Water Pollution Water Pollution Overview Types Sources Effects




















































































- Slides: 100

Water Pollution


Water Pollution Overview Types, Sources, Effects, Management

The Chesapeake Bay

Chesapeake Bay • The largest Estuary in the US – Receives water from numerous rivers and streams that mixes with salt water of the ocean to produce an extremely productive estuary. – Drains water from a large watershed of urban, suburban and agricultural areas.

Bay contains an abundance of • nutrients, – Million of lbs of Nitrogen & Phosphorus→ algae blooms • sediments, – Current estimate = 8. 2 billion kg of sediment each year. • chemicals. – Damage to fish endocrine systems → hermaphrodites • All causing poor water quality, decreased biodiversity

Chesapeake Bay • 2000 Chesapeake Bay Action Plan • 2010 many goals were being met – ↑water clarity – ↓nitrogen – ↑crab pop – ↑water quality

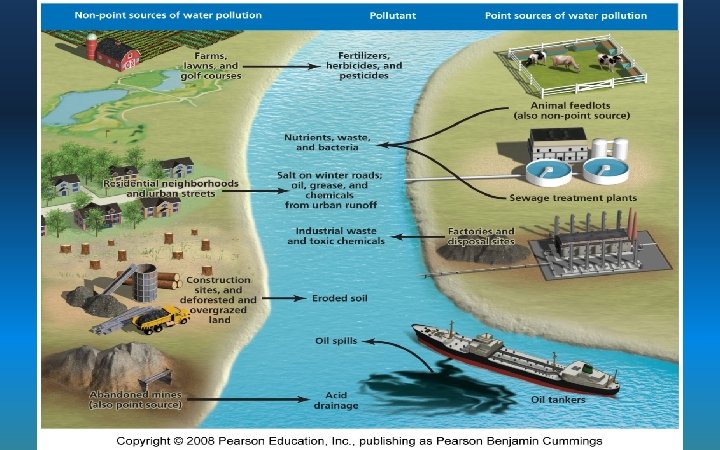
The Basics • Water Pollution: Any chemical, biological or physical change in water quality that has a harmful effect on living organisms (including humans!) or makes it unusable for agriculture – Point Sources - Discharge pollution from specific locations (single point). EASY TO MONITOR/REGULATE • Factories, power plants, oil wells – Non-Point Sources - Scattered or diffuse, having no specific location of discharge. HARDER TO CONTROL!! • Agricultural fields, feedlots, golf courses

Point source Non Point

15_21. JPG

Major Water Pollutants 1. Human wastewater: sewage & gray water -- Decomposition of organic waste → Huge O 2 demand – Excess nutrients (N & P) eutrophication – Disease-causing organisms (esp. E. Coli & Cholera) 2. Metals (Pb, Hg, As, Cd) 3. Acid 4. Synthetic Organics (pharma; pesticides; Military & Industrial compounds) 5. Oil 6. Solid Waste (especially plastic!) 7. Sediment 8. Thermal & Noise pollution

Water Quality Indicators • The measurements that are used to decide if the water quality is in good condition or not. • Water must meet different requirements for different uses (fishing versus drinking) – Drinking water must have 0 coliform bacteria colonies, very low turbidity and low nutrient levels – Water for fishing can have thousands of coliform bacteria colonies and high turbidity, but must have at dissolved oxygen levels of at least 6 ppm to support life.

Temperature • What is measured – the temperature (o. C or o. F) of the water body. Should change according to hydrology and season. • How to measure – use a thermometer • Desired range – varies depending on location, but should fit the range of tolerance of native species and change seasonally as appropriate to the region • Impacted by – stream flow and lake depth, thermal pollution from industry or power plants, turbidity level

p. H • What is measured – the concentration of H+ ions. Indicates how acidic or basic the water is. • How to measure – use a p. H probe or litmus paper • Desired range – most species can survive between 5 and 8 • Impacted by – acid rain, acid mine drainage, buffering capacity, geology of the watershed • p. H = -log[H+]

Nitrate (NO 3 -) • What is measured – the concentration (ppm) of nitrate ions. Indicates how nutrient rich the water is. • How to measure – use a digital probe or a nitrate titration kit • Desired range – less than 1 ppm • Impacted by – agricultural runoff, fertilizers, feedlots, sewage treatment plants

Phosphate (PO 43 -) • What is measured – the concentration (ppm) of phosphate ions. Indicates how nutrient rich the water is. • How to measure – use a digital probe or a phosphate test kit • Desired range – less than 0. 1 ppm • Impacted by – agricultural runoff, fertilizers, detergents

Dissolved Oxygen (DO) • What is measured – the concentration (ppm or mg/L) of oxygen dissolved in the water. Indicative of the amount of life the water can support. • How to measure – use a digital probe or a DO test kit • Desired range – above 6 ppm • Impacted by – temperature, photosynthesis, nutrient levels, turbidity, organic wastes

Biochemical Oxygen Demand (BOD 5) • What is measured – the rate of oxygen use. Specifically the amount of oxygen consumed over 5 days. Indicative of the amount of organic matter in the water. • How to measure – Measure the DO, cover water sample and allow to sit for 5 days. Measure DO again. BOD = DOi. DOf • Desired range – • pristine rivers <1 mg/L, • polluted river 2 mg/L-8 mg/L • sewage effluent <20 mg/L • Impacted by – respiration, nutrient levels, organic wastes

Fecal Coliform • What is measured – the number of bacteria colonies present in the sample. • Indicator species - Indicates the presence of bacterial pathogens in the water. • How to measure – place a 1 m. L of of water on a agar petri dish. Incubate for 24 -48 hours. Count bacteria colonies. • Desired range – – Drinking water 0/100 m. L – swimming <200/100 m. L • naturally present in ecosystems • Impacted by – animal waste, sewage

