Water Electrolyte and Acid Base Balance Chapter 21

Water, Electrolyte and Acid. Base Balance Chapter 21

• Balance – a state of equilibrium – substances are maintained in the right amounts and in the right place in the body

Water Balance • Osmosis is the primary method of water movement into and out of body fluid compartments. • Osmosis is the net movement of water molecules through a selectively permeable membrane from an area of high water concentration to an area of lower water concentration.

• The concentration of solutes determines the direction of water movement. • Most solutes in the body are electrolytes – inorganic compounds which dissociate into ions in solution. • “Where sodium goes, water follows. ”

• About 40 Liters (10. 56 gallons) of body water • Babies – 75% water • Men – 63 % • Women – 52%
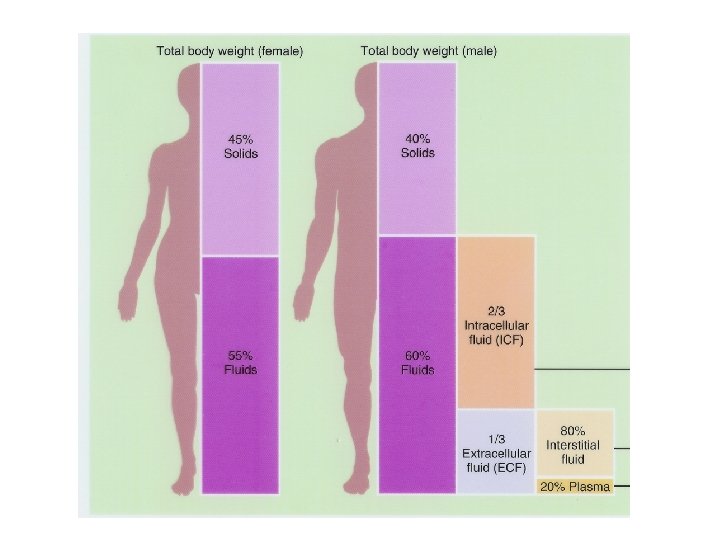

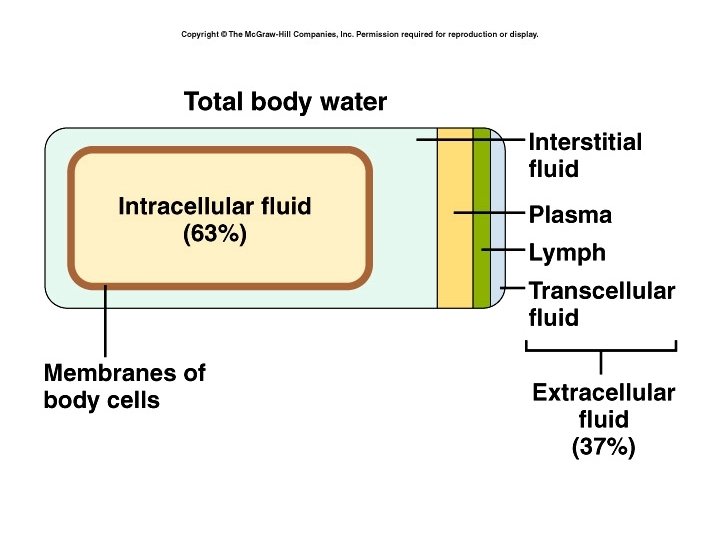
Fluid compartments • Separated by selectively permeable membranes • Intracellular – 2/3 (63%) of total body water • Extracellular – 1/3 (37%) – Interstitial fluid – 80 % of extracellular water – Blood plasma – 20 % of extracellular water

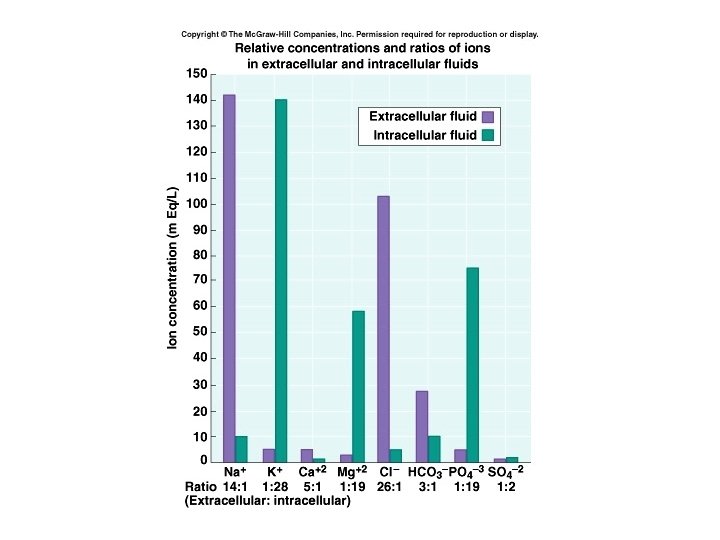
Composition of compartments • Extracellular fluids: – High in Na+, Cl-, Ca++, HCO 3 • Blood plasma has more protein than interstitial fluid and lymph • Intracellular fluids: – High in K+, phosphate, Mg++, and more protein than plasma

