Water Electrolytes and AcidBase Balance 1 Body Fluids

Water, Electrolytes, and Acid-Base Balance 1

Body Fluids • Intracellular – All fluids inside cells of body – About 40% of total body weight • Extracellular – All fluids outside cells – About 20% of total body weight – Subcompartments • Interstitial fluid and plasma; lymph, CSF, synovial fluid 2
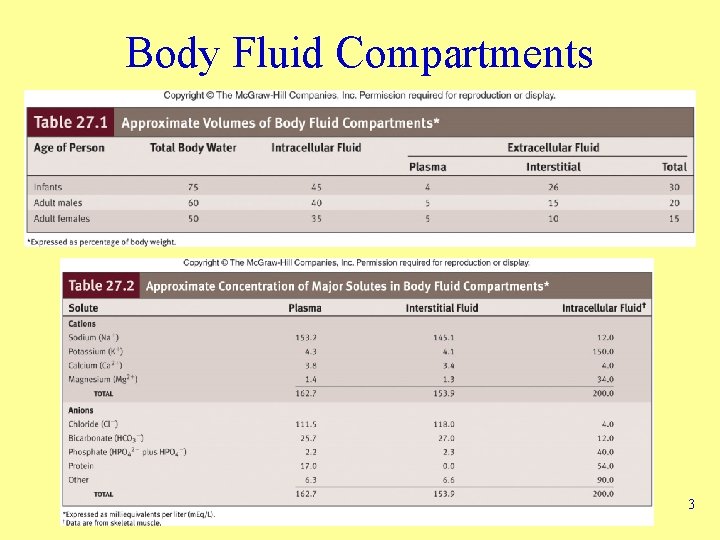
Body Fluid Compartments 3

Water Content Regulation • Content regulated so total volume of water in body remains constant • Kidneys primary regulator of water excretion • Regulation processes – – Osmosis Osmolality Baroreceptors Learned behavior • Sources of water – Ingestion – Cellular metabolism • Routes of water loss – Urine – Evaporation • Perspiration • Respiratory passages – Feces 4

Extracellular Fluid Osmolality • Osmolality – Adding or removing water from a solution changes this • Decreased osmolality – Inhibits thirst and ADH secretion • Increased osmolality – Triggers thirst and ADH secretion 5
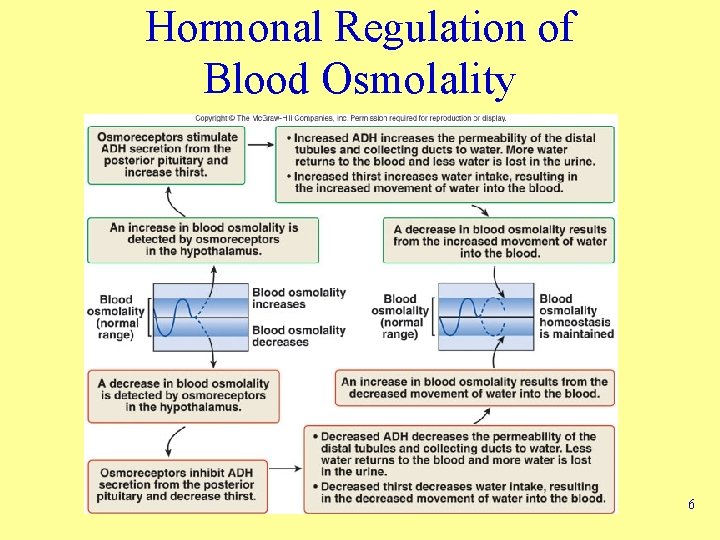
Hormonal Regulation of Blood Osmolality 6

Regulation of ECF Volume • Mechanisms – Neural – Renin-angiotensinaldosterone – Atrial natriuretic hormone (ANH) – Antidiuretic hormone (ADH) • Increased ECF results in – – Decreased aldosterone secretion Increased ANH secretion Decreased ADH secretion Decreased sympathetic stimulation • Decreased ECF results in – – Increased aldosterone secretion Decreased ANH secretion Increased ADH secretion Increased sympathetic stimulation 7