Turbidity • What is measured – the amount of light that can pass through water. Indicates the amount of suspended/dissovled solids in the water. • How to measure – Collect a sample and use a turbidity meter, compare to known samples, lower a Secchi disk and record depth at which it can be seen. • Desired range – Drinking water <1 NTU – Ecosystem < 5 NTU over background • Impacted by – sediment, flooding, animal waste, sewage, riparian habitat, land disturbances, nutrients

Macroinvertebrates / Species Diversity Good • What is measured – the size, diversity and distribution of Stoneflies organisms in the aquatic habitat • How to measure – – Macroinvertebrates – scoop and screen sediment samples to collect organisms. Use keys to identify and count species. Pond Mussels – Species diversity – use sampling techniques to estimate diversity and size of populations Bad Mayflies Snipefly larvae • Desired range – – Want a wide variety of native species Leeches – Healthy populations of pollution sensitive species – Low populations of pollutant tolerant species • Impacted by – all of the previously discussed indicators, but mostly DO Pouch Snails Tubifex worms Midgeflies larvae

http: //www. epa. gov/bioiweb 1/pdf/EPA-260 -R-08 -016 An. Introductionto. Freshwater. Fishesas. Biological. Indicators. pdf Fish Species Intolerant of Pollution Tolerant of Pollution Salmon and Trout Carp Streamline chub Creek Chub Lamprey Blackstripe Minnow

Emerging Contaminants • http: //www. teachersdomain. org/asset/frntc 10_vid_water/ • Sources of emerging contaminates – Excess drug concentrations are urinated out and end up in waste water. – Improper disposal of medication (i. e. flushing medication down the toilet) – Excess antibiotics and hormones fed to cattle enter the water cycle via waste lagoons – Chemicals in everyday products like cleaners, plastics and fragrances

Types of Water Pollution

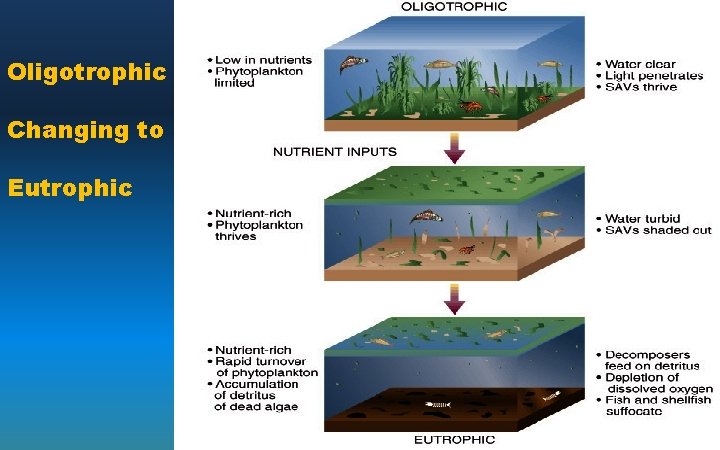
Eutrophication • Oligotrophic: nutrientpoor water • Eutrophic: nutrient-rich water Oligotrophic Eutrophic • As nutrients are added from pollution: – an oligotrophic condition rapidly becomes eutrophic.

Nutrients • Sources - agricultural runoff (fertilizer, feedlots), detergents (P only), disturbed soil, products of decomposition • Effects – Eutrophication nutrients ↑, algae grows, blocks sunlight, dies and decomposes, O 2 ↓ – Dead zones • Indicators – Elevated N and P levels – Decreased DO levels, increased BOD levels – Cloudy water: resembles pea soup from algae and cyanobacteria • Reduction - reduce runoff, treat stormwater, improve farming techniques, use organic fertilizer, avoid detergents with phosphates


Oligotrophic Changing to Eutrophic

Natural Vs. Cultural Eutrophication • Natural eutrophication – aquatic succession – occurs over several hundreds of years – mostly from runoff of plant nutrients from the surrounding land • Cultural eutrophication – driven by human activities – occurs rapidly – mostly nitrate- and phosphate-containing effluents – 85% of large lakes near major population centers in the U. S. have some degree of cultural eutrophication.

Combating Eutrophication Getting at root cause • Controlling point sources • Ban phosphate detergents • Sewage-treatment improvements • Reduce /control runoff from – Controlling nonpoint sources: Difficult to address runoff • Urban • Agricultural fields • Deforested woodlands • Overgrazed pastures

Combating Eutrophication • Attack the symptoms – Chemical treatment – Aeration – Harvesting aquatic weeds – Drawing water down

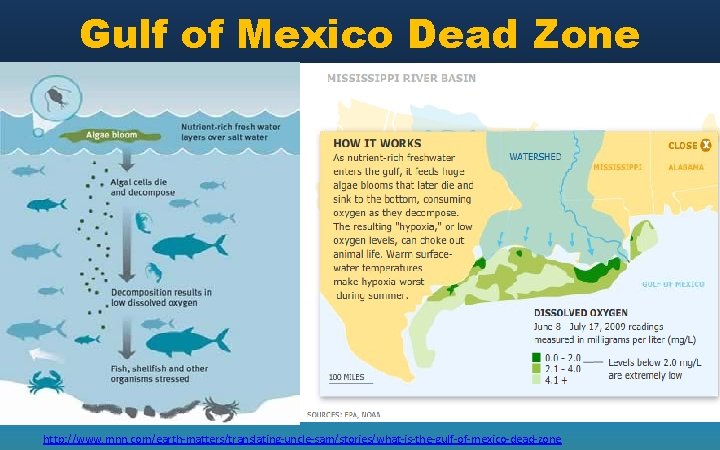
Excess nutrients Lakes – Eutrophication Put in order: a. b. c. d. e. f. Decomposers consume lots of oxygen, reducing DO levels in the lake Fish kills occur Algae grows, blocking sunlight Plants and other aquatic producers die due to lack of sunlight and space Algae and other plants die and sink to the bottom of the lake Fertilizer or animal waste runs off into the lake Ocean – dead zone • http: //www. pbslearningmedia. or g/asset/envh 10_vid_deadzone/

