Water Properties Lab Water is Polar Covalent Nonpolar

Water Properties Lab

Water is Polar Covalent Nonpolar Oxygen (even sharing of e-) Uneven sharing of e-
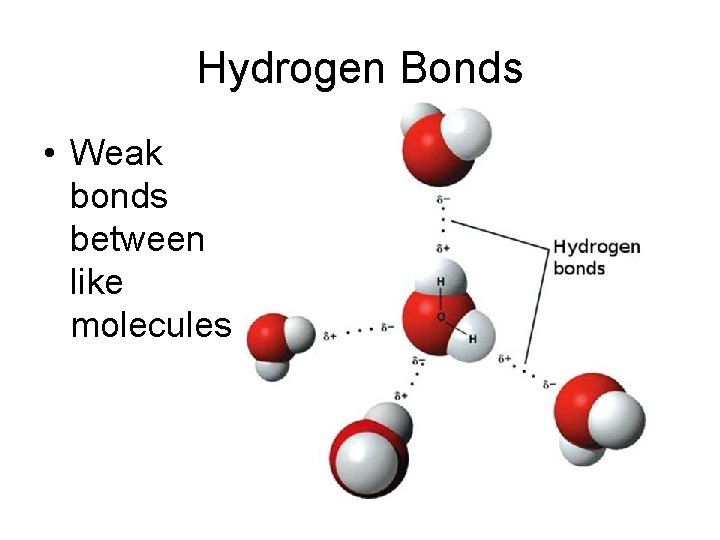
Hydrogen Bonds • Weak bonds between like molecules
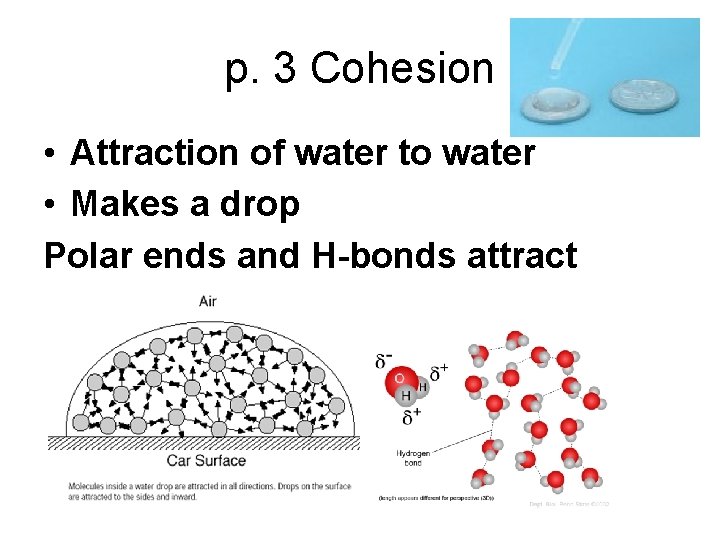
p. 3 Cohesion • Attraction of water to water • Makes a drop Polar ends and H-bonds attract

What forms a drop? • Cohesion causes water to form drops • surface tension causes them to be nearly spherical • adhesion keeps the drops in place.

Surface Tension • Water attracts itself and pulls in to form a “film” on its surface to form a drop.

Adhesion • Attraction of water to an unlike substance • (like glass) • Note the drop is flatter than on wax paper

p. 4 Water Drop Shapes • Rounder the drop, the stronger the H-bonds in the drop of water. • Round drop on wax paper (not strong adhesion to water). • As adhesion to glass is stronger + pulls the water, it makes a flatter drop.
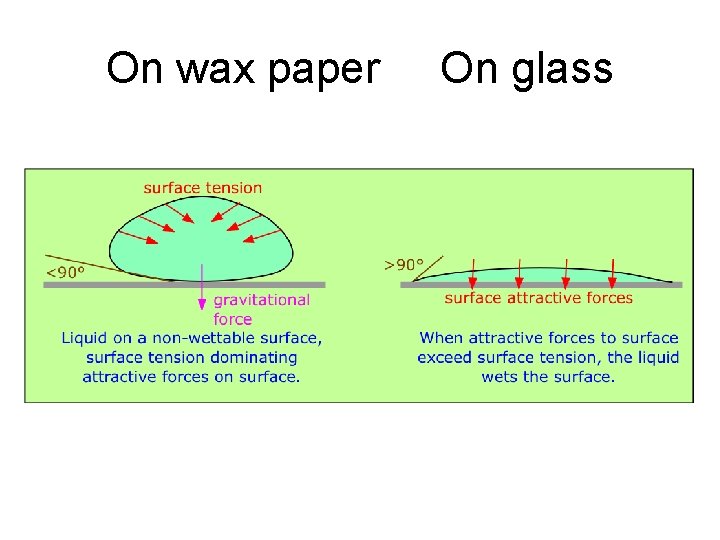
On wax paper On glass

p. 4 -#6 top Water on wax paper • Plain wax paper Soapy wax paper You can break surface tension with soap.