Movement of water • Hydrostatic pressure – pressure of fluids • Osmotic pressure – solute concentration (often Na+) – In blood referred to as colloid osmotic pressure (COP)

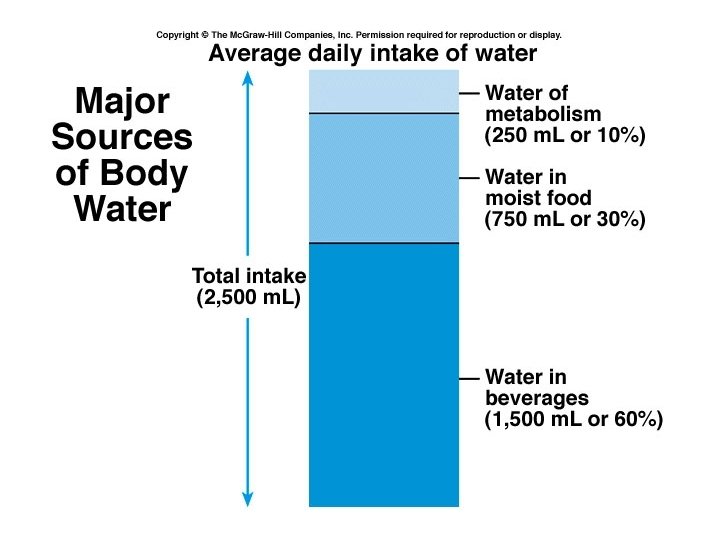
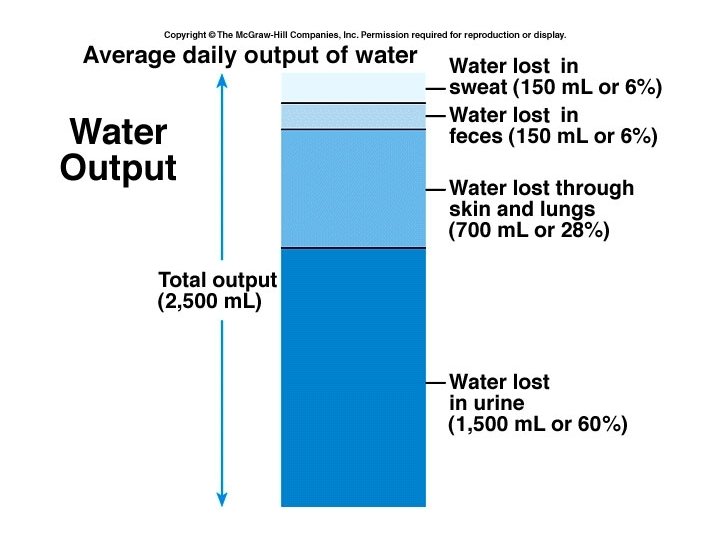
Water intake = Water loss • Average adult takes in about 2, 500 ml/day • Sources of water: – Preformed water: 2, 300 ml • Drinking water: 1, 500 ml (60%) • Moist food : 750 ml (30%) – Water of metabolism: 250 ml (10%) • Cellular respiration • Dehydration synthesis

Regulation of water intake • Main regulator is thirst. • Dehydration (output>intake) as little as 1% decrease in body water causes: – Decreased production of saliva – Increased blood osmotic pressure – stimulates osmoreceptors in the hypothalamus – Decreased blood volume – renin is produced

• The thirst center in hypothalamus is stimulated ( or mistakenly, the hunger center) and person feels thirsty • Wetting of the mouth and stretching of stomach or intestines decrease thirst before we take in too much water. • Water is absorbed, and blood osmotic pressure decreases.