Gulf of Mexico Dead Zone http: //www. mnn. com/earth-matters/translating-uncle-sam/stories/what-is-the-gulf-of-mexico-dead-zone

Biochemical Oxygen Demand (BOD) o BOD- the amount of oxygen a quantity of water uses over a period of time at a specific temperature. Lower BOD - water is less polluted and o higher BOD -more polluted by wastewater. o o Example: decomposition of leaves = BOD of 5 to 20 mg of oxygen o Decomp of human waste = BOD of 200 mg. o

Organic Matter (also called oxygen demanding waste) • Sources - agricultural runoff, sewage, paper mills, food processing – feeds microbes • Effects - Organic material must be broken down leading to an increase in biological activity and oxygen demand until waste is broken down. • Indicators – Slightly elevated N and P levels – Decreased DO levels, increased BOD levels – Possible increase in fecal coliform depending on source – Increase in turbidity – Loss of sensitive species • Reduction - reduce runoff, increase riparian habitat, treat sewage, improve farming techniques

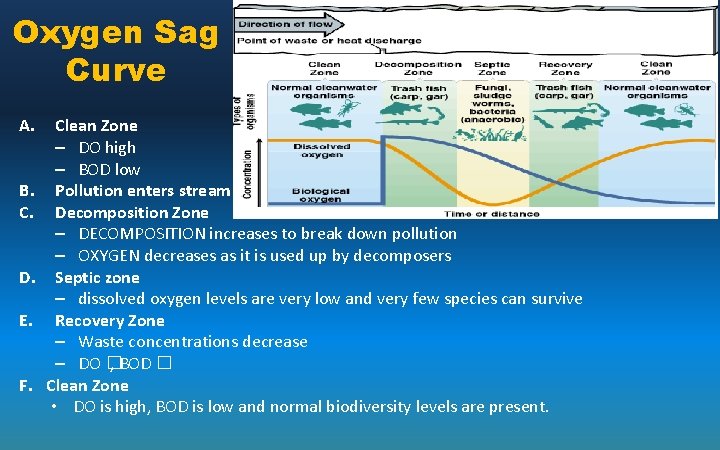
Oxygen Sag Curve A. B. C. D. E. F. Clean Zone – DO high – BOD low Pollution enters stream Decomposition Zone – DECOMPOSITION increases to break down pollution – OXYGEN decreases as it is used up by decomposers Septic zone – dissolved oxygen levels are very low and very few species can survive Recovery Zone – Waste concentrations decrease – DO � , BOD � Clean Zone • DO is high, BOD is low and normal biodiversity levels are present.

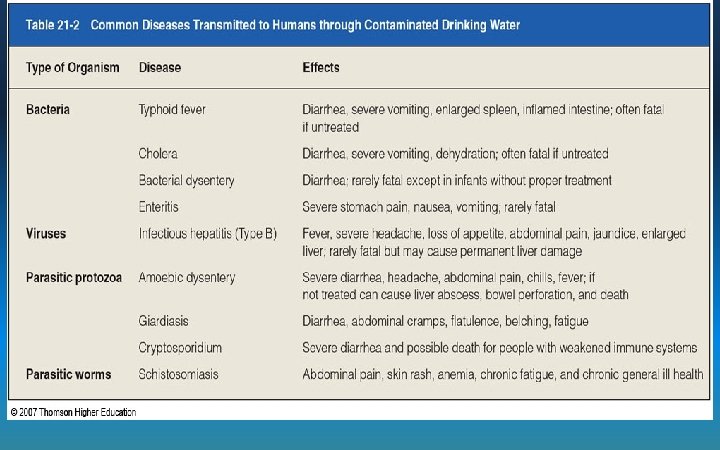
Pathogens • Sources - human and animal waste (fecal matter) • Effects – diseases in humans and animals: cholera, typhoid fever, hepatitis, Cryptosporidium, Giardia, E. coli, Guinea worm Cholera – Symptoms: nausea, vomiting, diarrhea, dehydration • Indicators – Presence of fecal coliform bacteria (these can be Rota Virus harmless themselves, but indicate the presence of other pathogens) • Reduction – treat sewage using disinfection, separate drinking and waste water, boil water before drinking, use filters for parasites Cryptosporidium E. Coli Giardia

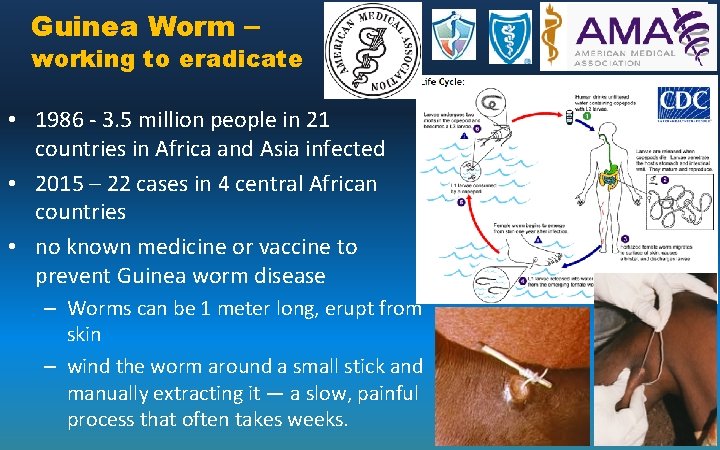
Guinea Worm – working to eradicate • 1986 - 3. 5 million people in 21 countries in Africa and Asia infected • 2015 – 22 cases in 4 central African countries • no known medicine or vaccine to prevent Guinea worm disease – Worms can be 1 meter long, erupt from skin – wind the worm around a small stick and manually extracting it — a slow, painful process that often takes weeks.


