Water Pollution Equipment for water pollution control 11
























- Slides: 34

Water Pollution / Equipment for water pollution control (11) Waste water treatment of colored waste water July 6, 2012 Japan Environmental Management Association for Industry (JEMAI) 1

CONTENT 1.Decolorization treatment of colored waste water 1.1 Physicochemical treatment 1.2 Biological treatment 1.3 Comparison of decolorization methods and their effect 2.Decomposition of waste liquid by incineration 3.Biological decolorization system of colored wastwater 4.Decolorization of waste water by using iron 5.Powder type coagulant made by natural mineral 2

1.Decolorization treatment of colored waste water 1.1 Physicochemical treatment (1) Coagulation-sedimentation method ・ Target particle size is about 0. 001~1 μm. (colloidal particle ) → Particle surface is charged negative. (Electrostatically particles repulsed each other. , zeta potential) → By adding positive ion to neutralize negative charge on particle surface, coagulation starts. By Van der Waal force particles begin coagulation. ・ Inorganic coagulant and organic coagulant. ・ Coagulant has adsorption function of generated gel. ・ The stirring of 1 st stage for neutralization of electricity should be done at high speed and 2 nd stage is at slow speed. 3

・Organic coagulant (Polymer coagulant) has function to neutralize electricity on particle surface and to bridge fine flock to become large flock. → Use of coagulant itself or co-use with inorganic coagulant such as aluminum sulfate. ・Need to confirm effect of decolorization with jar test in case of colored waste water. 4

(2) Coagulation-floatation method ・ Coagulate particles suspended in water then make air bubble adhere on the surface of coagulated particles to reduce specific gravity for floatation separation. ・ The treating time of coagulation-floatation is shorter than that of coagulation-sedimentation. (about 1~ 30 minutes. ) ・ Depending on the kinds of waste water coagulation-floatation can obtain better results than those of coagulation-sedimentation. 5

(3) Activated carbon adsorption method ・ Activated carbon has strong adhesive force for organic matters. ( hydrophobic property ) → It can apply to organic matters and decolorization of waste water which can not be removed or decomposed with coagulated- sedimentation and filtration ・ The lower solubility, the weaker ionicity, the larger particle size, and the higher concentration of absorbate in water, the larger adsorbed amounts on activate carbon are. ・ In general the adsorbed amount become much at p. H 2~3. → The effect of water temperature is small. 6

(4) Ozone oxidation method ・ Self decomposition of Ozone → In dry air half-life period of 1 wt% concentration of ozone is about 16 hours. At p. H 7 half-life period of 3 wt ppm concentration of ozone is about 30 minutes. The higher p. H is, the shorter half-life period is. ・ Atomic oxygen generated by decomposition of ozone has strong oxidation force. ・ Organic matters easily affected by oxidation of ozone Unsaturated compound which have C=C or C≡C bonds. Compounds which have aromatic ring. Amine, Sulfide, Sulfonic acid, Phosphate compound, Alcohol, Ether, Aldehyde. 7

・ Decoloring property Hydrophilic dyestuffs and hydrophobic dye tuffs (Disperse dyes ) → Hydrophilic dyestuff are highly affected by decolorization by ozone. Dyestuffs which have good decolorization property by ozone. Reactive dyestuff, Cationic and acidic hydrophilic dyestuff Dyestuffs which have bad decolorization property by ozone. Disperse dyestuff (Vat dyestuff, Sulphur dyestuff, Naphthol dye stuff, Oil color ) 8

(5) Fenton oxidation method ・ Utilize oxidation force of hydroxyl radical generated from hydrogen peroxide. Hydrogen atom abstraction reaction R-H + ・OH → ・R + H 2 O R : alkyl group X-: anion Charge transfer reaction X- + ・OH → ・X + OH- Addition reaction of hydroxyl radical to unsaturated bond. ・ By the reaction with hydroxyl radical, organic matters are decomposed by oxidation reaction to CO 2, H 2 O or low-grade organic acids such oxalic acid and acetic acid. 9