Sources of water loss • Through kidneys in urine – 1500 ml (60%) • Through intestines 150 ml (6%) – Can be significant in vomiting and diarhhea • From skin (sweat) 150 ml (6%) • From lungs and skin 700 ml (28%) • Last is called insensible loss – (menstruation)

Regulation of Water Output • Through regulating urine formation • ADH – production stimulated by ↑ blood tonicity of decrease in volume. – Acts on distal convoluted tubules and collecting ducts of kidney – permits reabsorption of water

• Aldosterone – production is stimulated by angiotensin II through renin production – Causes sodium ( and water) to be reabsorbed • ANP – causes sodium (and water) loss when pressure in right atrium is too high

Water imbalances • Dehydration is the imbalance seen most often. – Prolonged diarrhea or vomiting – Excessive sweating
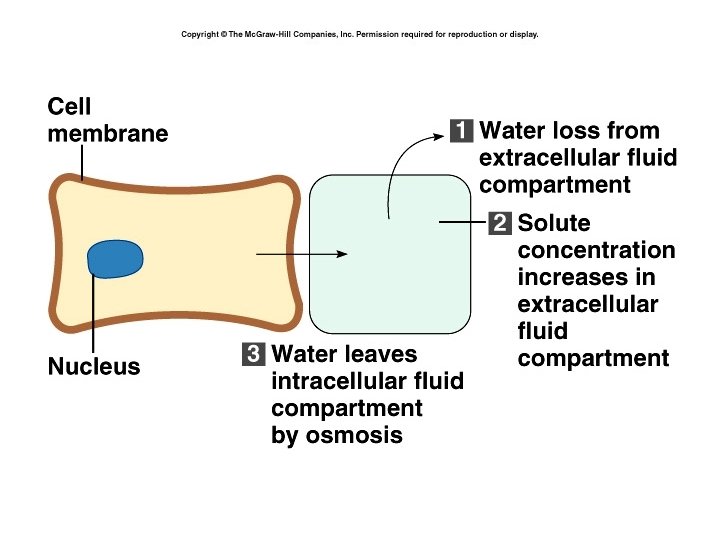

Water toxicity • If lose water by sweating, we also lose sodium. • Rapidly drinking large quantities of water decreases plasma sodium concentration initially, then see decrease in ISF as well. • Water is drawn into cells • This increases ISF tonicity, and water is drawn from blood • Add salt when replacing fluids like this!
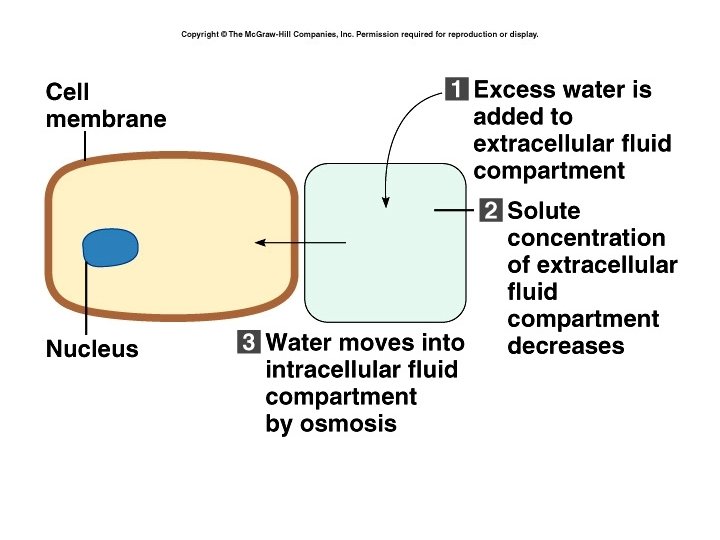

Overhydration • Can occur if I. V. fluids are given too rapidly or in too large amounts. • Extra fluid puts strain on heart