Global Problems • According to the World Health Organization, an estimated 1. 1 billion people (1/6 of pop) do not have access to safe drinking water • Diarrheal diseases kill around 3. 1 million people globally each year, – mostly children in developing countries. •

Metals • • Lead (pipes) Mercury (coal & garbage burning) Arsenic (rock breakup – natural & mining) Cadmium (electronics in landfills) • Tend to bioaccumulate up the food chain and cause damage to nervous system or cancers

Heavy Metals • Sources - natural deposits (arsenic, mercury, etc) – – mining (all) coal burning (mercury) e-waste (cadmium, mercury, lead) industrial processes like smelting (lead, mercury, arsenic, etc) • Low p. H, high salinity, and O 2 deficiencies increase metal concentrations in water • Effects – heavy metals bioaccumulate and biomagnify – Often have the largest impacts at the top of the food chain – Most are toxic at low doses and nuerotoxins • Arsenic (natural) • Indicators - Positive tests for metals • Reduction – – http: //ca. pbslearningmedia. org/res ource/envh 10. sci. life. nathis. arseni c/in-small-doses-arsenic/ minimize mining and perform effective remediation burn coal in larger coal plants with advanced air pollution control devices recycle e-waste regulate industry and require the use of advanced air pollution control devices

Health Effects • Lead and mercury are poisonous. • linked to a variety of conditions such as – – – mental retardations cerebral palsy developing delays kidney disorders several damage to nervous and cardiovascular systems. n. Low levels of in the brain cause neurological problems such as headache, depression, and quarrelsome behavior.

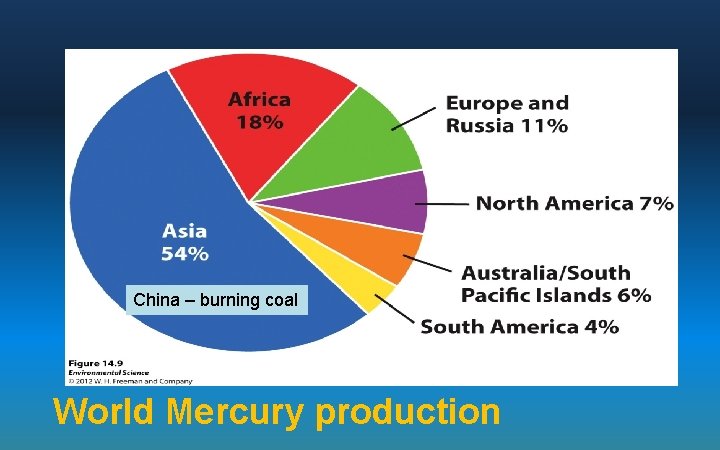
Heavy Metals: Mercury & Lead • Mercury - burning coal and mining gold • The mercury dumped in Minamata Bay by a factory entered humans through their diet of fish by bioaccumulation caused Minamata disease. – People showed mental impairments, birth defects, and death caused by the chronic effects of exposure to mercury. • Lead - mining and refining processes but is also in paint, water pipes, lead glazes or ceramics and burning fossil Mercury and lead are toxic heavy metals

China – burning coal World Mercury production

Arsenic - a Natural Threat • Toxic Arsenic (As) can naturally occur at high levels in soil & rocks. • Drilling into aquifers can release As into drinking water supplies. • According to WHO, more than 112 million people are drinking water with As levels 5 -100 times the 10 ppb standard. – Mostly in Bangladesh, China, and West Bengal, India.

Synthetic Organic Compounds • Chemicals that contain carbon atoms. • Most of the thousands of organic compounds found in water are synthetic chemicals that are produced by human activities; • these included pesticides, solvents, industrial chemicals and plastics, and seepage from landfills. • Wide variety of affects – Toxic – Cancers – Genetic defects – Interferes w/ hormones • Growth & sexual development • Gender benders

Gender Benders A study by US Geological Survey & (USGS) US Fish & Wildlife Service • Study on male fish - released 12/2015 • Conducted at 19 national wildlife refuges in the Northeast U. S • 85% of male smallmouth bass and 27% of male largemouth bass were intersex • Intersex - one sex develops characteristics of the opposite sex • a global issue, as wild-caught fish affected by endocrine-disrupting chemicals have been found in locations across the world. • endocrine-disrupting chemicals can come from many sources, – Pharmaceuticals, agrochemicals, & industrial compounds that enter the waterways. – birth control pills, natural sex hormones in livestock manures, herbicides and pesticides. Bisphenol A (BPA), plasticizers, polychlorinated biphenyls (PCBs), DDT, and some other pesticides, dioxins • adverse developmental, reproductive, neurological, and immune effects in both humans and wildlife

