WATER INDUSTRY IWA Specialist Conference Valladolid Spain May









![Catalyst reuse and stability (BVAl. Fe 2 C-Et. OH 25 -0. 5 g/L) [Fe]leached Catalyst reuse and stability (BVAl. Fe 2 C-Et. OH 25 -0. 5 g/L) [Fe]leached](https://slidetodoc.com/presentation_image_h2/0ebf8d2ae145cd7706d619c2c03a8a4d/image-18.jpg)



- Slides: 21

WATER & INDUSTRY IWA Specialist Conference. Valladolid, Spain. May - 2011 Removal of natural organic matter from a surface feedstock for drinking water production by Al/Fe-PILC – catalyzed wet peroxide oxidation: Effect of catalyst preparation from concentrated precursors L. A. Galeano, P. F. Bravo, C. D. Luna, M. A. Vicente, A. Gil, M. Rosero, S. X. Delgado, G. Rodríguez Corresponding Author E-mail: alejandrogaleano@udenar. edu. co 1

Outline • • • Introduction (NOM; AOPs; PILCs) Materials and Methods Results Conclusions Perspectives Acknowledgments 2

Introduction 3

Phenol, substituted phenols, light carboxylic acids, azo-dyes, PCBs, organo-chlorinated compounds, pesticides, herbicides, VOCs, hormones, etc. Natural water resources NOM Negative impact on surrounding ecosystems and humans even under trace concentrations “Poorly defined mix of organic substances with variable properties in terms of acidity, MW and molecular structure” * Toxic and hazardous compounds. Biorefractory Predominantly phenolic and carboxylic functionalities, plus alcohol, purine, amine and ketone groups * Sharp et al. (2006); Sci. Total Environ. 363, 183 -194. DBPs precursors 4

Problems derived from the presence of NOM in drinking water treatment processes* Negative effect on water quality by colour, taste and odor problems 2. Increased coagulant and disinfectant dose requirements, which in turn results in increased sludge and potential harmful formation of DBPs. 3. Promoted biological growth in distribution systems, and 4. Increased levels of complexed heavy metals and adsorbed organic pollutants. 1. * Matilainen & Sillanpää (2010); Chemosphere 80, 351 -365. 5

Conventional technologies for the removal of organic contaminants Biological processes Several kinds of bacteria strains, but…. Ø ~ 97% max. Removal Ø Slow degradation Ø Sludge is formed Ø Strongly inhibited in the presence of highly toxic substrates Separation techniques Ø Ø Extraction with solvents Phase transfer …. . only affordable for very high OM contents Coagulation – flocculation processes Membrane filtration 6

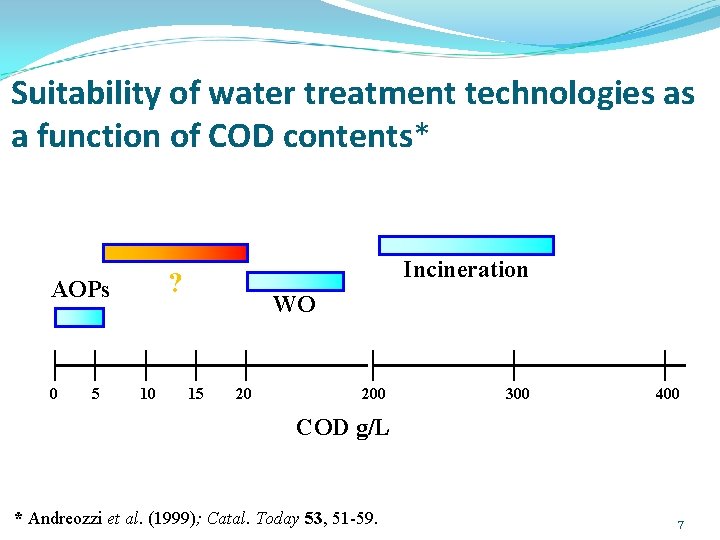
Suitability of water treatment technologies as a function of COD contents* ? AOPs 0 5 Incineration 10 WO 15 20 200 300 400 COD g/L * Andreozzi et al. (1999); Catal. Today 53, 51 -59. 7