・Organization of system : Consist of three steps of Fenton oxidation step, neutralization step and sedimentation step. → Add hydrogen peroxide, ferrous chloride and sulfuric acid in to oxidation vessel. → Oxidize ferrous chloride to ferric chloride by adjusting p. H with caustic soda etc. → Add coagulation-sedimentation agent. (Solid-liquid separation) ・As catalyst ferrous salts are generally used because which price is cheep. →The reaction speed is higher at acidic condition. ((Addition of sulfuric acid. ) ・At waste water treatment organic matters are decomposed by oxidative reaction by mixing waste water with hydrogen peroxide and iron catalyst then neutralize the solution to precipitate iron catalyst as iron hydroxide and remove them. 10

・ Possible to remove offensive components and inorganic COD such as nitrous acid, sulfurous acid, sulfide and thiocyanic compounds. ・ Adoption of biological treatment at before-stage is effective to I increase efficiency. 11

(6) Electrolytic oxidation method (a) Oxidation-decomposition method ・As the waste water of dye effluent contains chlorine ion or chlorite ion these chlorine or chlorite ion are oxidized to chlorine gas by electrolysis. Then by using oxidation force of chlorine gas the waste water of dye effluent is decolorized. ・Chlorine gas reacts with hydroxide ion which generates at cathode to generate hypochlorite ion. This hypochlorite ion decolorizes color components by oxidation-decomposition reaction. Cathode reaction 2 Cl- → Cl 2 + H 2 Anode reaction H 2 O + 2 e → 2 OH- + H 2 Chemical reaction in solution Cl 2 + 2 OH-→ 2 ClO- + H 2O Decolorization reaction Cl. O- → Cl- + (O) + colored substances → decolor (bleach) 12

(b) Electrisys-floatation method ・ In waste water when turning on weak electricity between electrodes water is electrolyzed to generate oxygen gas at anode and generate hydrogen gas at cathode. As this electrolyzed gas is very fine and generate much amount the much amount gas adheres on SS. → Rising rate of flock is fast. ・ On the surface of electrode cathodic oxidation and anodic reduction proceed, then because of removal of COD and BOD the degree of color decrease also. ・ Cathode material : lead dioxide electrode Anode material : stainless steel electrode 、 13

1.2 Biological treatment (1) Aerobic biological treatment ・ Activated sludge can remove colored materials to some extent by adsorption. → At beginning stage hydrophobic dyestuff (sulphur dyestuff and disperse dyestuff) can be much removed by activated sludge by adsorption. In case of hydrophilic dyestuff removal by adsorption is expected but biological decolorization is difficult. (Some cases exist by using well domesticated activated sludge. ), (2) Anaerobic biological treatment 14

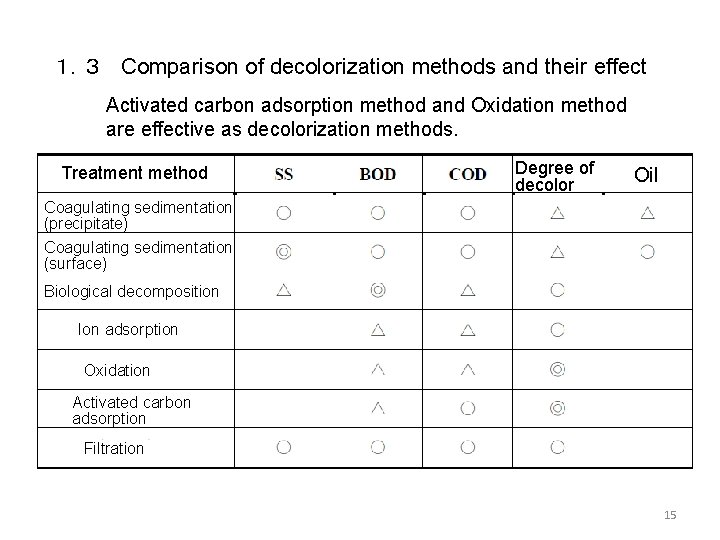
1.3 Comparison of decolorization methods and their effect Activated carbon adsorption method and Oxidation method are effective as decolorization methods. Treatment method Degree of decolor Oil Coagulating sedimentation (precipitate) Coagulating sedimentation (surface) Biological decomposition Ion adsorption Oxidation Activated carbon adsorption Filtration 15