• Water that moves back into capillaries depends on concentration of plasma proteins. • Decrease in blood proteins caused by: – Dietary deficiency in proteins – Liver failure – Blockage of lymphatic system – Increased capillary permeability • Burns, infection
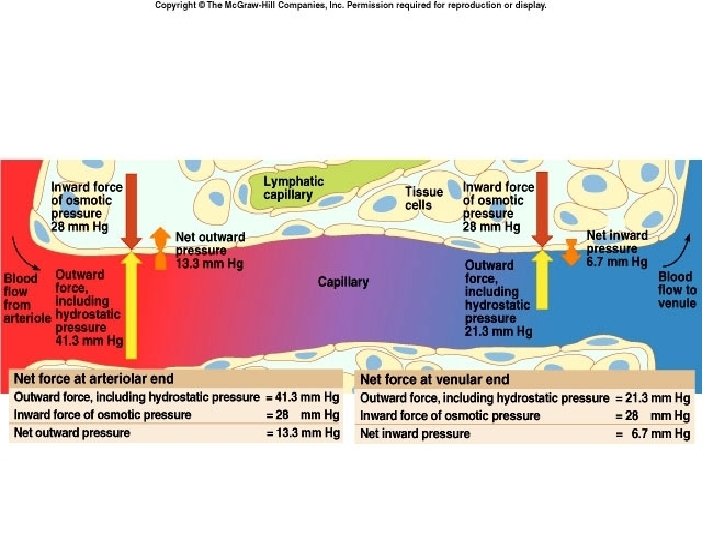

• Fluid moves from the blood to the interstitial fluid. • Get large amounts of fluid in the intercellular spaces – Edema
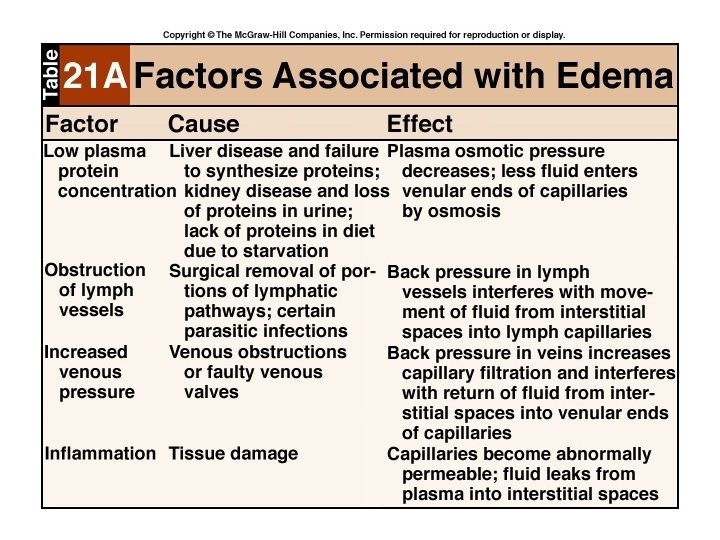

• Of the three main compartments (IVF, ICF and ISF) the interstitial fluid varies the most.

Edema • Can be caused by: – Decrease in plasma proteins – Retention of electrolytes, esp. Na+ – Increase in capillary blood pressure

Electrolyte Balance • Cations – positively charged ions • Anions – negatively charged ions • Body fluids also contain charged organic molecules • Only a small percentage of molecules in fluids are non-electrolytes: glucose, urea, creatinine

Functions of electrolytes • Certain ions control the osmosis of water between body compartments • Ions help maintain the acid-base balance necessary for cellular activity • Ions carry electric current, which allows for action potentials and secretion of neurotransmitters • Several ions are cofactors needed for the optimal activity of enzymes

Electrolyte intake • Food and water • Produced by metabolism • Salt craving

Electrolyte loss • Sweat • Feces • Urine

Osmolarity • The total concentration of dissolved particles determines osmolarity. • Glucose – one dissolved particle • Na. Cl – dissolves into two particles • One mole of Na. Cl = 2 osmoles • Osmoles/L = osmolarity of solution

Sodium (Na+) • 90 % of extracellular cations and half the osmolarity of extracellular solutions • Necessary for action potentials in nerve & muscle cells • Aldosterone increases reabsorption from DCT and collecting ducts – ↓ blood volume, ↓ extracellular Na+ , ↑ extracellular K+ • ANP causes loss of Na+

Potassium (K+) • Most numerous intracellular cation • Membrane potential and repolarization • Controlled by aldosterone – causes loss of K+ in urine

Calcium (Ca++) • Part of bone, most abundant mineral in body. 98% of Ca is in bone • Extracellular cation • Needed for blood clotting, nerve and muscle function • PTH causes reabsorption of bone and increases reabsorption from G. I tract and glomerular filtrate • Calcitonin inhibits osteoclasts and stimulates osteoblast, so calcium is removed from blood

Chloride (Cl-) • Most common extracellular anions • Cl- diffuses easily between compartments – can help balance charges (RBC’s) • Parietal cells in stomach secrete Cl- & H+ • Aldosterone indirectly adjusts Cl- when it increases the reabsorption of Na+ - Clfollows the Na+

