WATER RELATIONS Water potential Osmotic potential Pressure potential







- Slides: 18

WATER RELATIONS • • • Water potential Osmotic potential Pressure potential Matric potential Absorption and translocation of water Stomatal regulation.

Water Potential • • • Difference b/w free energy of water in that system and free energy of pure water at atmospheric pressure and a defined temperature. Water potential is the potential energy of water per unit volume relative to pure water in reference conditions. Water potential quantifies the tendency of water to move from one area to another due to osmosis, gravity, mechanical pressure and matrix effects such as capillary action

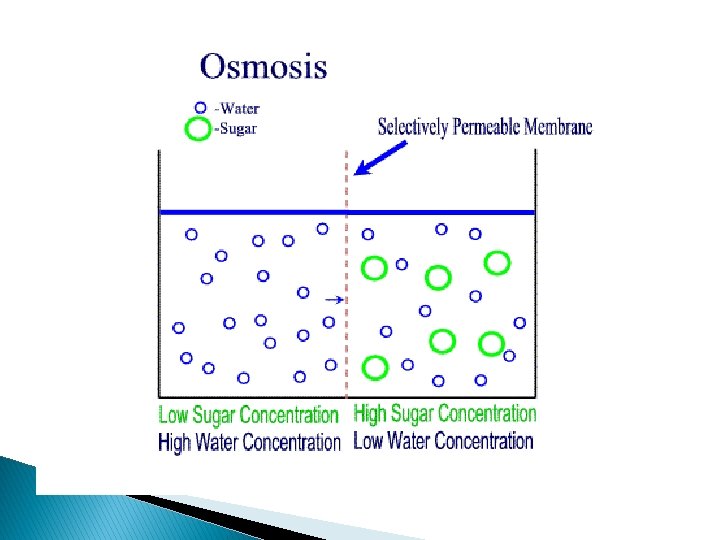
• • • Unit of measurement: Energy units, Joules per m 3, Pascals Pure water =0 Adding solute lowers potential Less free water molecules Water moves from a higher water potential to a lower water potential Less concentrated (hypotonic) to a more concentrated (hypertonic)

Osmosis and Diffusion Osmosis � Movement of water molecules from a region of higher water potential to a region of lower water potential through a semipermiable membrane. Diffusion • Net movement from one point to another because of random kinetic activities of molecules or ions from a region of their own higher concentration to a region of their lesser concentration. • It is spontaneous process.