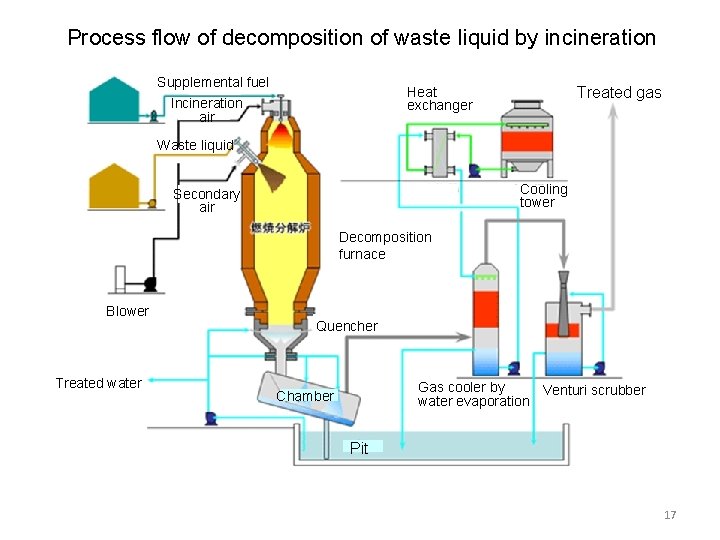
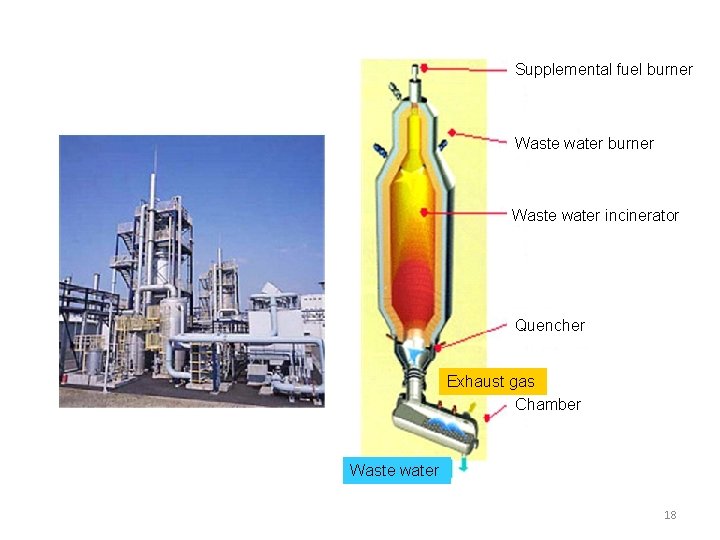
2.Decomposition of waste liquid by incineration Source:Asahi chemical engineering (1) Outline ・By spraying waste water into furnace organic matters are immediately oxidized and decomposed and inorganic matters melt. ・At quencher and chamber located bottom part of furnace, exhaust gas and melted salt were rapidly cooled. ・In gas cooler by cooling exhaust gas with water prevent the generation of white smoke (steam). Absorb acid gas. ・Venturi scrubber can catch particulate matter. (2) Feature ・ Possible to apply to high concentration organic waste liquid and colored waste liquid. ・ Possible to apply to the waste liquid of high concentration inorganic salt (pigment). ・ Waste acid liquid and waste alkaline liquid can be treated. 16

Process flow of decomposition of waste liquid by incineration Supplemental fuel Treated gas Heat exchanger Incineration air Waste liquid Cooling tower Secondary air Decomposition furnace Blower Treated water Quencher Gas cooler by Venturi scrubber water evaporation Chamber Pit 17

