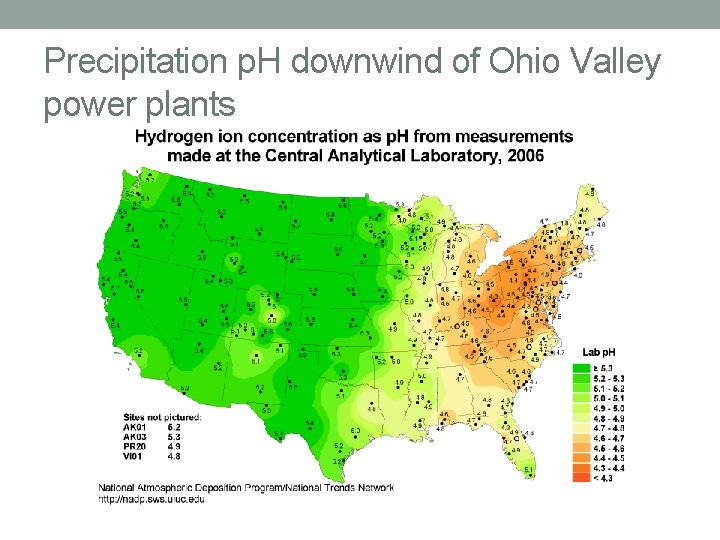
Precipitation p. H downwind of Ohio Valley power plants
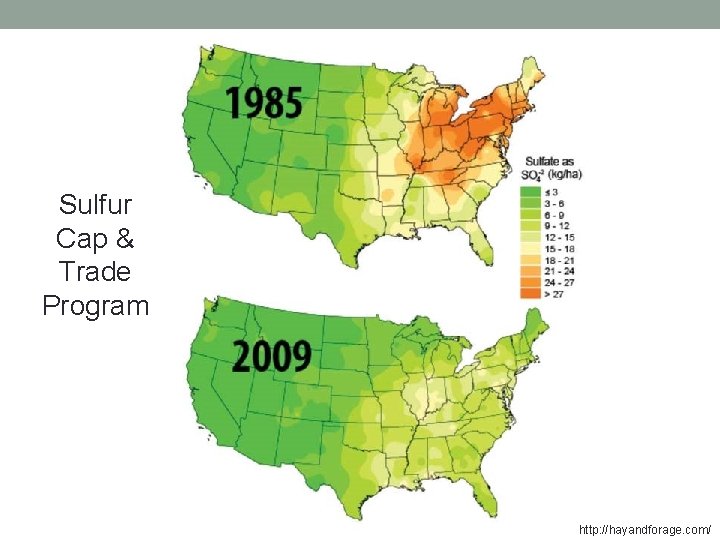
Sulfur Cap & Trade Program http: //hayandforage. com/