• Pesticides – Developed during WWII • Herbicides, fungicides, insecticides – Unintended effects • Kill not just intended target, but wide variety – Mosquito insecticide kills amphibians • Alter other physiological functions – DDT: target insect nervous system – accumulates in food chain and thins eggshells • Inert ingredients – Additives aren’t necessarily harmless » Roundup additive to penetrate waxy leaves – ruptures tadpole gills

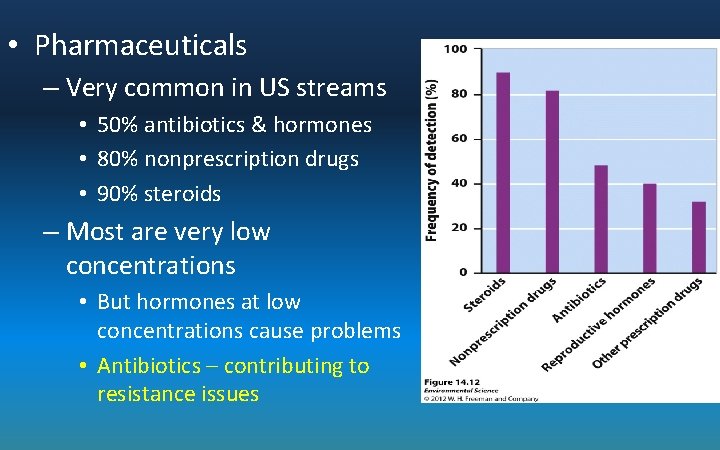
• Pharmaceuticals – Very common in US streams • 50% antibiotics & hormones • 80% nonprescription drugs • 90% steroids – Most are very low concentrations • But hormones at low concentrations cause problems • Antibiotics – contributing to resistance issues

• Military compounds – Perchlorates • In soil from rocket fuel – Affect thyroid & metabolism

Industrial compounds – Cuyahoga River in OH caught fire several times in 50’s and 60’s – mostly from industrial waste – PCB’s – polychlorinated biphenyls • To make plastic, insulation, • Lethal, persistent, bioaccumulates • Dump into rivers – settles in sediments • In 2009, GE finally dredge river from dumping 1947 -77 • No longer manufactured/used in US – Banned by Stockholm Convention in 2001 – dirty dozen – PBDE’s - polybrominated diphyenyl ethers • Flame retardants • Brain damage • Banned in EU and several states

Acid Rain • Sources - burning fossil fuels (SOx, NOx), especially coal (SOx) – In the eastern US, most acid rain is caused by SO 2 from coal burning – In So. Cal, most acid rain is caused by NOx emissions from cars – NOx + SOX + H 2 O H 2 SO 4 and HNO 3 • Effects – – Loss of the most sensitive species first (very young, very old) Increased leaching of metal from sediment Loss of biodiversity food web disruption HNO 3 can also cause eutrophication • Indicators – Normal rain is around 5. 5 -6. 5, acid rain is below 5. 5 – Reduced p. H in water bodies – Reduced soil p. H • Reduction – Remove SO 2 and NOx from emissions before release (install scrubbers on power plants) – Switching to less polluting alternative fuels – Add lime to aquatic ecosystems to absorb H+ ions (expensive)

Acid Mine Drainage

Thermal Pollution • Sources - industry, power plants, reduced water flow, increased sediment • Effects – increased temperatures reduce the amount of oxygen the water can hold, thus reducing DO levels and causing fish kills • Indicators – Increased water temperature – Decreased DO – Fish kills – Increased algae & bacteria growth • Reduction - reduce runoff, cool water before releasing, maintain adequate water flow, maintain healthy riparian zones for shade

Sediment Pollution • Sources - erosion, runoff from farms, construction, mining, etc • Effects – clogs waterways, blocks sunlight, may choke fish, increases water temperatures, fish kills • Indicators – Increased turbidity – Increased temperatures – Possible decrease in DO • Reduction – protect/replace riparian habitat, minimize erosion, practice soil conservation strategies, maintain wetlands around water bodies

• • Destruction of spawning beds (river/lake bottoms) Adsorption and transport of other pollutants Reduced light penetration, aquatic vegetation Greater nutrients loadings, oxygen demand

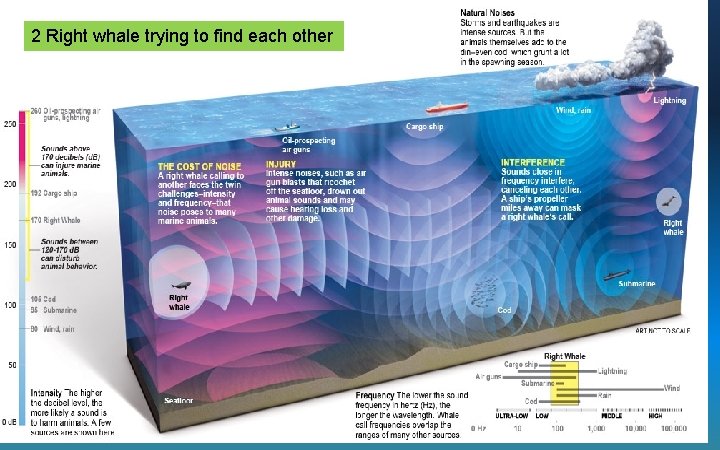
Noise Pollution • Sounds travels better underwater than thru air • Sounds from ships & subs – Especially sonar • Interferes with marine animal communication – Beached whales

2 Right whale trying to find each other


Groundwater Pollution • Over 50% of people in the U. S. rely on groundwater for drinking • pollutants leech into groundwater EPA - organic chemicals contaminate – – – Land fills agricultural lands underground storage tanks Deep well injection - fracking wastewater Impoundment ponds Superfund & brownfield sites about 45% of municipal groundwater supplies. By 2003, EPA - cleanup of 297, 000 underground tanks leaking gasoline, diesel fuel, home heating oil, or toxic solvents.