Supplemental fuel burner Waste water incinerator Quencher Exhaust gas Chamber Waste water 18

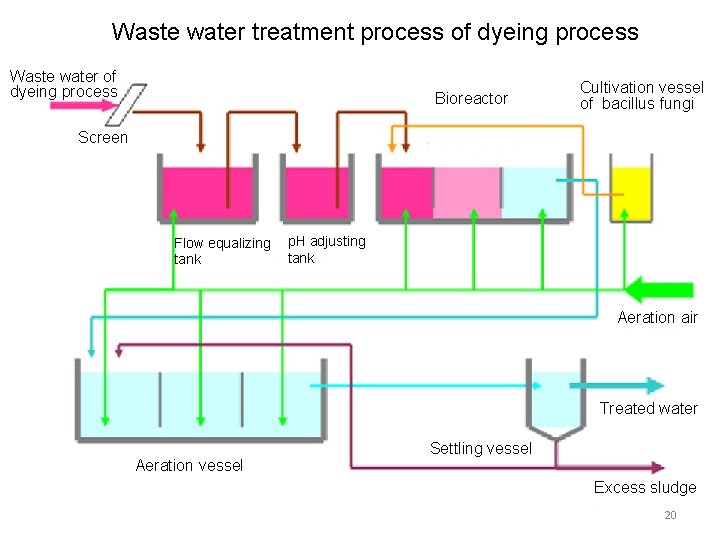
3.Biological decolorization system of colored wastwater Source:HP of Mikiriken Industrial Co. , LTD ・This system is that by using bacillus fungi which exists in soil, withered grass and even in human body, colored waste water containing azo dyestuff which is used at most of dye works can be decolorized biologically. ・ Cultivate bacillus fungi in system then introduce cultivated bacillus fungi into bioreactor (decolorizing tank) and decompose dyestuff. The combination of following activated sludge vessel can remove BOD components etc. 19

Waste water treatment process of dyeing process Waste water of dyeing process Bioreactor Cultivation vessel of bacillus fungi Screen Flow equalizing tank p. H adjusting tank Aeration air Treated water Aeration vessel Settling vessel Excess sludge 20

Electron micrograph of Bacillus OY 1 -2 21

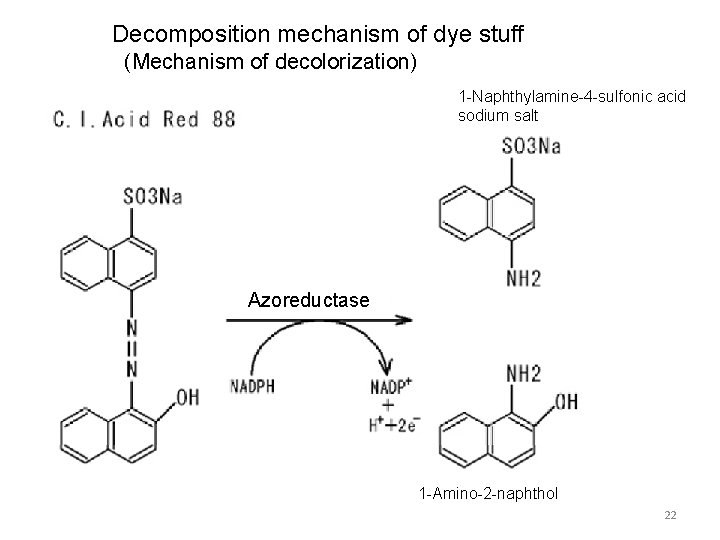
Decomposition mechanism of dye stuff (Mechanism of decolorization) 1 -Naphthylamine-4 -sulfonic acid sodium salt Azoreductase 1 -Amino-2 -naphthol 22

Carrier of bacillus fungi Enlarged photo of carrier of bacillus fungi by one hundred times 100 23