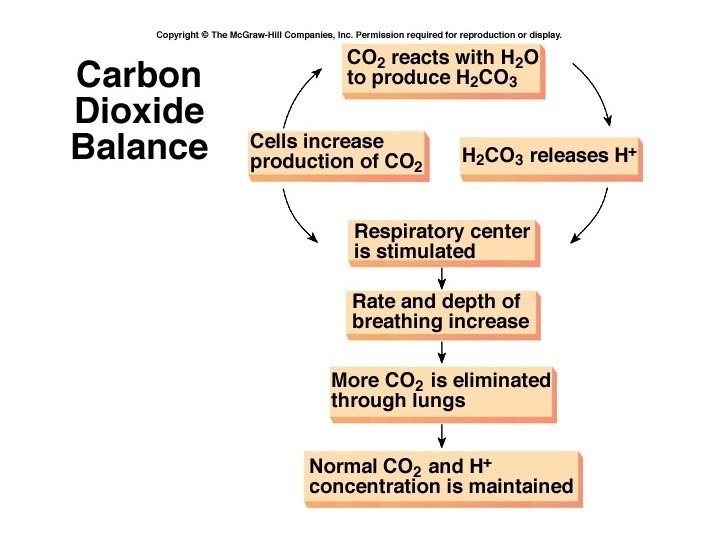
Bicarbonate (HCO 3 -) • Part of the body’s chief buffer and transports CO 2 in blood stream. • CO 2 + H 2 O ↔H 2 CO 3 ↔ H+ + HCO 3 • The kidneys are the main regulators of bicarbonate: they form bicarb when levels are low and excrete it when levels are high.

Phosphate (HPO 42 -) • Like calcium, most of the phosphate is found in the bones. • 15% is ionized • Found in combination with lipids, proteins, carbohydrates, nucleic acids and ATP. • Three different forms • Part of the phosphate buffer system • PTH causes phosphate to be released from bones and to be excreted by the kidneys. Calcitonin removes phosphate by encouraging bone formation.

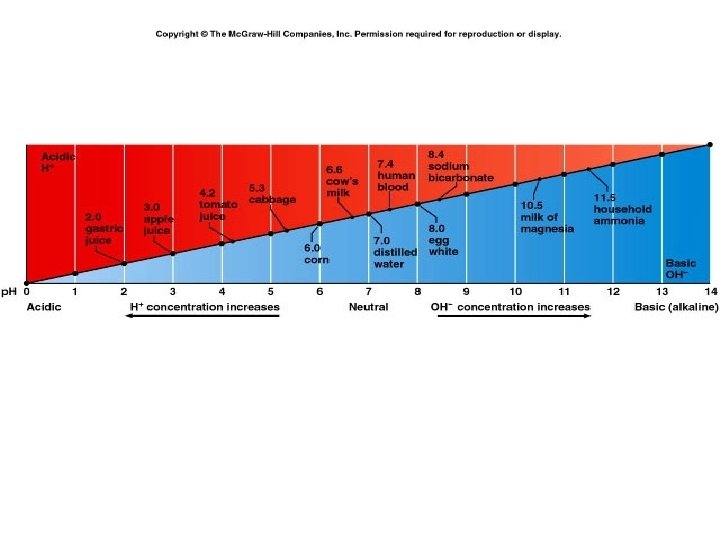
Acid-Base Balance • • p. H – negative log of H+ concentration Affects functioning of proteins (enzymes) Can affect concentrations of other ions Modify hormone actions (proteins)