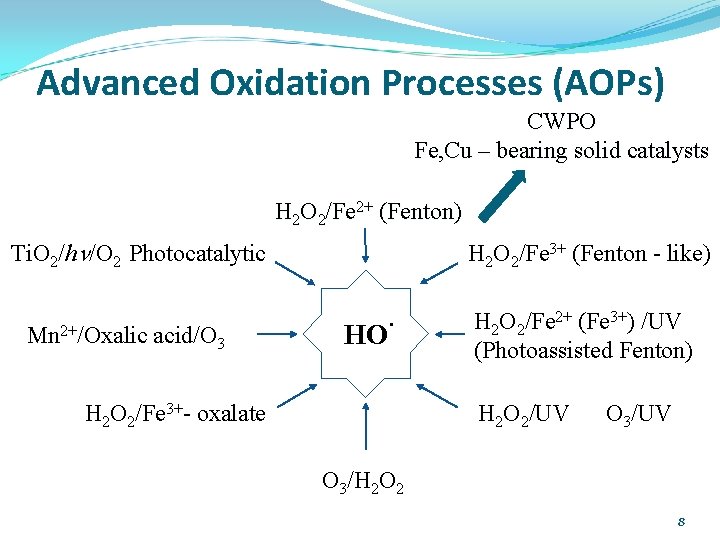
Advanced Oxidation Processes (AOPs) CWPO Fe, Cu – bearing solid catalysts H 2 O 2/Fe 2+ (Fenton) Ti. O 2/hn/O 2 Photocatalytic Mn 2+/Oxalic acid/O 3 H 2 O 2/Fe 3+ (Fenton - like) HO· H 2 O 2/Fe 3+- oxalate H 2 O 2/Fe 2+ (Fe 3+) /UV (Photoassisted Fenton) H 2 O 2/UV O 3/H 2 O 2 8

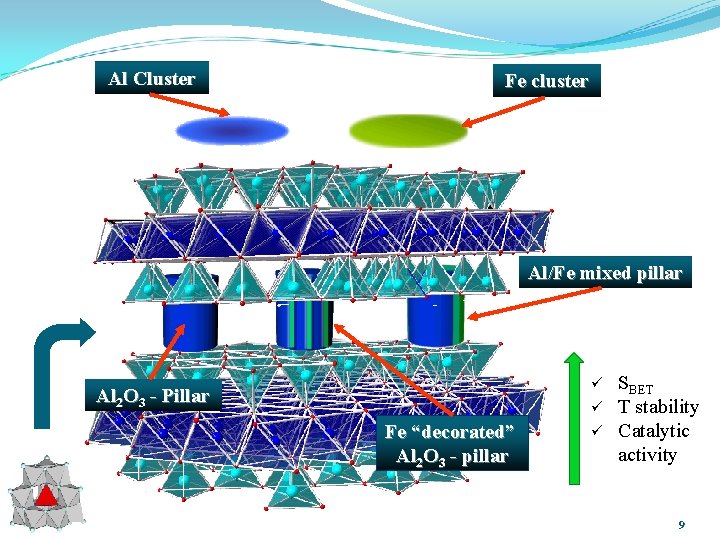
Al Cluster Fe cluster Al/Fe mixed pillar ü Al 2 O 3 - Pillar ü Fe “decorated” Al 2 O 3 - pillar ü SBET T stability Catalytic activity 9

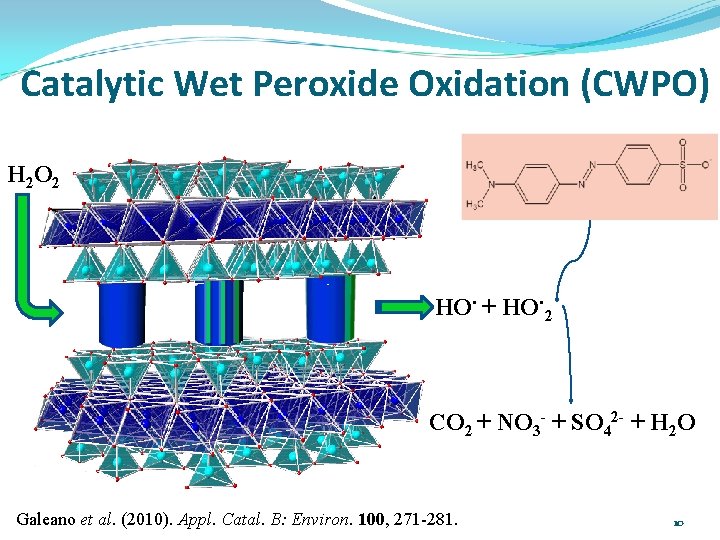
Catalytic Wet Peroxide Oxidation (CWPO) H 2 O 2 HO. + HO. 2 CO 2 + NO 3 - + SO 42 - + H 2 O Galeano et al. (2010). Appl. Catal. B: Environ. 100, 271 -281. 10

