Water Potential Water potential Potential energy of water





- Slides: 11

Water Potential

Water potential? ? • Potential energy of water per unit area compared to pure water – Allows us to figure out where water will flow • Measured in Psi – Remember Posidon, Greek god of ocean had trident • Formula

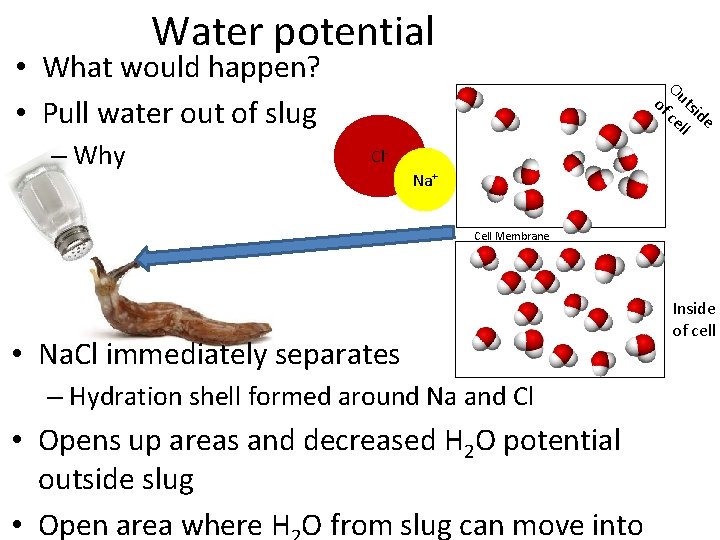
Water potential • What would happen? • Pull water out of slug – Why of Ou ts ce ide ll Cl. Na+ Cell Membrane • Na. Cl immediately separates – Hydration shell formed around Na and Cl • Opens up areas and decreased H 2 O potential outside slug • Open area where H O from slug can move into Inside of cell

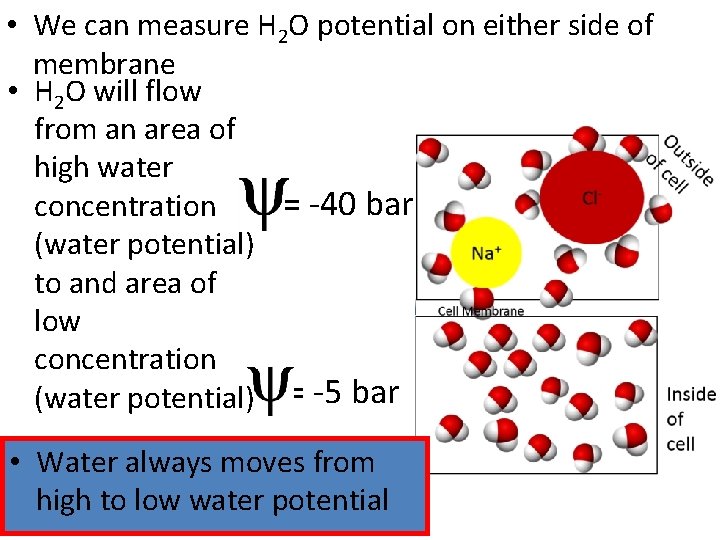
• We can measure H 2 O potential on either side of membrane • H 2 O will flow from an area of high water = -40 bar concentration (water potential) to and area of low concentration (water potential) = -5 bar • Water always moves from high to low water potential

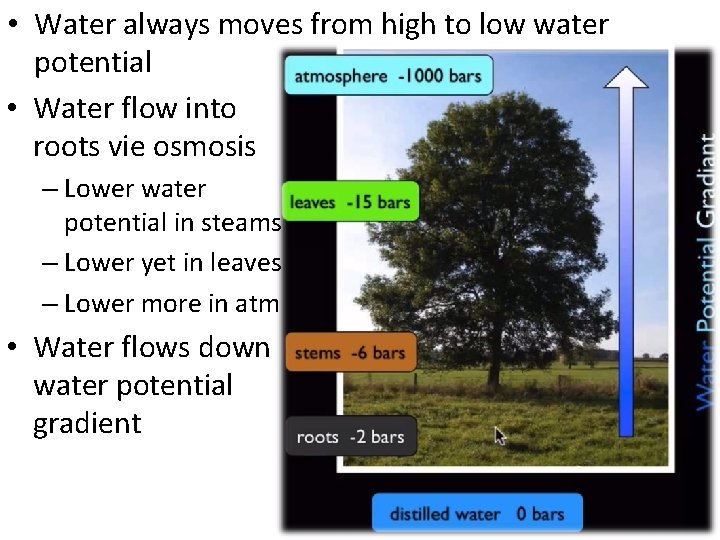
• Water always moves from high to low water potential • Water flow into roots vie osmosis – Lower water potential in steams – Lower yet in leaves – Lower more in atm • Water flows down water potential gradient

