Water Properties Science Guy In ATie com Biomedical

Water Properties Science. Guy. In. ATie. com Biomedical Sciences Program 1
2

Importance of water • Overview: The Molecule That Supports All of Life • Water is the biological medium here on Earth • All living organisms require water more than any other substance 3

Water Facts • Three-quarters of the Earth’s • surface is submerged in water The abundance of water is the main reason the Earth is habitable Figure 3. 1 4

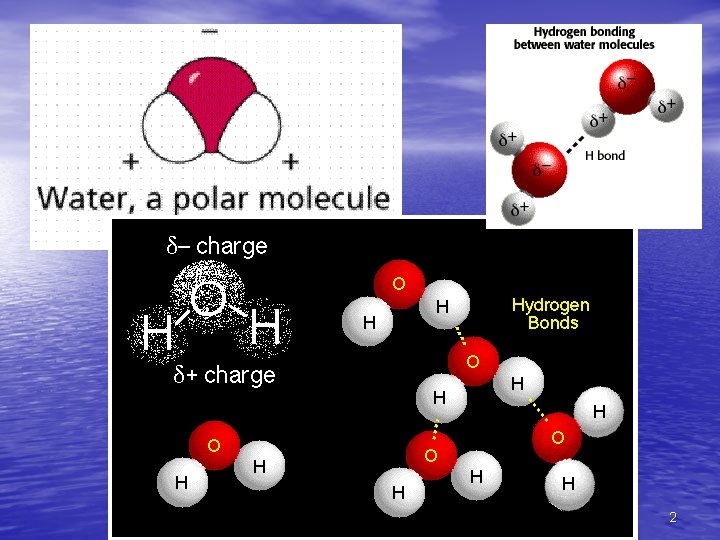
Water Facts • The polarity of water molecules • results in hydrogen bonding The water molecule is a polar molecule 5
6

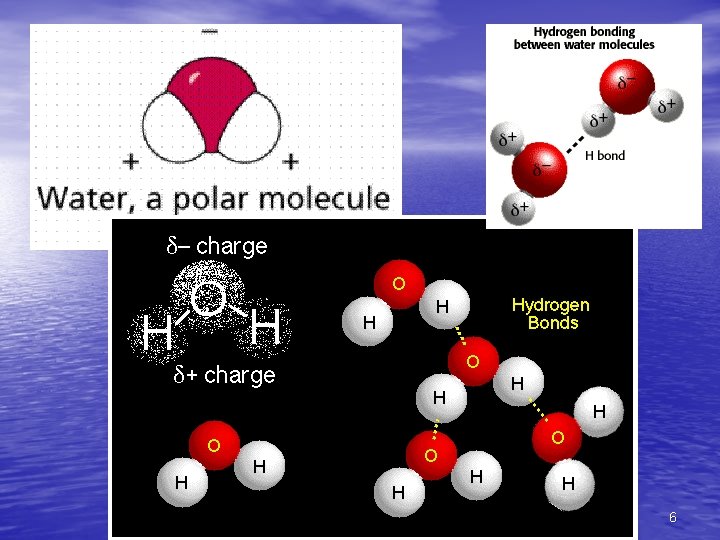
Water’s Polarity • The polarity of water molecules – Allows them to form hydrogen bonds with each other – Contributes to the various properties water exhibits – Hydrogen bonds + H + Figure 3. 2 – – + H + – 7
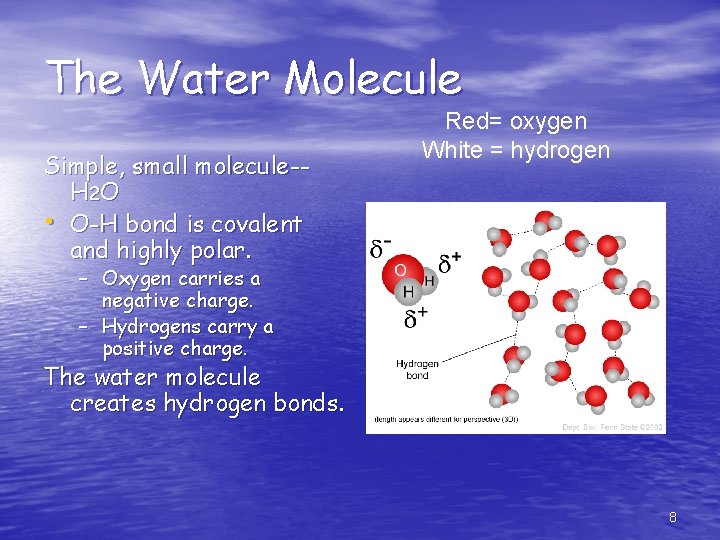
The Water Molecule Simple, small molecule-H 2 O • O-H bond is covalent and highly polar. Red= oxygen White = hydrogen – Oxygen carries a negative charge. – Hydrogens carry a positive charge. The water molecule creates hydrogen bonds. 8