Groundwater Pollution • Sources - underground storage tanks, older unlined landfills, modern landfill leaks, buried waste, etc • Effects – aquifer contamination, drinking water contamination, plume movement into surface water • Indicators – Positive well tests • Reduction – line landfills and lagoons – bury waste in appropriate hazardous waste facilities – cleanup superfund and brownfield sites – replace underground storage tanks before leaks occur.

Oil Spills • Sources - 1. natural oil seeps 2. surface runoff 3. transport 4. extraction • Effects – See case studies • Indicators – Presence of oil byproducts including benzene, toluene, xylene, napthalene, benzo(a)pyrene • Reduction – Improve safety standards for extracting and transporting oil – Perform routine inspections – Require back-up systems and a disaster management plan in case of unexpected event – Research oil spill cleanup techniques prior to a spill

Effects of Oil Spills • Volatile Organics Compounds (VOCs) • immediately kill many aquatic organisms (especially plankton and larvae!) • Floating oil - coats birds and ocean mammals; reduces natural insulation and buoyancy, causing deaths • Heavy oil - sinks to ocean bottom and washes into estuaries where it contaminates crabs, oysters, mussels, clams, etc. • On beaches - harm intertidal life and cause economic losses to tourism and fishing industries


Cleaning Up an Oil Spill

Oil Cleanup Options • • • Place boom around - containment Skimmer boats - vacuum it up Absorbent material Dispersants (toxic? ) Coagulants Burn it off (surface only) GM Bacteria Let it sink to the bottom Wait until it eventually is decomposed naturally

• March 24, 1989, tanker in Prince William Sound, Alaska, worst oil spill in U. S. waters Exxon Valdez Oil Spill • The most ecologically destructive oil spill in history • Coated 1, 600 of shoreline • killed wildlife, and caused serious contamination • Exxon spent billions for clean up and fines © Brooks/Cole Publishing Company / ITP Water Resources and Water Pollution by Paul Rich

Deepwater Horizon, 2010 • • Pipe broke 1 mile deep Took 3 months to cap 206 million gallons of oil Effects will be seen for decades

Ocean Pollution • We have used Oceans as our trash can • The only substance that is illegal to dump anywhere in the ocean is plastic • NOAA Ocean Pollution

Solid Waste • Plastic pollution is a huge problem!


debris not visible to satellites. The ocean still looks blue because most of the junk is barely visible tiny bits of plastic. Microplastics - no bigger than 5 mm from many sources. broken bits of larger plastic pieces, synthetic fibers from clothing, plastic beads from toothpastes and face washes that escape cleaning filters at wastewater treatment plants & end up in ocean. These small pieces of plastic – collect microbes not normally in seawater, absorb & concentrate pollutants – all enter food chain

Cleaning the ocean Baltimore’s Mr. Trash Wheel removed at least 213 tons of trash from the city’s Inner Harbor last year, stopping plastic and other debris before it reached the ocean. Its 2015 roundup included 118, 670 plastic bottles and 2. 6 million cigarette butts. V-shaped array of floating barriers to collect ocean plastic. With the help of ocean currents, plastic pieces concentrate in the center of the V and are scooped up by a conveyer belt driven by solar panels and dropped in a collecting station for recycling.

Preventing Water Pollution • Protect wetland riparian habitat • Use less chemicals, energy, and manufactured products that contain plastic or metal • Minimize the use of agricultural chemicals (inorganic fertilizer, pesticide, soil stabilizers, etc) • Develop effective storm water runoff systems • Develop and enforce strict safety requirements for oil drilling and transport • Minimize runoff from all sources • Treat contaminated water prior to release into the environment (waste water treatment)

Cleaning Up Polluted Water • Immediately reduce or eliminate pollution discharges into the water body • Bacteria can be used to clean up organic or oil pollution • Dredge contaminated soil out of the water body – will temporarily increase water pollution

Sewage Treatment ‣ Sewage includes all waste water that has been used by a household or industry. It does not include storm-water from road and property runoff, which is usually diverted directly into waterways. In some cities, sewage and storm-water systems may be partly combined, and sewage may overflow into storm-water during high rainfall. Trickling filters at a waste treatment plant Sewage pipes discharging into a holding tank

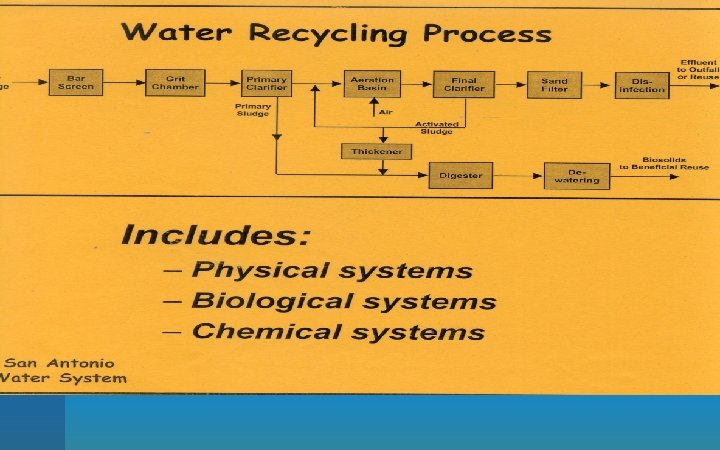
Ways to handle wastewater • Same basic approach to all. – Break down organic matter with bacteria turning it into carbon dioxide and inorganic compounds. • Two most widespread systems are the – septic system – sewage treatment plant. • Manure from livestock fields usually handled by a manure lagoon.