CES, Alkalinity, Acid Rain’s Effect • Carbonate solids • Common source of alkalinity in natural waters (as they dissolve) • Lakes and streams with alkalinity can accept acid rain without big p. H changes • Alkalinity buffers acid rain • Acid rain can damage water bodies that don’t have alkalinity
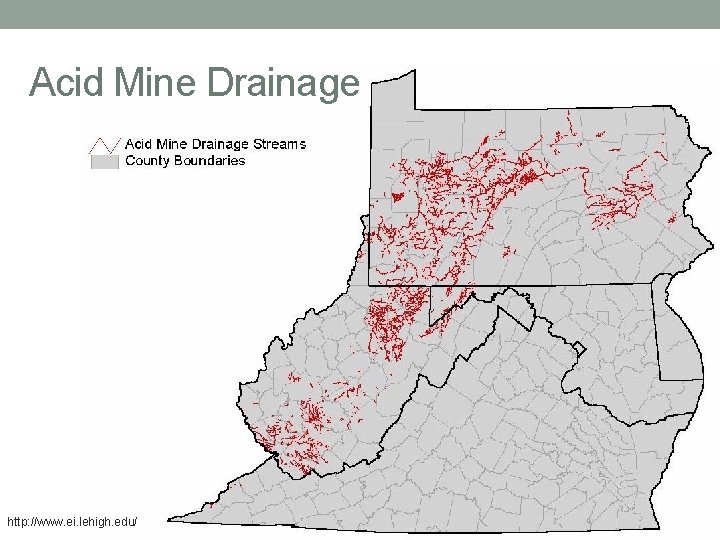
Acid Mine Drainage http: //www. ei. lehigh. edu/
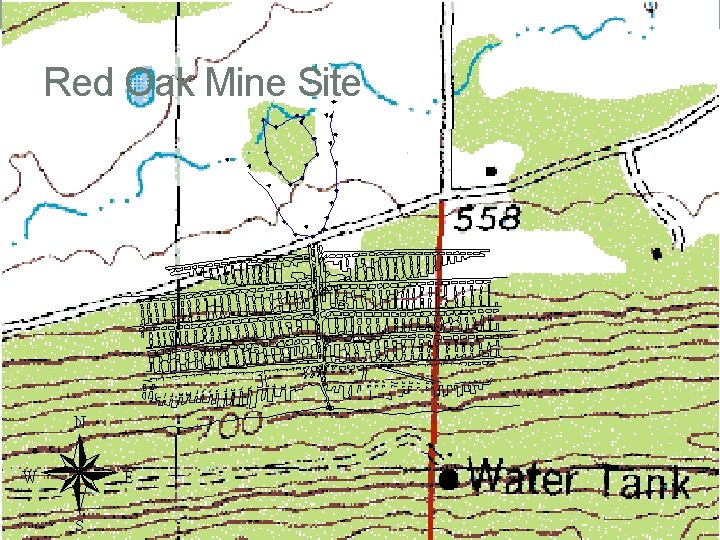
Red Oak Mine Site
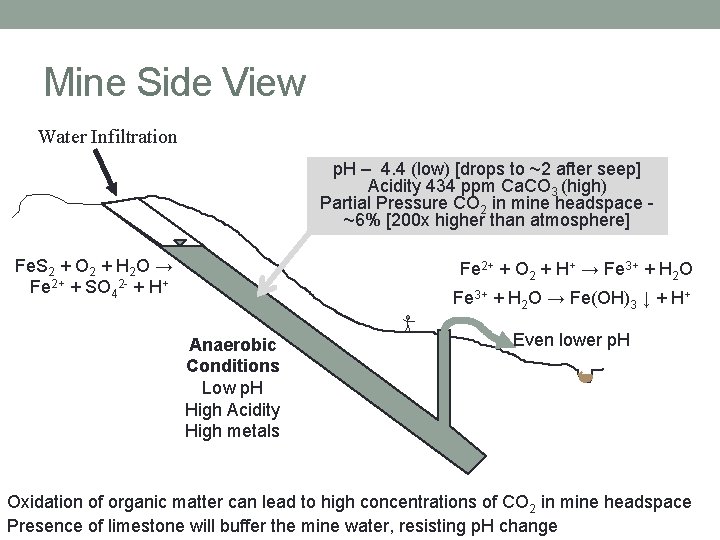
Mine Side View Water Infiltration p. H – 4. 4 (low) [drops to ~2 after seep] Acidity 434 ppm Ca. CO 3 (high) Partial Pressure CO 2 in mine headspace ~6% [200 x higher than atmosphere] Fe. S 2 + O 2 + H 2 O → Fe 2+ + SO 42 - + H+ Fe 2+ + O 2 + H+ → Fe 3+ + H 2 O → Fe(OH)3 ↓ + H+ Anaerobic Conditions Low p. H High Acidity High metals Even lower p. H Oxidation of organic matter can lead to high concentrations of CO 2 in mine headspace Presence of limestone will buffer the mine water, resisting p. H change

Red Oak Seep seep discharge
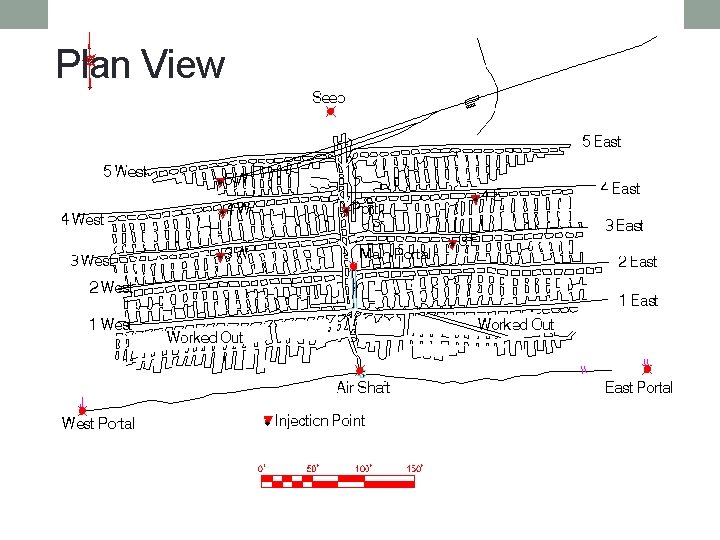
Plan View Not to scale

Red Oak Remediation Post injection, seep characteristics were p. H - 6. 3, Alkalinity - 150 mg/L as Ca. CO 3, Fe- 120 mg/l; however, this level of treatment lasted only 15 months.
- Slides: 42