Test results of decolorization Test result under anaerobic condition (more effective than aerobic condition) Test result of decolorization of dye stuffs Dye stuffs Number of samples Number of decolorized Reactive dyestuffs Disperse dyestuffs Threne dyestuffs About 90% of Reactive dyestuffs and Disperse dyestuffs and 50% of Threne dyestuffs were decolorized. 24

Reactive dyestuffs Red reactive dyestuffs Blue reactive dyestuffs Yellow disperse dyestuffs Red disperse dyestuffs Disperse dyestuffs 25

Threne dyestuffs Blue threne dyestuffs Red threne dyestuffs 26

Test result of actual waste waater of dye works ・Test result of waste water containing various kinds of dyes. Before treatment After treatment 27

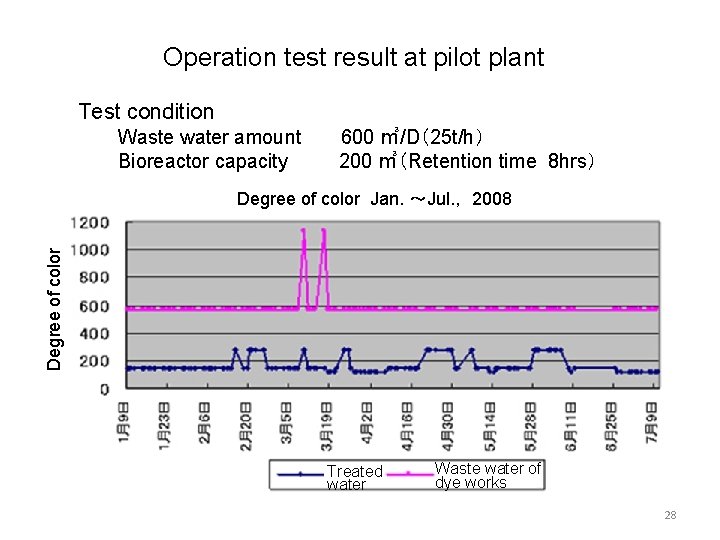
Operation test result at pilot plant Test condition Waste water amount Bioreactor capacity 600 ㎥/D(25 t/h) 200 ㎥(Retention time 8 hrs) Degree of color Jan. ~Jul. , 2008 Treated water Waste water of dye works 28

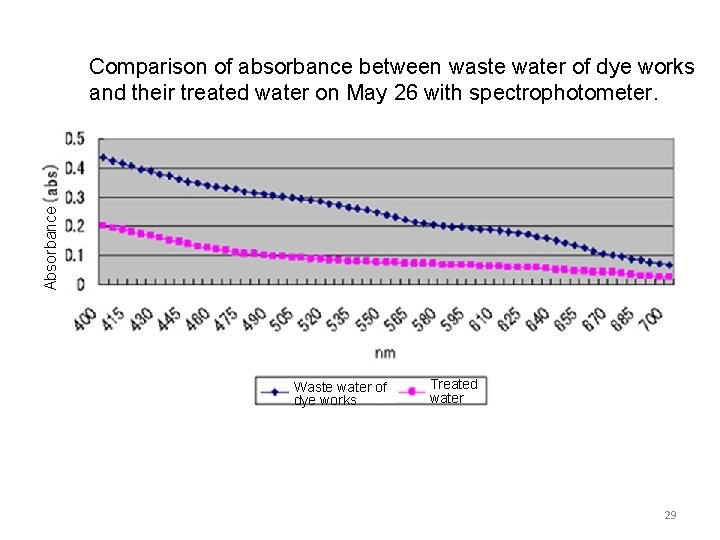
Absorbance Comparison of absorbance between waste water of dye works and their treated water on May 26 with spectrophotometer. Waste water of dye works Treated water 29