Materials and methods 11

Starting materials and methods Starting clay: clay Natural Ca-bentonite (BV); Mostly dioctahedral character; Montmorillonite type. CEC = 89 meq. /100 g. Chemical composition: 60. 5% Si. O 2, 24. 7% Al 2 O 3, 10. 2% Fe 2 O 3, 0. 05% Mn. O, 3. 07% Mg. O, 0. 64% Ca. O, Ca. O 0. 20% Na 2 O, 0. 58% K 2 O Water sample: sample Pasto River. COD = 40. 14 mg/L; Colour 455 = 42 PCU; TStored = 4 °C Catalytic runs: runs Semi-batch reactor. Catalyst loading = 0. 5 g/L or 5. 0 g/L; [H 2 O 2]added = 0. 047 mol/L; H 2 O 2 addition flow rate = 6. 0 m. L/h; [H 2 O 2]/[COD]0 = 1. 0; T = 18 ± 2. 0 ºC; P = 0. 7 atm. ; Sampling = 4. 0 m. L; treaction = 240 min; p. H = 3. 7. Analytical methods: methods COD: Closed reflux colorimetric method 5220 D True colour: Spectrophotometric single wavelength method 2120 C at 455 nm. [Fe]240 min: AAS 12

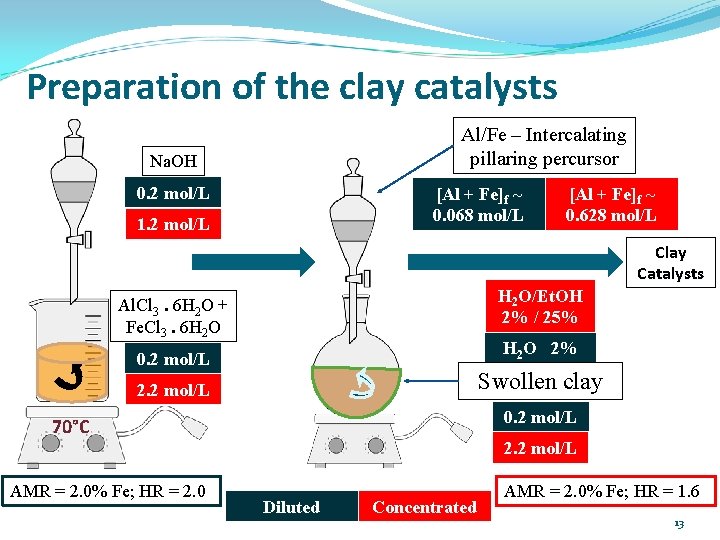
Preparation of the clay catalysts Al/Fe – Intercalating pillaring percursor Na. OH 0. 2 mol/L [Al + Fe]f ~ 0. 068 mol/L 1. 2 mol/L [Al + Fe]f ~ 0. 628 mol/L Clay Catalysts H 2 O/Et. OH 2% / 25% Al. Cl 3. 6 H 2 O + Fe. Cl 3. 6 H 2 O 2% 0. 2 mol/L Swollen clay 2. 2 mol/L 0. 2 mol/L 70°C AMR = 2. 0% Fe; HR = 2. 0 2. 2 mol/L Diluted Concentrated AMR = 2. 0% Fe; HR = 1. 6 13

Results 14

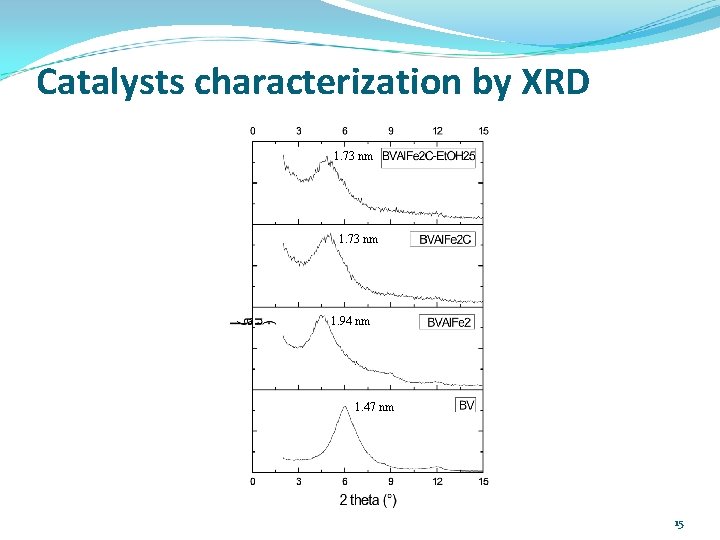
Catalysts characterization by XRD 1. 73 nm 1. 94 nm 1. 47 nm 15