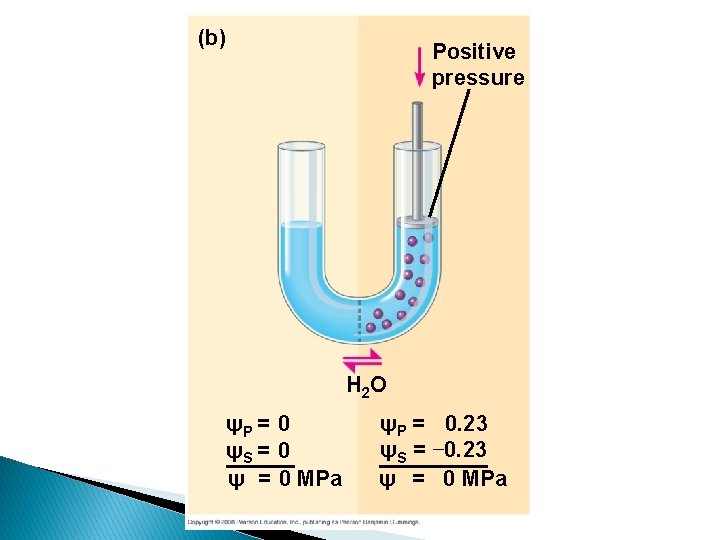
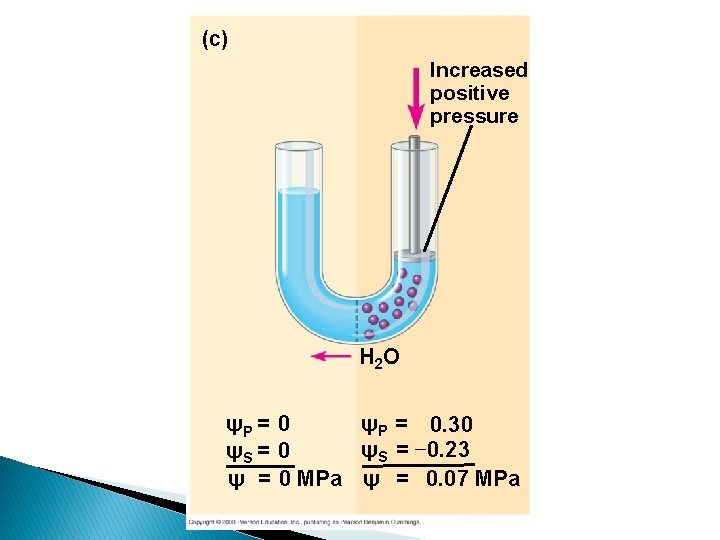
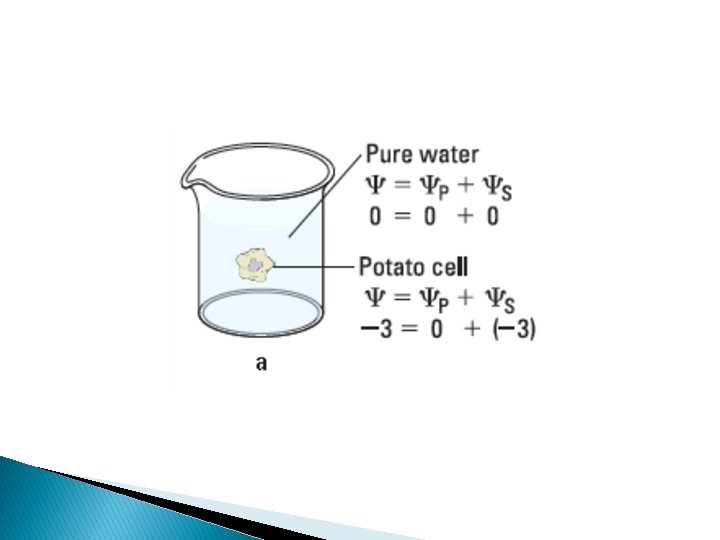
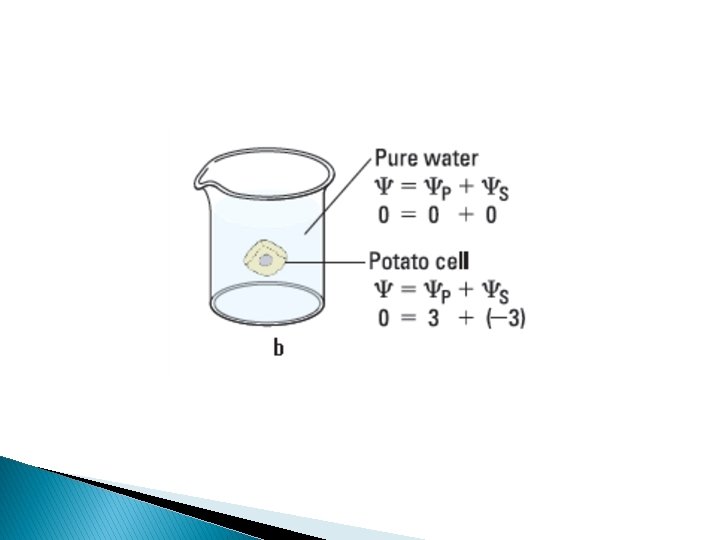
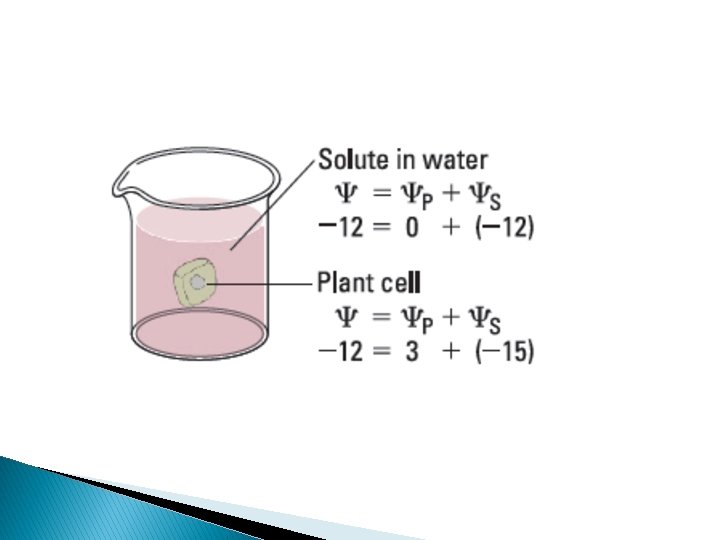
Water potential = Solute potential + Pressure potential = s + p Water potential • Tendency of water to leave the system • Higher water potential, greater tendency to leave • Water passes from one system to the other, through membrane by osmosis Osmotic potential • Solute potential • Pressure that a solution have to build to increase its chemical potential to that of pure water. • Solute potential is always negative. Pressure potential • Equivalent to pumpimg water from one place to another. • If pressure greater than atmospheric pressure is applied to pure water or a solution, its water potential increases.

