Water Purification Required Practical Compare potable and pure

Water Purification (Required Practical) Compare potable and pure water. LAST LESSON Suggest how we could separate a mixture of salt and water. LAST YEAR January 2022 Define particulate and suggest their effect on our health. LAST WEEK Explain why carbon monoxide is so dangerous. LAST TOPIC Challenge: Create 2 questions where the answer requires the use of at least two key words in an explanation Carry out and complete the RP into water purification.

Water Purification (Required Practical) January 2022 Define the word pure. A substance not mixed or contaminated with any other substance or material. Stretch: Describe how we could investigate the pureness of water. Carry out and complete the RP into water purification.

Water Purification (Required Practical) January 2022

Carry out and complete the RP into water purification. Pure Distillation p. H Water Solids Seperation Identify methods to test p. H and dissolved substances in water. Carry out an investigation to test p. H and dissolved substances in water.
Water Purification (Required Practical) January 2022 Suggest how we could test the p. H of a substance. Suggest how we could test the presence of dissolved solids in a substance. You must write in full sentences and use a minimum of 23 words. Carry out and complete the RP into water purification.

Water Purification (Required Practical) Required practical activity January 2022 Apparatus and techniques Analysis and purification of water samples AT 2, AT 3, AT 4 from different sources, including p. H, dissolved solids and distillation. In this investigation you will test 3 water samples from different sources of p. H and the presence of dissolved solids. After distillation of the sea water, you will test the water again to check that dissolved solids have been removed, making the water fit to drink. Carry out and complete the RP into water purification.
Water Purification (Required Practical) *Take notes* Carry out and complete the RP into water purification. January 2022
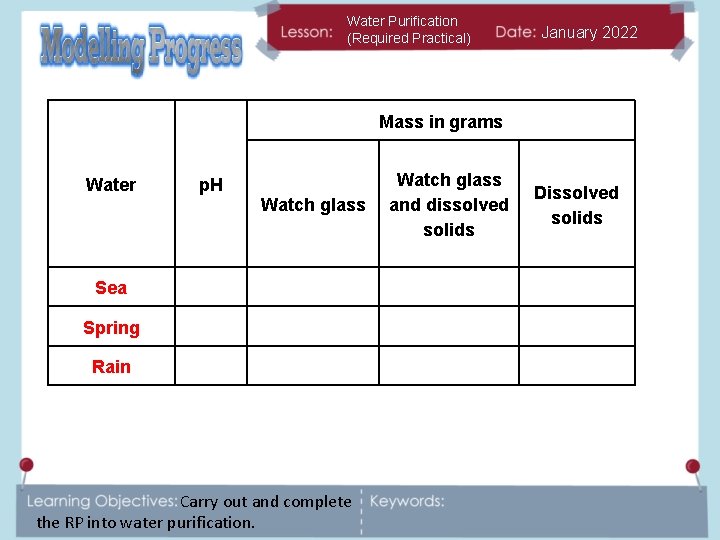
Water Purification (Required Practical) January 2022 Mass in grams Water p. H Watch glass Sea Spring Rain Carry out and complete the RP into water purification. Watch glass and dissolved solids Dissolved solids
Water Purification (Required Practical) January 2022 Required practical - Water - AQA - GCSE Chemistry (Single Science) Revision - AQA - BBC Bitesize Hypothesis: I predict that… Equipment: List the equipment you will be using… Variables: Control variables: (what you keep the same) Independent variable: (what you change) Dependent variable: (what you measure) Carry out and complete the RP into water purification. Method: Detail the steps you will take during your investigation… Results table: Construct a suitable for your results. You will be doing the test 3 times and then calculating an average. The IV (independent variable) goes in the left column.

Watch Glass Evaporation – Sea Water 1)Weigh a dry watch glass. Record its mass in the table. Pour 4 cm 3 sea water (less if your watch glass is small) into it and place it above a beaker acting as a water bath as shown in the diagram. 2) Allow all the water to evaporate from the watch glass. Do not let the water bath boil dry. 3) You should see dissolved solids on the glass. Remove the watch glass with tongs and allow to cool. Dry the bottom of the watch glass with a cloth and reweigh it. Record the new mass in the table. Subtract the mass of the watch glass alone and record the mass of the dissolved solids. Wash the watch glass and dry it. BE CAREFUL AS THE EQUIPMENT IS HOT AND MAY NOT BE STABLE.
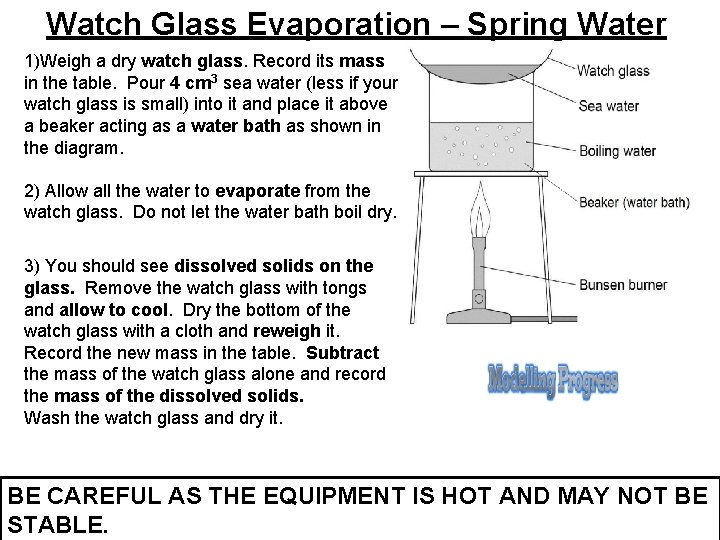
Watch Glass Evaporation – Spring Water 1)Weigh a dry watch glass. Record its mass in the table. Pour 4 cm 3 sea water (less if your watch glass is small) into it and place it above a beaker acting as a water bath as shown in the diagram. 2) Allow all the water to evaporate from the watch glass. Do not let the water bath boil dry. 3) You should see dissolved solids on the glass. Remove the watch glass with tongs and allow to cool. Dry the bottom of the watch glass with a cloth and reweigh it. Record the new mass in the table. Subtract the mass of the watch glass alone and record the mass of the dissolved solids. Wash the watch glass and dry it. BE CAREFUL AS THE EQUIPMENT IS HOT AND MAY NOT BE STABLE.