Hormonal Regulation of Blood Volume 8
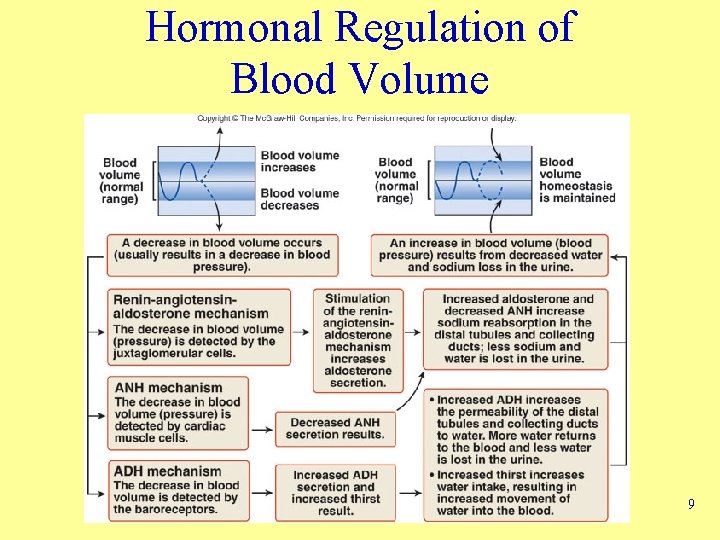
Hormonal Regulation of Blood Volume 9
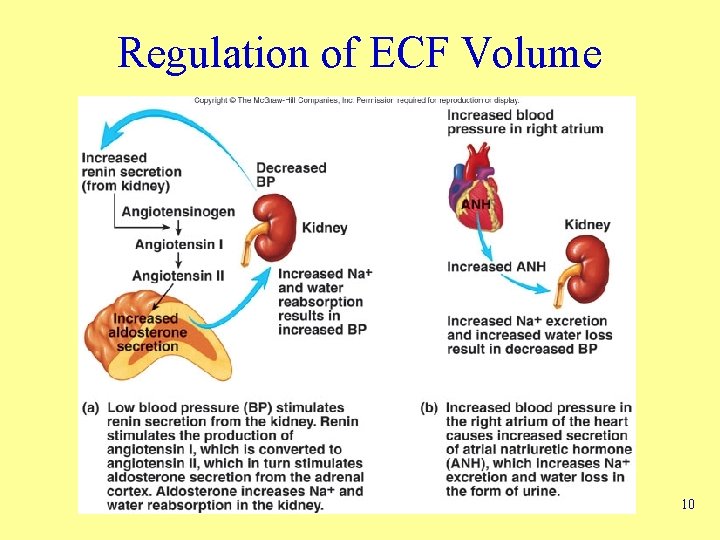
Regulation of ECF Volume 10
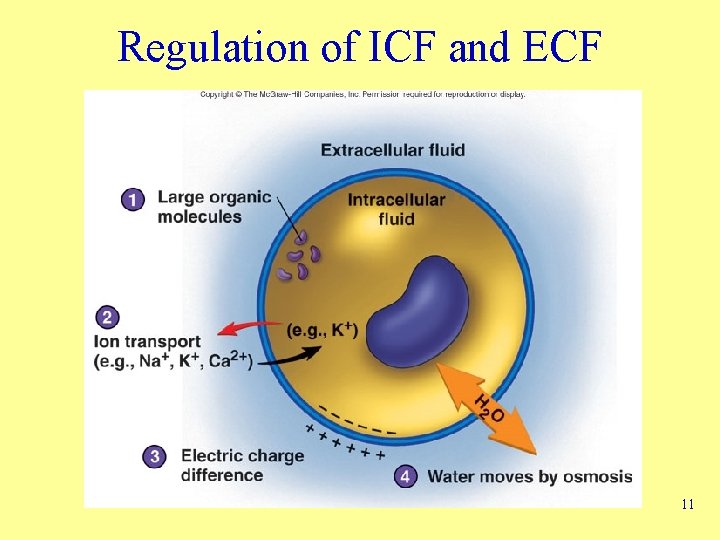
Regulation of ICF and ECF 11

Regulation of Electrolytes in ECF • Electrolytes – Molecules or ions with an electrical charge • Water ingestion adds electrolytes to body • Kidneys, liver, skin, lungs remove from body – Concentration changes only when growing, gaining or losing weight • Na+ Ions – Dominant ECF cations – Responsible for 90 -95% of osmotic pressure • Regulation of Na+ ions – Kidneys major route of excretion – Small quantities lost in sweat • Terms – Hypernatremia – Hyponatremia 12