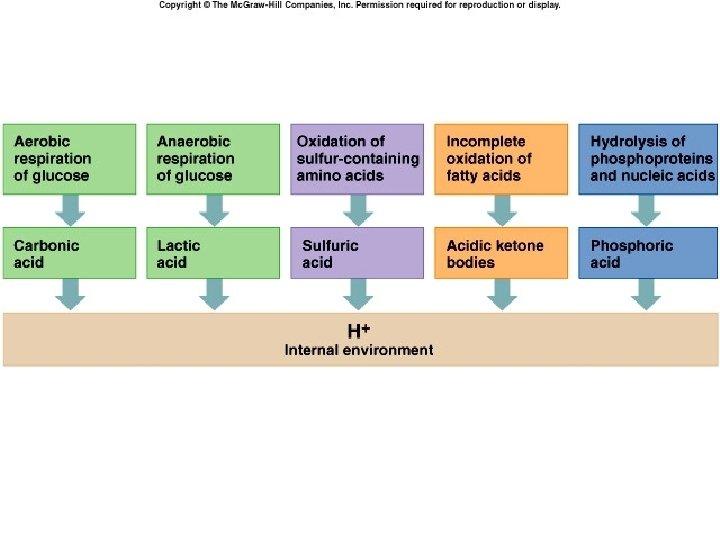
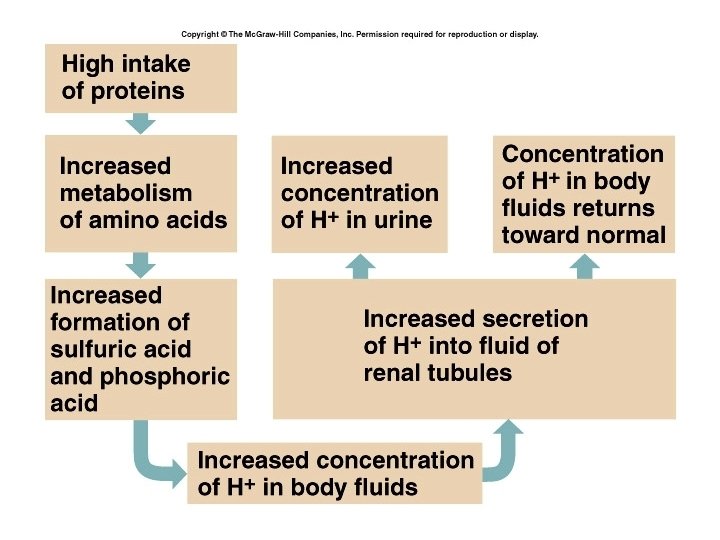
Acid intake • Foods • Produced by cellular metabolism

Strengths of Acids and Bases • Acids and bases that ionize (break apart) completely are strong acids and bases. (HCl; Na. OH) • Acids and bases that do not completely dissociate in solution are weak acids and bases. (lactic acid, carbonic acid)

• Remember, blood needs to stay between 7. 35 and 7. 45 for the body to function properly. • Since more acids than bases are formed, p. H balance is mainly a matter of controlling excess H+.

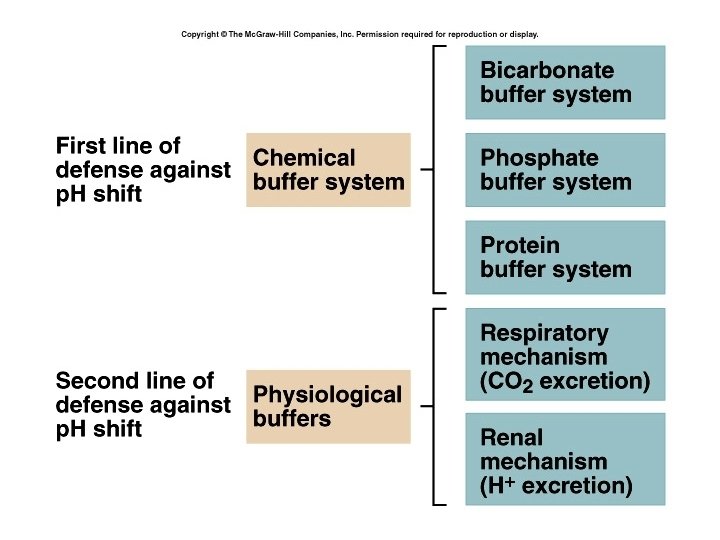
Control of Acid-Base Balance 1. Buffer systems 2. Exhalation of carbon dioxide 3. Kidney excretion

Buffers • Are pairs of chemical substances that prevent a sharp change in the p. H of a solution. • Buffers exchange strong acids for weaker acids that do not release as much H+ and thus change the p. H less.

Bicarbonate Buffer System • Na. HCO 3 + sodium bicarbonate H 2 CO 3 carbonic acid Addition of a strong acid: HCl + Na. HCO 3 → H 2 CO 3 + Na. Cl Carbonic acid does not dissociate completely, and p. H is changed much less.

• Addition of a strong base: • Na. OH + H 2 CO 3 → Na. HCO 3 + H 2 O • Water dissociates very little, and p. H remains nearly the same.