Watch Glass Evaporation – Rain Water 1)Weigh a dry watch glass. Record its mass in the table. Pour 4 cm 3 sea water (less if your watch glass is small) into it and place it above a beaker acting as a water bath as shown in the diagram. 2) Allow all the water to evaporate from the watch glass. Do not let the water bath boil dry. 3) You should see dissolved solids on the glass. Remove the watch glass with tongs and allow to cool. Dry the bottom of the watch glass with a cloth and reweigh it. Record the new mass in the table. Subtract the mass of the watch glass alone and record the mass of the dissolved solids. Wash the watch glass and dry it. BE CAREFUL AS THE EQUIPMENT IS HOT AND MAY NOT BE STABLE.
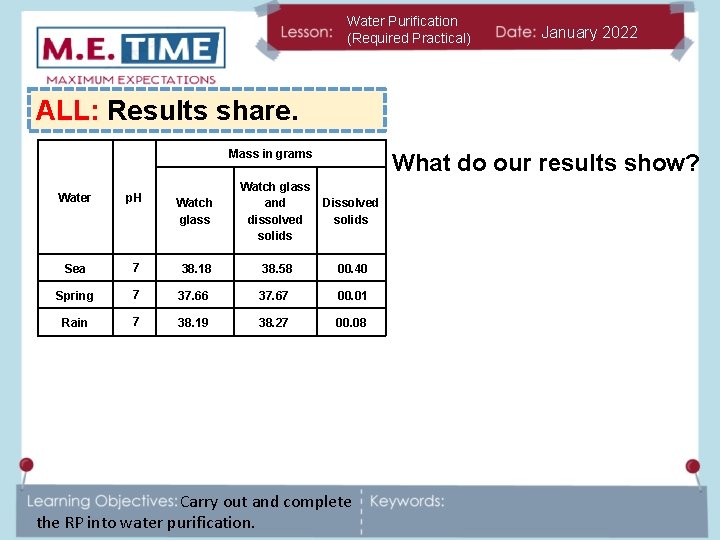
Water Purification (Required Practical) January 2022 ALL: Results share. Mass in grams What do our results show? Watch glass and dissolved solids Dissolved solids 7 38. 18 38. 58 00. 40 Spring 7 37. 66 37. 67 00. 01 Rain 7 38. 19 38. 27 00. 08 Water p. H Sea Carry out and complete the RP into water purification.

Water Purification (Required Practical) Conclusion The results show… The investigation has shown… Evaluation Over all the experiment well because… The experiment could have been improved by… If I was to carry out this experiment again I would… One error present was… I could reduce this by… January 2022 P- point? E – evidence from your results E – explain A – anything else? PEE again? What went well? What didn’t go so well? How could you improve? YOU MUST INCLUDE THE WORDS: accuracy, repeatability, ‘X’ error and reliability. STRETCH: Complete the exam style questions. Carry out and complete the RP into water purification.

Water Purification (Required Practical) January 2022 Todays Lesson Compare pure and potable. Last Lesson Explain why we make water potable. Last Lesson Suggest 3 resources we get from Earth. Carry out and complete the RP into water purification.

Water Purification (Required Practical) January 2022 1. Which water sample is most acidic? Rainwater • Why might this be? Acid rain – due to sulphur dioxide / nitrogen oxides dissolving in rainwater 2. Which water sample contains the most dissolved solids? Sea water • Why might this be? Contains all the salt added to the sea from rivers 3. During the distillation step, why is the test tube placed in a beaker of ice water? To cool and condense the gas produced by boiling 4. Name the piece of equipment that could be used instead of the beaker of ice water. Condenser 5. How did the sea water sample change after being distilled? Dissolved solids removed 6. In the real world, when is desalination of water necessary? outsupplies and complete When there. Carry are no of fresh water / ground the RP water into water purification.
- Slides: 16