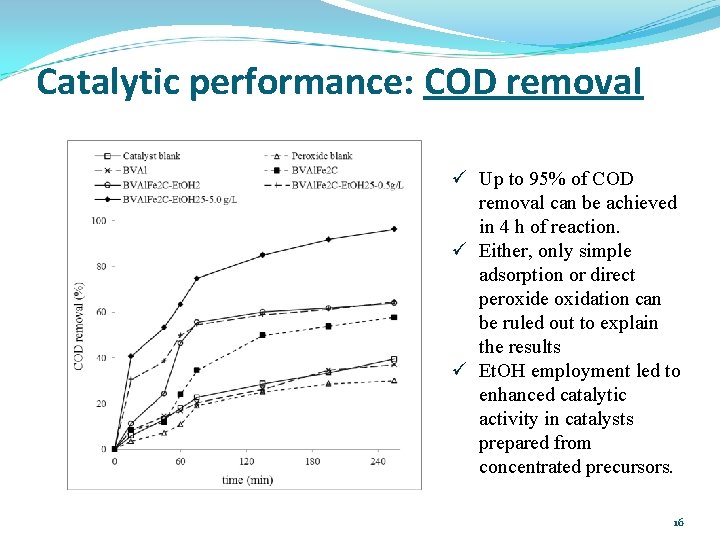
Catalytic performance: COD removal ü Up to 95% of COD removal can be achieved in 4 h of reaction. ü Either, only simple adsorption or direct peroxide oxidation can be ruled out to explain the results ü Et. OH employment led to enhanced catalytic activity in catalysts prepared from concentrated precursors. 16

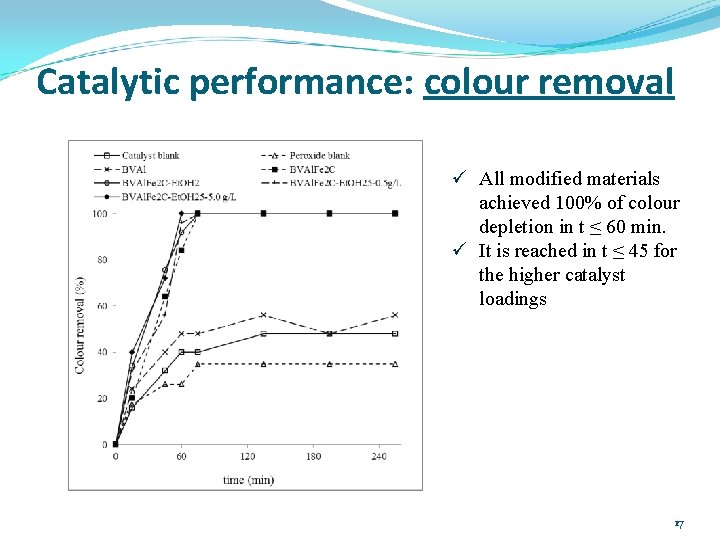
Catalytic performance: colour removal ü All modified materials achieved 100% of colour depletion in t ≤ 60 min. ü It is reached in t ≤ 45 for the higher catalyst loadings 17
![Catalyst reuse and stability BVAl Fe 2 CEt OH 25 0 5 gL Feleached Catalyst reuse and stability (BVAl. Fe 2 C-Et. OH 25 -0. 5 g/L) [Fe]leached](https://slidetodoc.com/presentation_image_h2/0ebf8d2ae145cd7706d619c2c03a8a4d/image-18.jpg)
Catalyst reuse and stability (BVAl. Fe 2 C-Et. OH 25 -0. 5 g/L) [Fe]leached < 0. 5 mg/L 18

Conclusions Al/Fe-pillared clays from concentrated precursors for CWPO NOM in surface water. Catalyst preparation from concentrated both ethanol-based suspension and mixed Al/Fe oligomeric precursor was advantageous by lower suspension V and improved catalytic performance. Catalyst exhibited excellent NOM removal from raw surface water: Depleted over 95 % of the COD in t = 4 h and 100 % of the true colour 455 in t < 45 min of reaction. High stability of the catalysts against chemical leaching of the active metal along several catalytic tests. 19

Perspectives Optimization studies are still needed mainly for the following reaction factors: Catalyst loading, concentration and addition rate of peroxide solution. Transition from batch to continuous reactor should be studied. A more detailed NOM characterization could be useful to improve the catalytic performance of CWPO reaction on surface feedstock waters. Further research to establish the true bactericidal potential of the technique could provide still added value to the application of CWPO in drinking water production. 20

21