• Usually the body is called upon to buffer weaker organic acids, such as lactic acid. • Carbonic acid is formed, and amount of bicarbonate ion decreases. • Blood needs to maintain a 20: 1 ratio of bicarbonate ion : carbonic acid. • H+ concentration increases slightly • p. H drops slightly
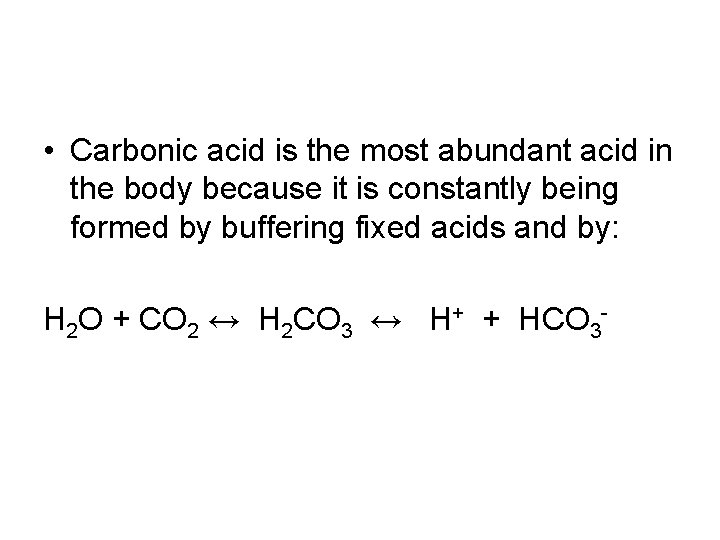
• Carbonic acid is the most abundant acid in the body because it is constantly being formed by buffering fixed acids and by: H 2 O + CO 2 ↔ H 2 CO 3 ↔ H+ + HCO 3 -

Phosphate Buffer System • Is present in extracellular and intracellular fluids, most important in intracellular fluids and renal tubules. • H+ + HPO 42 - → H 2 PO 4 monohydrogen phosphate dihydrogen phosphate • OH- + H 2 PO 4 - → H 2 O + HPO 42 -

Protein Buffer System • The most abundant in body cells and plasma. • Carboxyl group -COOH ↔ -COO- + H+ • Amino group –NH 2 ↔ -NH 3+

Respiratory Mechanisms – Exhalation of CO 2 • Because carbonic acid can be eliminated by breathing out CO 2 it is called a volatile acid. • Body p. H can be adjusted this way in about 1 -3 minutes • p. H also affects breathing rate • Powerful eliminator of acid, but can only deal with carbonic acid.

Kidney excretion of + H • Metabolic reactions produce large amounts of fixed acids. • Kidneys can eliminate larger amounts of acids than the lungs • Can also excrete bases • Can excrete acids while conserving bicarbonate ion • Can produce more bicarbonate ion • Kidneys are the most effective regulators of p. H; if kidneys fail, p. H balance fails

The regulators work at different rates • Buffers are the first line of defense because they work almost instantaneously. • Secondary defenses take longer to work: – Respiratory mechanisms take several minutes to hours – Renal mechanisms may take several days

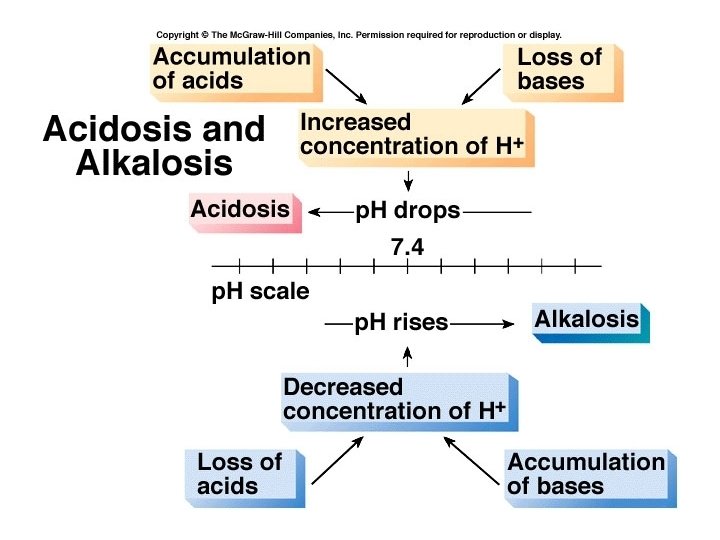
p. H imbalances • The normal blood p. H range is 7. 35 – 7. 45 • Any p. H below this range is considered to be a condition of acidosis • Any p. H above this range is considered to be a condition of alkalosis • The body response to acid-base imbalance is called compensation: Compensation may be complete if the blood p. H is brought back to normal, or partial if it is still outside the norms.

