Water molecules are polar and hydrogen bonds form

• Water molecules are polar and hydrogen bonds form between them. • Hydrogen bonding and dipolarity explain the cohesive, adhesive, thermal and solvent properties of water. • Application: Use of water as a coolant in sweat. • Guidance: Students should know at least one example of a benefit to living organisms of each property of water. 1

Molecules 1: Properties of water Objectives Describe how hydrogen bonding and dipolarity explain the special properties of water. Explain how water’s special properties benefit living organisms Warm Up What are hydrogen bonds? In table groups, work out your best definition. Please change title of previous lesson to “Molecules 0” 2

What are hydrogen bonds? 3

What are hydrogen bonds? 4
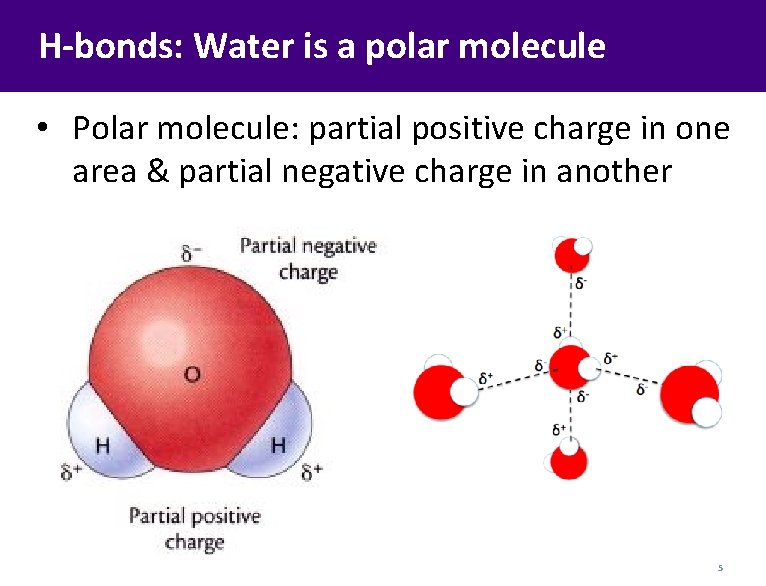
H-bonds: Water is a polar molecule • Polar molecule: partial positive charge in one area & partial negative charge in another 5
H-bonds: Opposites charges attract 6

H-bonds: Definition • Hydrogen bond = attractive force between hydrogen attached to electronegative atom of one molecule and electronegative atom of a different molecule. 7

H-bonds: Visual representation 8

Today: Special properties of water • • Cohesion Adhesion High heat of vaporization Solvent 9

Today: By the end of the recitation: • Each student will leave with 4 flash cards describing 4 special properties of water • Flashcards are in notebook: tape or make pocket 10
Revised jigsaw: expert pairs A A B B C C D D 11

Revised jigsaw: mixed SUPERTABLES! A A C C B B D D 12

Flashcards Property of water Sketch/diagram Definition Benefit to living orgs. ________________________ ________________________ 13

Flashcard guidelines: • Flashcards are brief bullet points, not paragraphs • Given picture is not always the best sketch/diagram! 14

Guidelines for experts and learners: • Experts explain verbally and answer questions • Learners ask clarifying questions 15

Flashcards Property of water Sketch/diagram Definition Benefit to living orgs. ________________________ ________________________ 16

Expert order • • Expert 1: Cohesion Expert 2: Adhesion Expert 3: High heat of vaporization Expert 4: Solvent 17

18
- Slides: 18