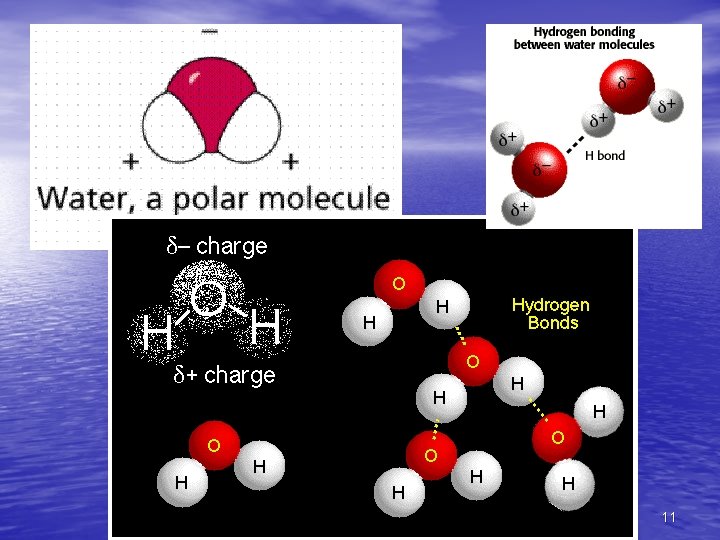
Cohesion • Water molecules exhibit cohesion • Cohesion – Is the bonding of a high percentage of the molecules to neighboring water molecules – Is due to hydrogen bonding – Like molecules bonding to each other (water attracting other water molecules) 9

Surface Tension • Surface tension – Is a measure of how hard it is to break the surface of a liquid – Is related to cohesion Figure 3. 4 10
11

The Solvent of Life • Water is a versatile solvent due to its polarity • It can form aqueous solutions • Called the universal solvent because so many substances dissolve in water 12
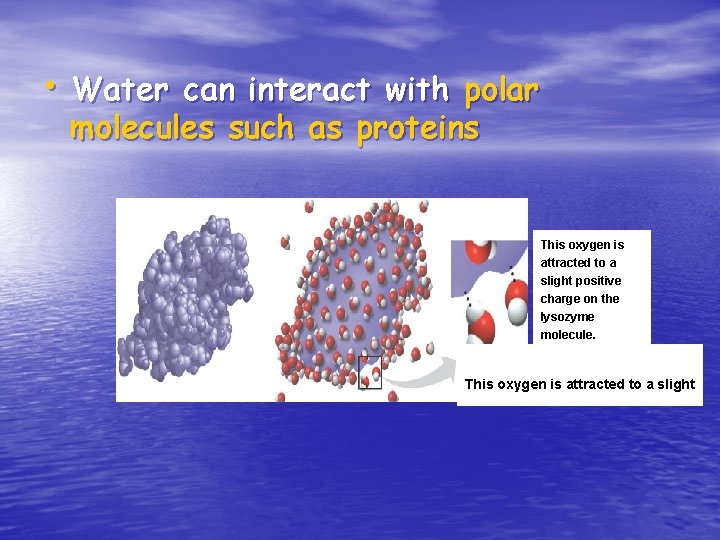
• Water can interact with polar molecules such as proteins This oxygen is attracted to a slight positive charge on the lysozyme molecule. This oxygen is attracted to a slight

Hydrophilic and Hydrophobic Substances • A hydrophobic substance – Does not have an affinity for water – Nonpolar – lipids • A hydrophilic substance – Has an affinity for water – Polar or ionic – Carbohydrates, salts 14
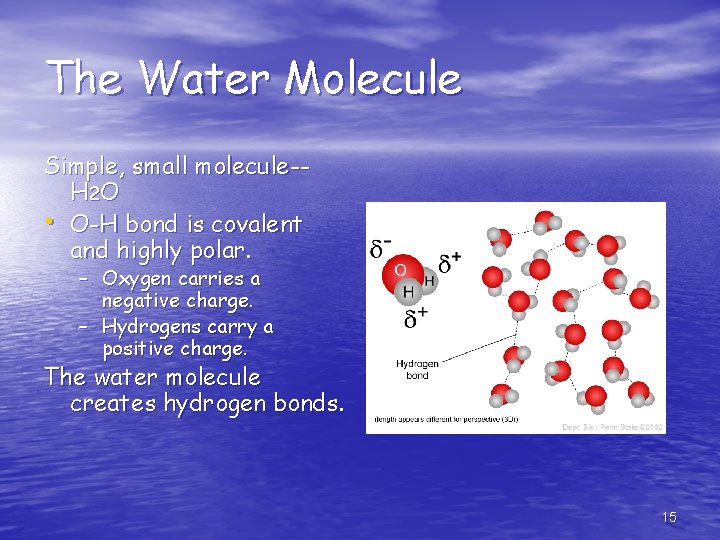
The Water Molecule Simple, small molecule-H 2 O • O-H bond is covalent and highly polar. – Oxygen carries a negative charge. – Hydrogens carry a positive charge. The water molecule creates hydrogen bonds. 15

Hydrogen Bonding • These are bonds that exist between molecules. – More specifically, hydrogen bonds exist between polar molecules. – The positive end of one polar molecule lines up with the negative end of another molecule. 16

Good luck with the lab! 17
- Slides: 17