Mechanisms Regulating Blood Sodium 13

Mechanisms Regulating Blood Sodium 14

Abnormal Plasma Levels of Sodium Ions 15

Regulation of Chloride, Potassium, Magnesium Ions • Chloride ions – Predominant anions in ECF • Magnesium ions – Capacity of kidney to reabsorb is limited – Excess lost in urine – Decreased extracellular magnesium results in greater degree of reabsorption • Potassium ions – Maintained in narrow range – Affect resting membrane potentials – Aldosterone increases amount secreted • Terms – Hyperkalemia – Hypokalemia 16
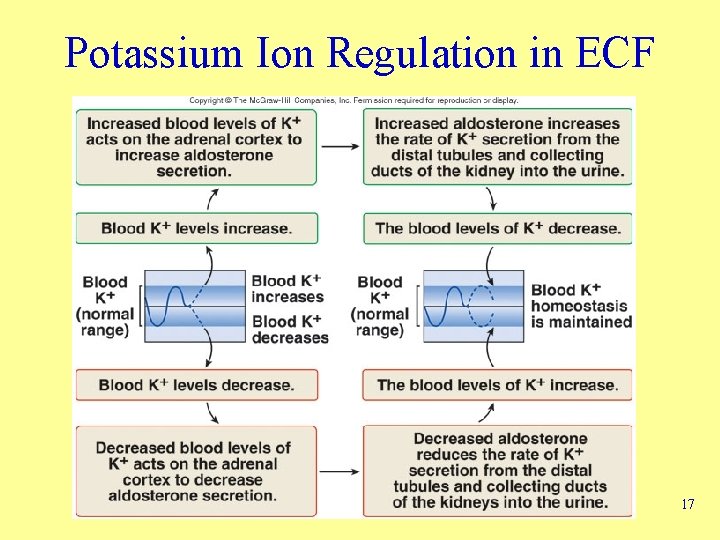
Potassium Ion Regulation in ECF 17

Abnormal Concentration of Potassium Ions 18

Abnormal Plasma Levels of Magnesium Ions 19
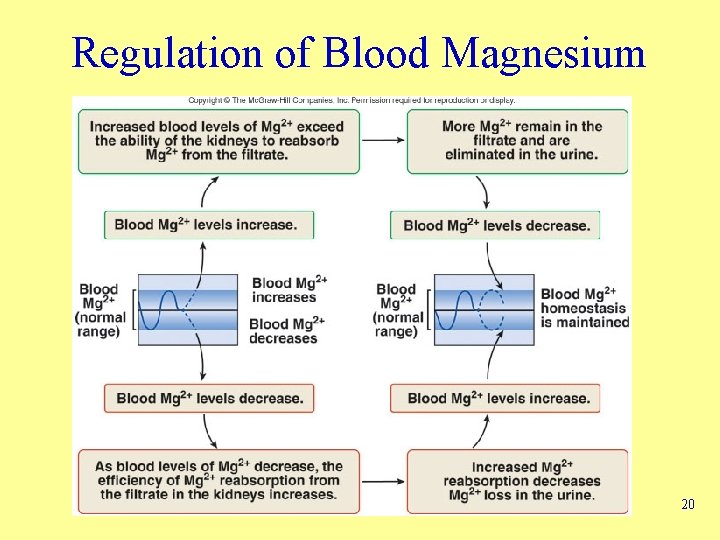
Regulation of Blood Magnesium 20

Regulation of Calcium Ions • Regulated within narrow range – Elevated extracellular levels prevent membrane depolarization – Decreased levels lead to spontaneous action potential generation • Terms – Hypocalcemia – Hypercalcemia • PTH increases Ca 2+ extracellular levels and decreases extracellular phosphate levels • Vitamin D stimulates Ca 2+ uptake in intestines • Calcitonin decreases extracellular Ca 2+ levels 21