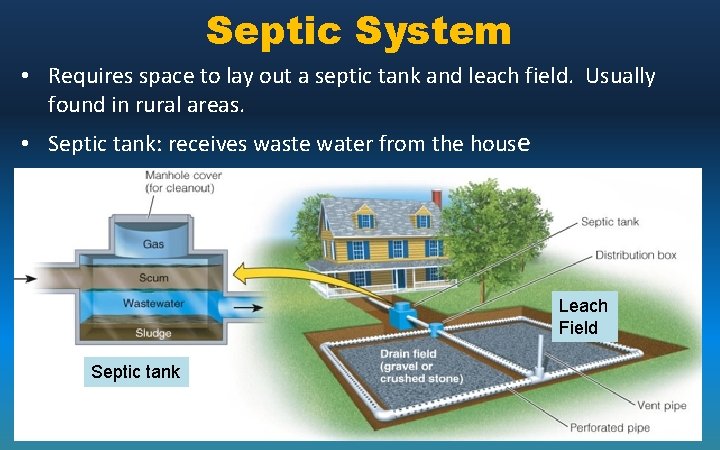
Septic System • Requires space to lay out a septic tank and leach field. Usually found in rural areas. • Septic tank: receives waste water from the house Leach Field Septic tank

• Harmful pathogens settle in sludge , decompose in tank or degraded by soil microbes in leach field – Three layers develop: – Scum - floats – Septage Flows to leach field – Sludge-pumped out every 5 -10 yrs. Septic System – Leach field – pipes with small holes – Wastewater slowly seeps and is filtered and decomposed – Nutrients are recycled by other plants / organisms

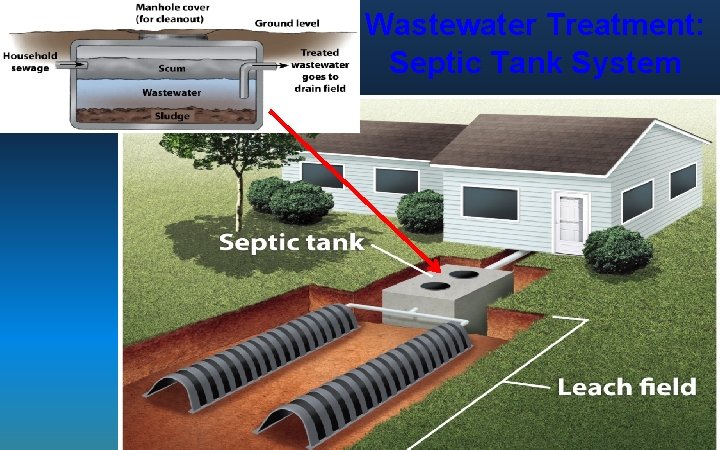
Wastewater Treatment: Septic Tank System 81

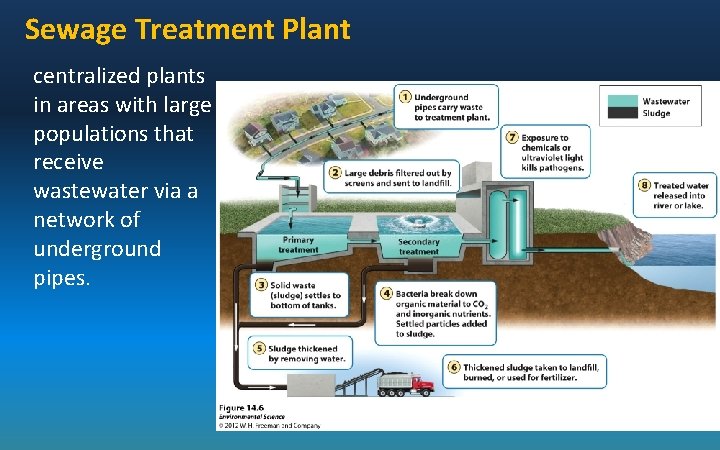
Sewage Treatment Plant centralized plants in areas with large populations that receive wastewater via a network of underground pipes.


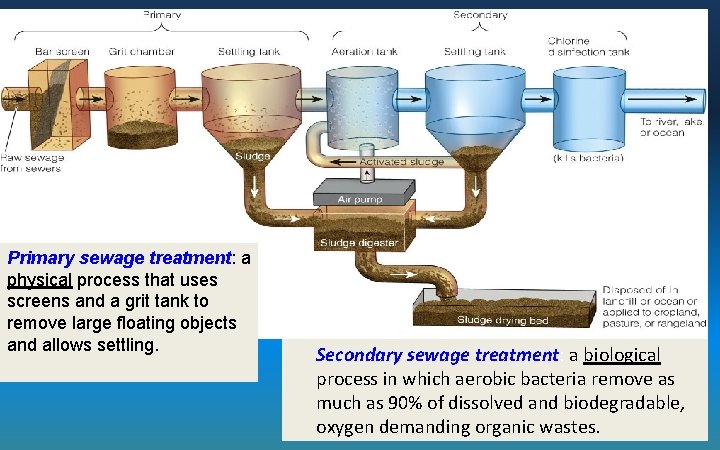
Primary sewage treatment: a physical process that uses screens and a grit tank to remove large floating objects and allows settling. Secondary sewage treatment: a biological process in which aerobic bacteria remove as much as 90% of dissolved and biodegradable, oxygen demanding organic wastes.