Analytical data of BOD and COD Waste water of dye works Treated water 30

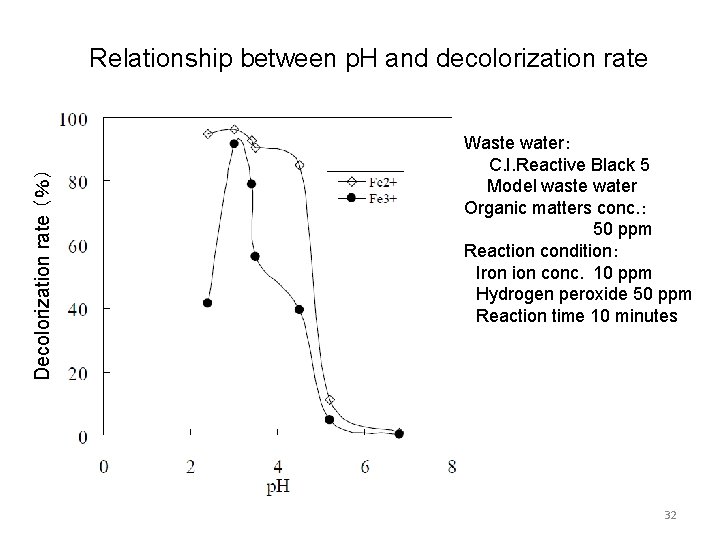
4.Decolorization of waste water by using iron Fenton reaction (one explanation) Fe 2+ + H 2 O 2 → Fe 3+ + OH- + ・OH Fe 3+ + H 2 O 2 → Fe 2+ + ・OOH + H+ (Regeneration of Fe 2+) But Fe 3+reacts with hydroxide ion to generate iron oxide which precipitates. Then when all Fe 2+ions are consumed the reaction finishes. Fe 3+ + n OH- → amorphous iron oxide ↓ ・ Fenton reaction is largely affected by p. H. 31

Decolorization rate (%) Relationship between p. H and decolorization rate Waste water: C. I. Reactive Black 5 Model waste water Organic matters conc. : 50 ppm Reaction condition: Iron ion conc. 10 ppm Hydrogen peroxide 50 ppm Reaction time 10 minutes 32

5.Powder type coagulant made by natural mineral Source:HP of KU・system Co. , Ltd ・ Powder of natural mineral ・ AQUARIA POP IN-100 ・ Experimental result of waste water of water based ink Add coagulant then start stirring. coagulant : 2, 000 ppm rotational frequency : 180 rpm 5 minutes later. Begin solid-liquid separation. 7 minutes later. Treated water after filtration. Stop stirring. Immediately start coagulation and settling. . 33

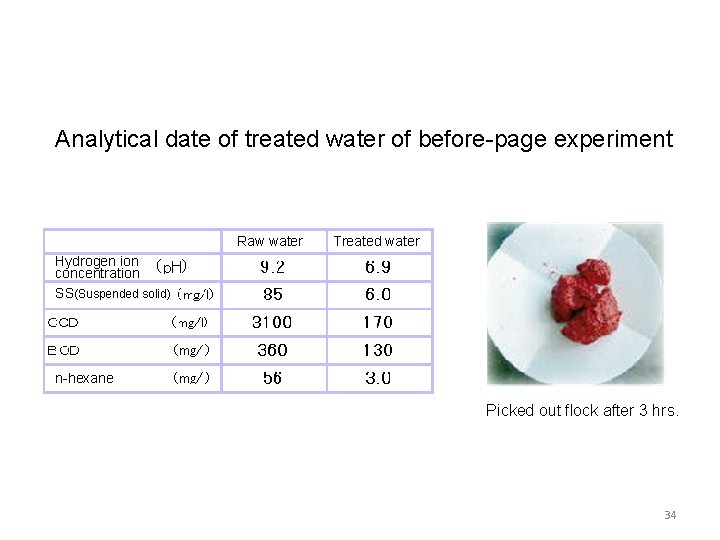
Analytical date of treated water of before-page experiment Raw water Treated water Hydrogen ion concentration SS(Suspended solid) n-hexane Picked out flock after 3 hrs. 34