Regulation of Calcium Ions 22

Regulation of Phosphate Ions • Under normal conditions, reabsorption of phosphate occurs at maximum rate in the nephron • An increase in plasma phosphate increases amount of phosphate in nephron beyond that which can be reabsorbed; excess is lost in urine 23
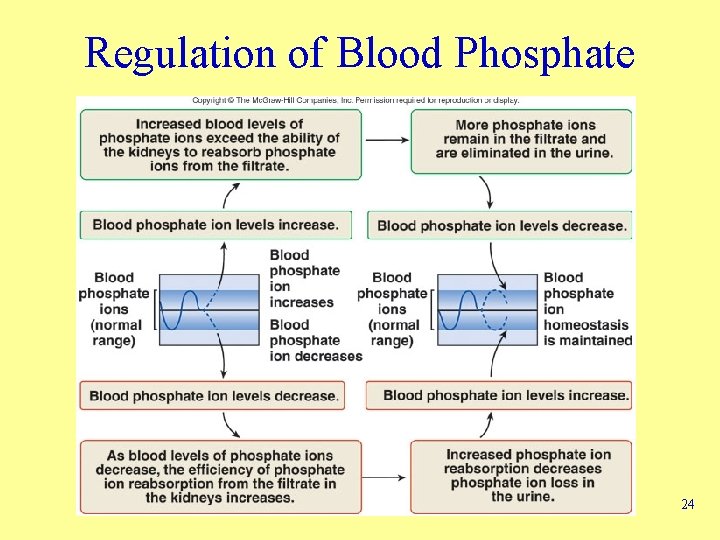
Regulation of Blood Phosphate 24

Acids and Bases and Buffers • Acids – Release H+ into solution • Bases – Remove H+ from solution • Acids and bases – Grouped as strong or weak • Buffers: Resist changes in p. H – When H+ added, buffer removes – When H+ removed, buffer replaces • Types of buffer systems – Carbonic acid/bicarbonate – Protein – Phosphate 25

Regulation of Acid-Base Balance 26
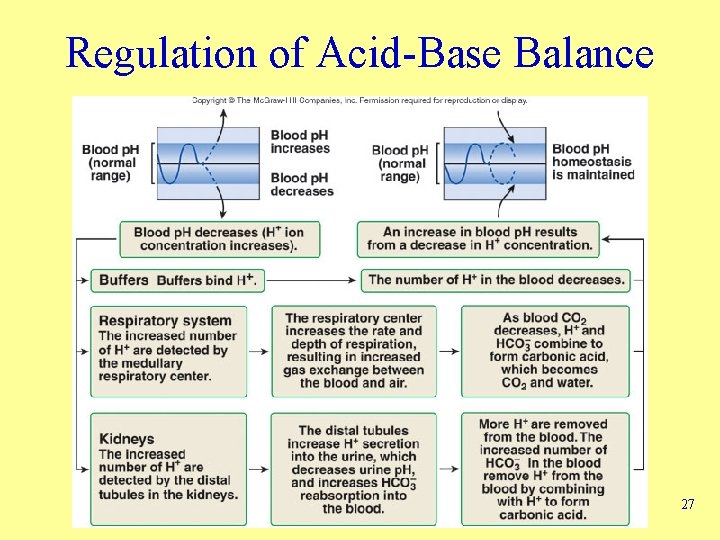
Regulation of Acid-Base Balance 27

Buffer Systems 28

Respiratory Regulation of Acid-Base Balance • Respiratory regulation of p. H is achieved through carbonic acid/bicarbonate buffer system – As carbon dioxide levels increase, p. H decreases – As carbon dioxide levels decrease, p. H increases – Carbon dioxide levels and p. H affect respiratory centers • Hypoventilation increases blood carbon dioxide levels • Hyperventilation decreases blood carbon dioxide levels 29
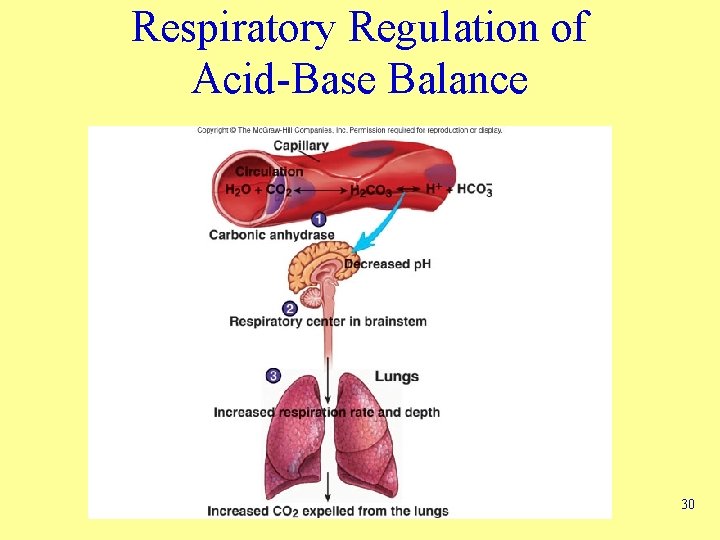
Respiratory Regulation of Acid-Base Balance 30