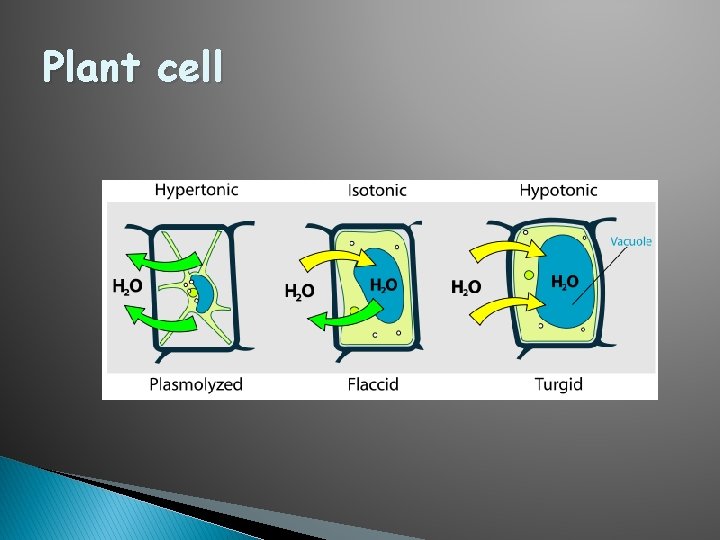
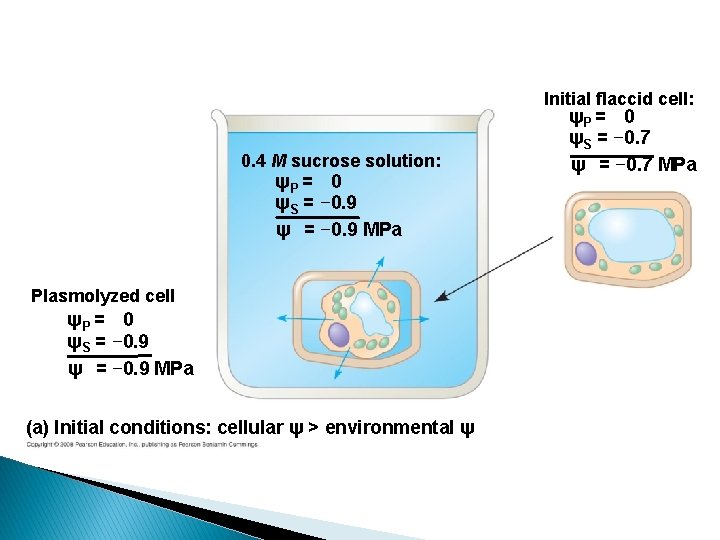
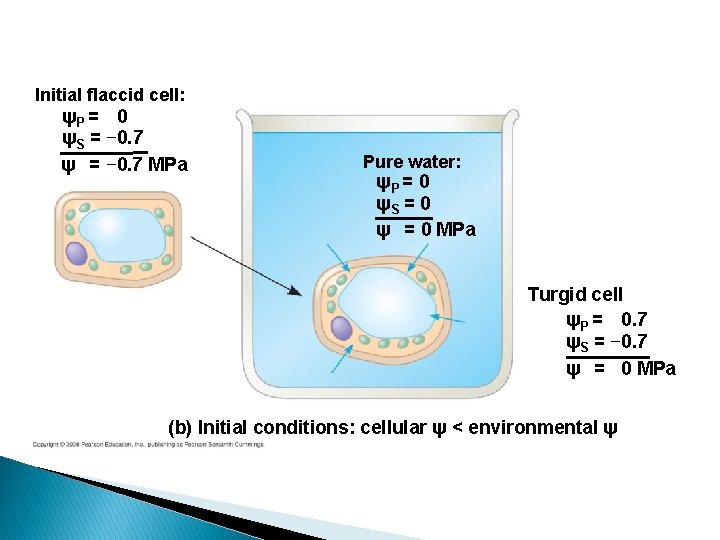
Plant cells � Flaccid: Limp-lost water � Turgid: Firm-gained water � Plasmolysis: Plant cell shrinks from cell wall Lost water � Deplasmolysis: Plant cell resumes turgidity Gained water