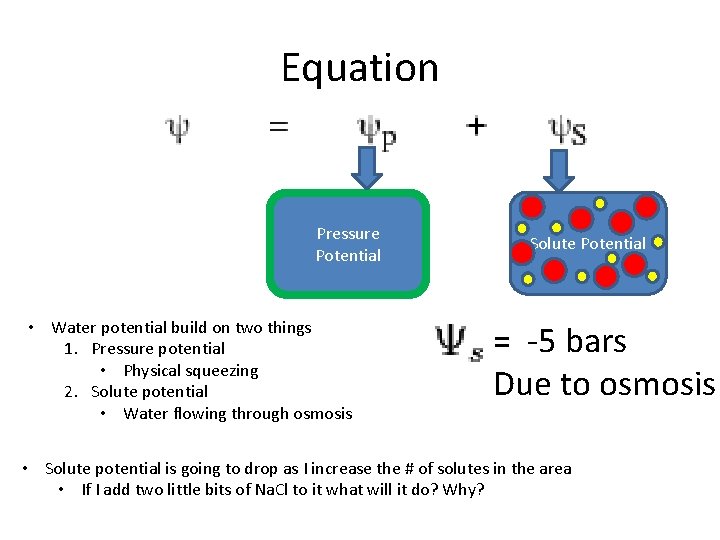
Equation Pressure Potential • Water potential build on two things 1. Pressure potential • Physical squeezing 2. Solute potential • Water flowing through osmosis Solute Potential = -5 bars Due to osmosis • Solute potential is going to drop as I increase the # of solutes in the area • If I add two little bits of Na. Cl to it what will it do? Why?

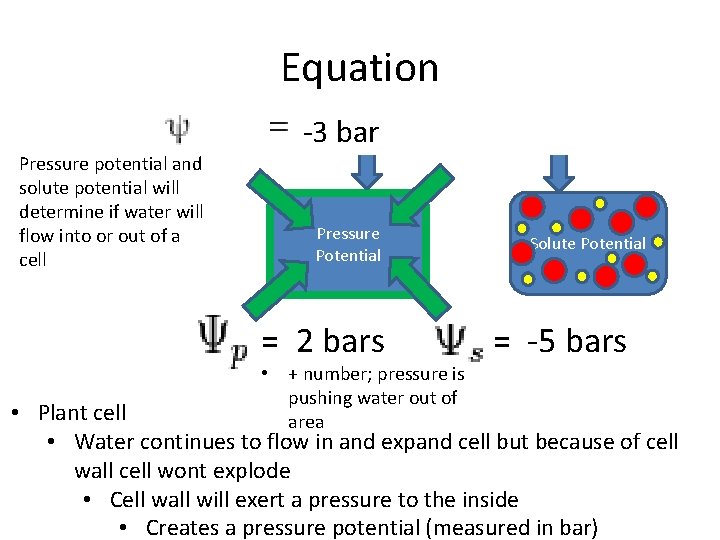
Equation -3 bar Pressure potential and solute potential will determine if water will flow into or out of a cell Pressure Potential = 2 bars • + number; pressure is pushing water out of area Solute Potential = -5 bars • Plant cell • Water continues to flow in and expand cell but because of cell wall cell wont explode • Cell wall will exert a pressure to the inside • Creates a pressure potential (measured in bar)

Solute potential equation = -i. CRT i= ionization constant (factor between 1 and 2) C= concentration (moles/ liter) R= pressure constant (0. 0831 liter/bar/mole K) T = temperature in Kalvin (273 + o. C)

CYU • The molar concentration of a sugar solution in an open beaker has been determined to be 0. 2 M. Calculate the solute potential at 22 degrees Celsius. = - (1)(. 2 moles/liter)(. 0831 liter/bars/mole K)(295 K) = - (1)(. 2)(. 0831 bars)(295) = - (1)(. 2)(. 0831)(295) bars = - 4. 9029 bars = - 5 bars

What is the overall water potential? • We know that = -5 bars • In the problem it is given that our solution is in an open container – So that makes our what? • Zero 0 + -5 bar

• AND DONE!