Polar coheres to Polar not Nonpolar • Water (Polar) has no adhesion to the wax (nonpolar) paper. It would roll right off. • Water has more adhesion to a glass plate.

p. 6 Climbing Property of Water • CHROMATOGRAPHY: • Black is Colors are separated composed of all by densities. Less the colors. dense colors go to Paper has the top. pores, open spaces • Capillary action cohesion (water to water) + adhesion (water to paper) • Polar water + polar ink (dissolves)

Capillarity – water climbs up small spaces • The small spaces (pores of the paper) provide adhesion to the water. • The water makes a column by cohesion to itself. (water to water because they are polar)
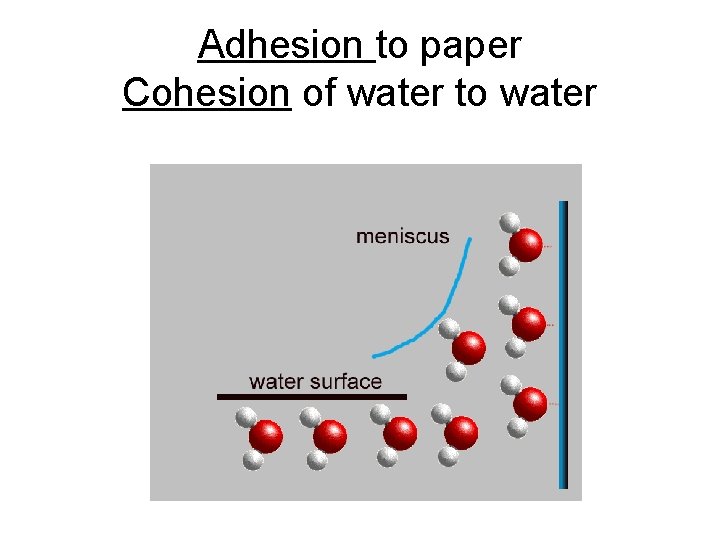
Adhesion to paper Cohesion of water to water

p. 8 #6 Oil is hydrophobic • Oil and water do not mix. • H-bonding is only in polar Oil is on top polar Non-polar nonpolar Water is on bottom Polar polar
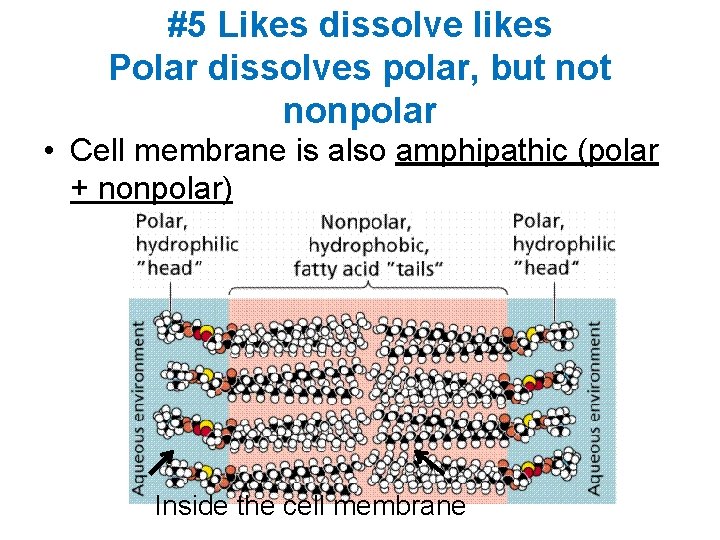
#5 Likes dissolve likes Polar dissolves polar, but nonpolar • Cell membrane is also amphipathic (polar + nonpolar) Inside the cell membrane

p. 9 # 6 Food coloring dissolves in water. • Water is polar +H-bonds • Food coloring is polar. • Makes a solution (water surrounds the dye) • Oil is nonpolar (no charge). • And no H- bonds. Polar molecules dissolve polar molecules.
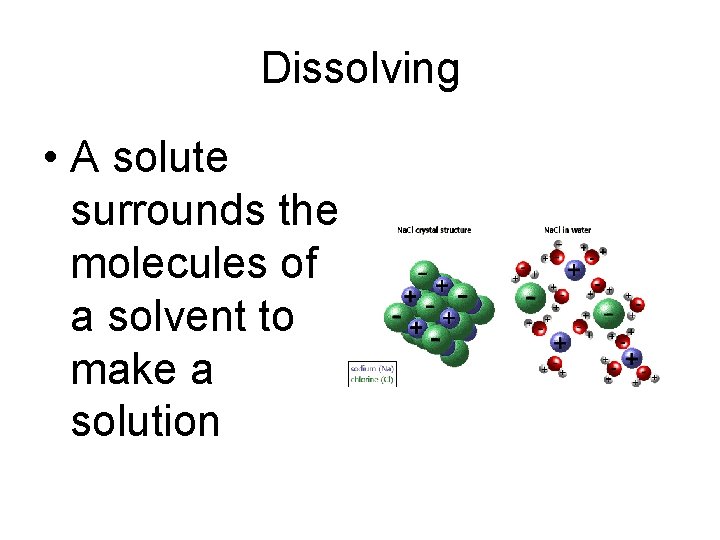
Dissolving • A solute surrounds the molecules of a solvent to make a solution

Stirring oil and water • The oil will go back to being separate from the water.

D. Oil sheen on water (oil on top of water in a thin layer)

Interpret #6 Getting rid of oil on water How. Stuff. Works "How do you clean • Detergent breaks up the oil into very small droplets. up an oil spill? “ (pick a video) Burn Off Booms Hi-pressure water

Slick Sack • Absorbent pad Hand washing

Oil Spills Problems • • • a. dead sea life b. human life ills c. cost of clean-up

p. 9 Interpret 5 Amphipathic (bipolar) • molecules have (hydrophobic) nonpolar end and to attract oil structure, but also have a region that is polar (hydrophilic) attracts water to wash it away. Like detergent.

Soap is a surfactant • That reduces the surface tension • Breaks the cohesion of water molecules
- Slides: 25