Plant cell

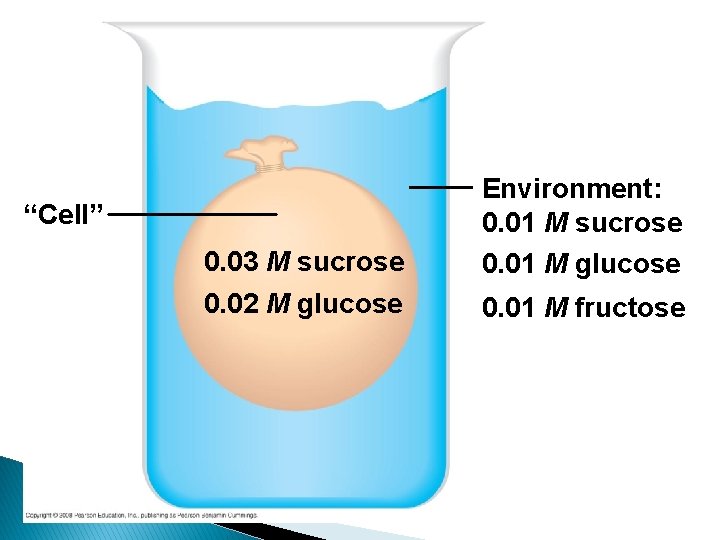
“Cell” 0. 03 M sucrose 0. 02 M glucose Environment: 0. 01 M sucrose 0. 01 M glucose 0. 01 M fructose

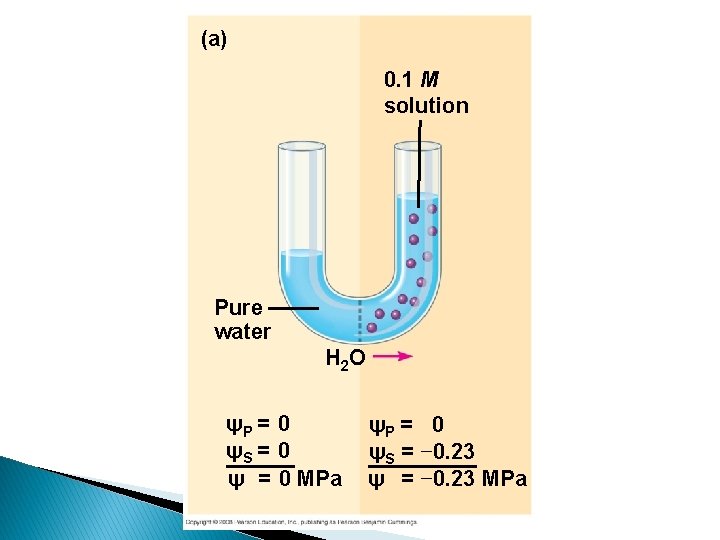
(a) 0. 1 M solution Pure water H 2 O ψP = 0 ψS = 0 ψ = 0 MPa ψP = 0 ψS = − 0. 23 ψ = − 0. 23 MPa

(b) Positive pressure H 2 O ψP = 0 ψS = 0 ψ = 0 MPa ψP = 0. 23 ψS = − 0. 23 ψ = 0 MPa

(c) Increased positive pressure H 2 O ψP = 0. 30 ψP = 0 ψS = − 0. 23 ψS = 0 ψ = 0 MPa ψ = 0. 07 MPa

Initial flaccid cell: ψP = 0 ψS = − 0. 7 ψ = − 0. 7 MPa 0. 4 M sucrose solution: ψP = 0 ψS = − 0. 9 ψ = − 0. 9 MPa Plasmolyzed cell ψP = 0 ψS = − 0. 9 ψ = − 0. 9 MPa (a) Initial conditions: cellular ψ > environmental ψ

Initial flaccid cell: ψP = 0 ψS = − 0. 7 ψ = − 0. 7 MPa Pure water: ψP = 0 ψS = 0 ψ = 0 MPa Turgid cell ψP = 0. 7 ψS = − 0. 7 ψ = 0 MPa (b) Initial conditions: cellular ψ < environmental ψ

� Ψs = -i. CRT � i = ionization constant � Sucrose=1. 0 (sucrose does not ionize water) � C = Molar concentration (from experiment) � R = Pressure constant (R=0. 0831 liter bars/mole K) � T = temperature in K (273 + C)