Disinfection Options • Chlorine – the most common disinfectant – – not effective against cryptosporidium can create dangerous byproducts must be removed prior to discharge into ecosystems Residual left in water for residual disinfection (pro/con – depends) • UV – new technology, very effective, damages DNA – Can catalyze undesirable reactions – Ineffective against adenoviruses – No residual disinfection (for drinking water) • Ozone – new technology – – more expensive ozone is a regulated air pollutant No residual disinfection (for drinking water) Can form unintended byproducts

Problems with Waste Water Treatment • Water is often removed from one source and returned to another, this is unsustainable (open loop) • Many municipalities only use primary and secondary treatment for residential waste water – Nitrogen, phosphorus, pharmaceuticals, synthetic organics not removed. – Need expensive tertiary step • may use no treatment for storm water • Expensive to build and maintain

Animal Wastewater Concentrated Animal Feeding Operations Feed Lots - Manure lagoons- large, human-made ponds lined with rubber to prevent the manure from leaking into the groundwater. After the manure is broken down by bacteria, it is spread onto fields as fertilizers.

Using Nature to Purify Sewage • Ecological wastewater purification by a living machine. – Uses the sun and a series of tanks containing plants, snails, zooplankton, crayfish, and fish (that can be eaten or sold for bait). Figure 21 -1

Reducing Water Pollution through Sewage Treatment • Natural and artificial wetlands and other ecological systems can be used to treat sewage. – California created a 65 hectare wetland near Humboldt Bay that acts as a natural wastewater treatment plant for the town of 16, 000 people. • The project cost less than half of the estimated price of a conventional treatment plant.

Green Waste Water • Modern waste water treatment plants expensive • Use a series of greenhouses & wetlands - Purify water: effective & affordable – Aesthetically pleasing – no odor – Many of the same processes of traditional treatment plants • Waste water flows into holding tanks • Pumped into series of transparent tanks • Microbes, snails algae, plants – grow & consume nutrients • Air is bubbled thru to get aerobic decomp - Anaerobic decomp = smell • Liquid coming out held – solids settle • Water is filtered thru sand then artificial wetlands • Denitrifying bacteria removes Nitrogen • Water is exposed to UV light to kill viruses & bacteria • Released to leach field to by filtered by soil

Water Quality Laws

Clean Water Act • The Clean Water Act is a 1977 amendment to the Federal Water Pollution Control Act of 1972 – regulates discharges of pollutants in the US – EPA to set water quality standards for industry and for all contaminants in surface waters • Designed to protect fishing, swimming and the ecosystem health • Goal: Make lakes & streams fishable & swimmable

Clean Water Act • Sets up the National Pollutant Discharge Elimination System (NPDES) – Regulates point sources of pollution by requiring a permit to discharge any pollutant into a navigable waterway. • Total Maximum Daily Load (TMDL) – sets daily limits for total pollutant discharges into impaired water bodies – Set based on the maximum pollution levels the water body can receive and still meet water quality standards


Safe Drinking Water Act • The Safe Drinking Water Act (1974) was established to protect the quality of drinking water in the U. S • This law focuses on all waters actually or potentially designed for drinking use, whether from above ground or underground sources

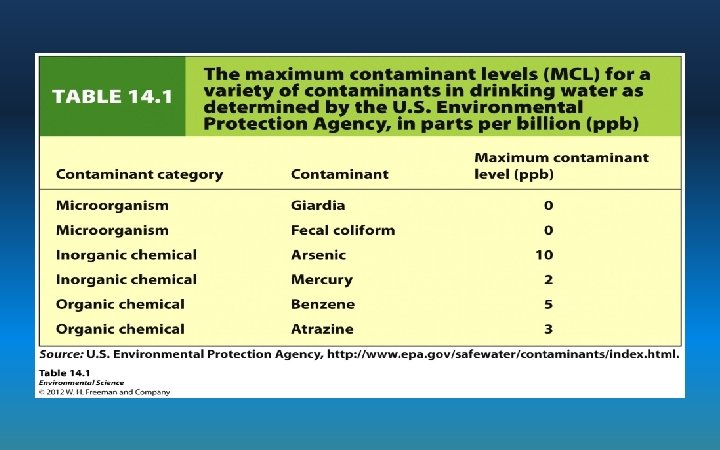
Safe Drinking Water Act • EPA sets Maximum Contaminant Levels (MCLs) for drinking water • Standards for dozens of contaminants – Subjective and sensitive to political pressure – Industry & cities: resist reducing levels - say it’s too expensive • http: //www. epa. gov/safewater/contaminants/index. html#mcls Pollutant Standard Health Impact Nitrate 10 mg/L Fertilizers: Blue Baby Syndrome Lead 0. 015 mg/L Lead pipes: Neurotoxin, decreased IQ Chloramines 4 mg/L Byproduct of chlorine disinfection: eye and nose irritant, stomach discomfort PCBs 0. 0005 mg/L Industrial processes: increased cancer risk, reproductive issues


Pollution in Developing Countries • Water in many of central China's rivers are greenish black from uncontrolled pollution by thousands of factories. Figure 21 -5

India’s Ganges River: Religion, Poverty, and Health • Daily, more than 1 million Hindus in India bathe, drink from, or carry out religious ceremonies in the highly polluted Ganges River. Figure 21 -6

India’s Ganges River: Religion, Poverty, and Health • Religious beliefs, cultural traditions, poverty, and a large population interact to cause severe pollution of the Ganges River in India. – Very little of the sewage is treated. – Open defecation (“more temples than toilets”) – Hindu believe in cremating the dead to free the soul and throwing the ashes in the holy Ganges. • Some are too poor to afford the wood to fully cremate. • Decomposing bodies promote disease and depletes DO.