Water Potential Measures the tendency of water to











- Slides: 15

Water Potential Measures the tendency of water to leave one place for another Ψ “psi” Used by botanist to predict water movement into or out of a cell.

Question If we soak potato CELLS in sucrose which way will water move? Water always moves from a higher water potential to a lower water potential.

Look on the Formula Sheet Ψ = Ψp + Ψs Water Potential Pressure Potential Solute Potential Water always moves to a lower, more negative, water potential.

Ψs= Solute Potential Ψs Solutes are always negative More solutes = more negative Which way will water move? What is happening to the pressure potential in cell?

Ψp= Pressure Potential Usually Positive

Water Potential in the Potato Cells Ø Revise your definition: Osmosis is the movement of water molecules through a selectively permeable membrane from a region of higher water potential to an area of lower water potential Ø Water always moves to a more negative water potential.

Water Potential Can we figure out the water potential of a cell in a solution at equilibrium? Ø Ψ= Ψp + Ψs Ø Where there is no % change in mass, the solution in the beaker has the same water m ir u cells. potential as the potato li ib y D m a n ic u q E

Water Potential at Equilibrium Ψ of Solution in Beaker = (Ψ= Ψp + Ψs) = = Ψ of Potato cells (Ψ= Ψp + Ψs)

Water Potential Solution (Ψ= Ψp + Ψs) = Potato Cells (Ψ= Ψp + Ψs) Pure water at atmospheric pressure has a water potential of zero. (open beaker) Ψ=Ψs+(Ψp= 0) = Ψ= Ψs Ψ= Ψp + Ψs = Ψ= Ψp + Ψs

Solute potential of a solution at equilibrium equals water potential of potato cells. Ø Ψs = ( Ψ= Ψp + Ψs ) Ψ We can calculate Ψs and then we will know Ψ. We don’t know individual components of water potential for the potato cells.

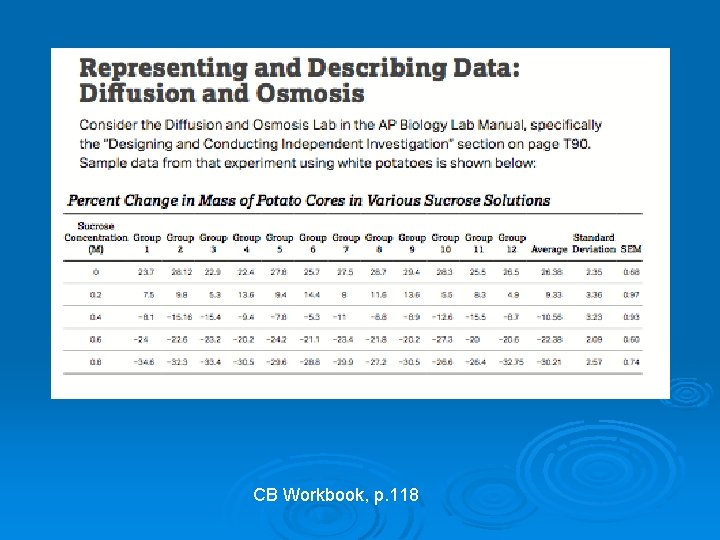
CB Workbook, p. 118

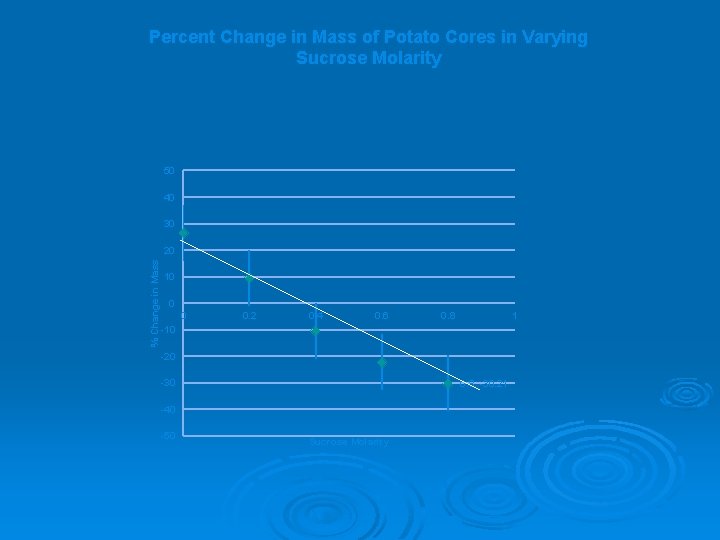
Percent Change in Mass of Potato Cores in Varying Sucrose Molarity 50 40 30 % Change in Mass 20 10 0 0 0. 2 0. 4 0. 6 0. 8 1 -10 -20 -30 0. 8, -30. 21 -40 -50 Sucrose Molarity


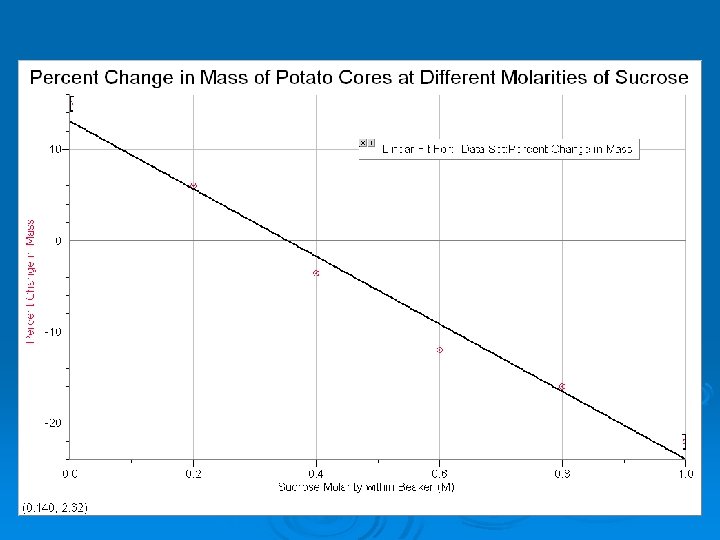
To Calculate Ψs Ψs = -i. CRT (Formula Chart) i= Ionization constant (sucrose is 1. 0 because it does not ionize). C= Molar Concentration (from line of best fit where the line crosses the x axis) R= Pressure Constant (0. 0831 liter bars/mole °K T= Temperature °K (273 + °C)

An example Ø Ψs = -i. CRT Ø Ψs= -(1. 0)(0. 36 mole/liter)(0. 0831 liter bar/mole °K)(295 °K) Ø -8. 83 bars Ø This equals the entire Ψ of the cell.