Renal Regulation of Acid-Base Balance • Secretion of H+ into filtrate and reabsorption of HCO 3 - into ECF cause extracellular p. H to increase • HCO 3 - in filtrate reabsorbed • Rate of H+ secretion increases as body fluid p. H decreases or as aldosterone levels increase • Secretion of H+ inhibited when urine p. H falls below 4. 5 31
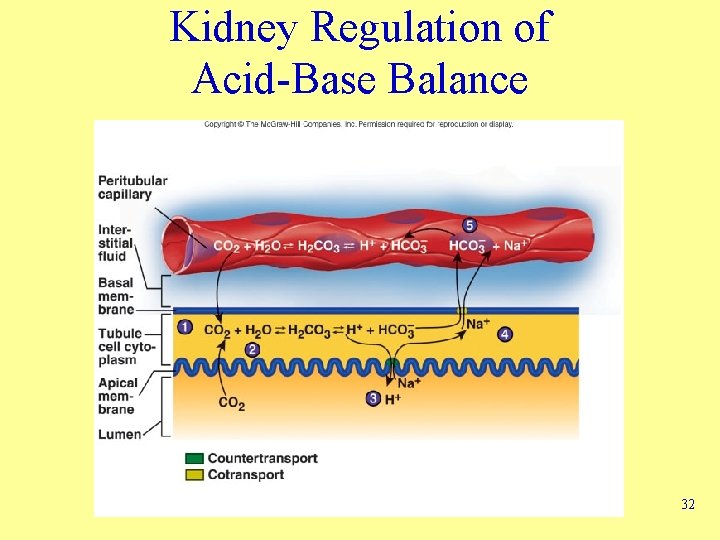
Kidney Regulation of Acid-Base Balance 32
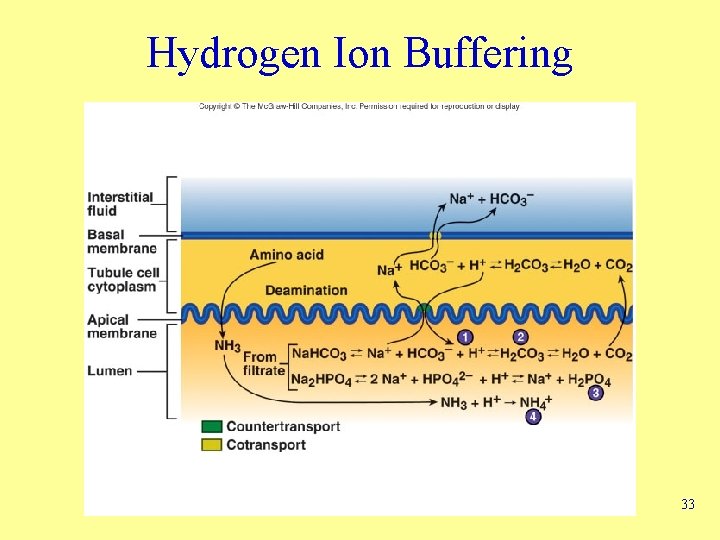
Hydrogen Ion Buffering 33

Acidosis and Alkalosis • Acidosis: p. H body fluids below 7. 35 – Respiratory: Caused by inadequate ventilation – Metabolic: Results from all conditions other than respiratory that decrease p. H • Alkalosis: p. H body fluids above 7. 45 – Respiratory: Caused by hyperventilation – Metabolic: Results from all conditions other than respiratory that increase p. H • Compensatory mechanisms 34

Acidosis and Alkalosis 35
- Slides: 35