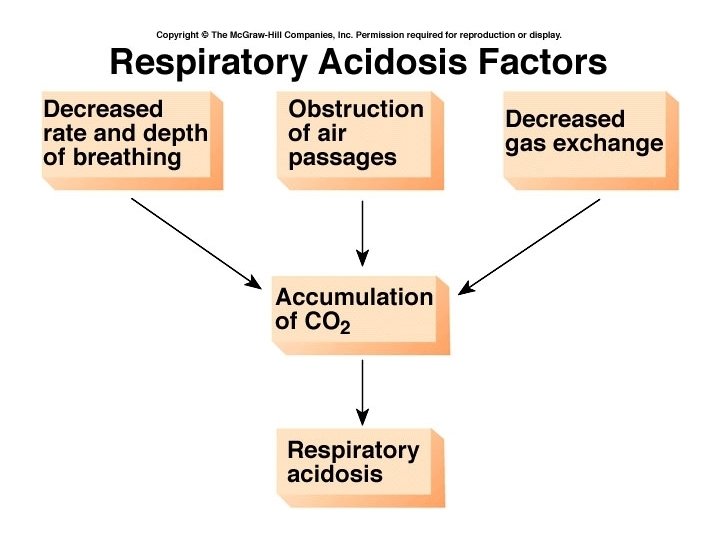
Respiratory problems • Respiratory acidosis is a carbonic acid excess (blood CO 2 is too high) • Respiratory alkalosis is a carbonic acid deficit (blood CO 2 is too low) • Compensation would occur through the kidneys
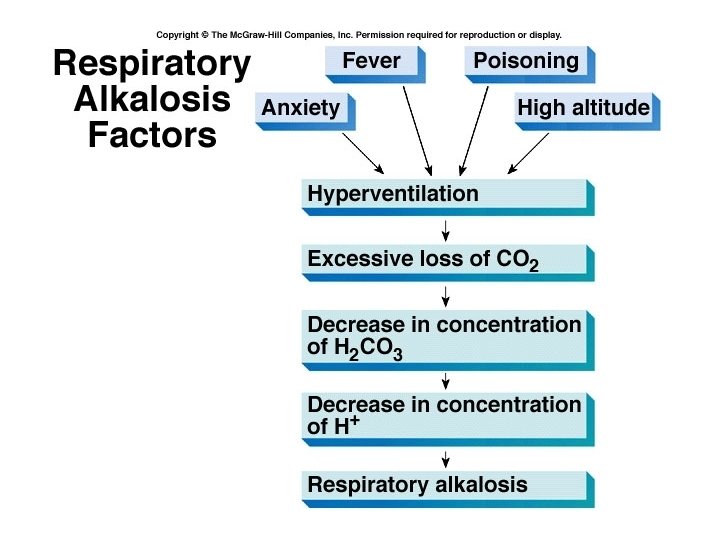

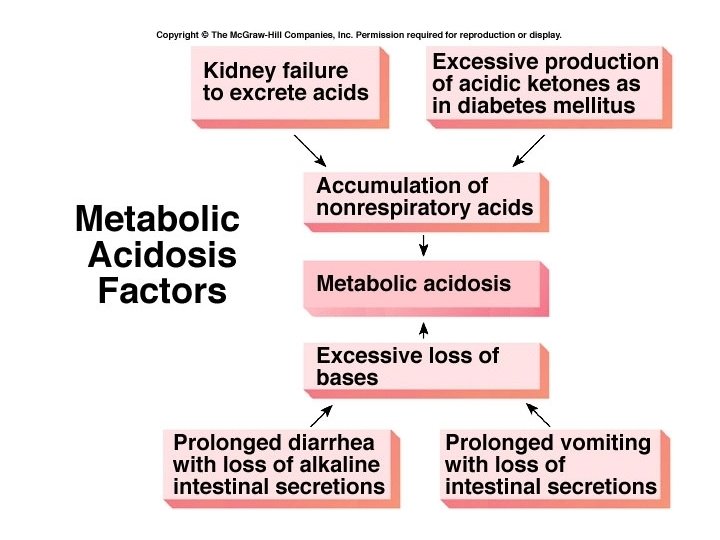
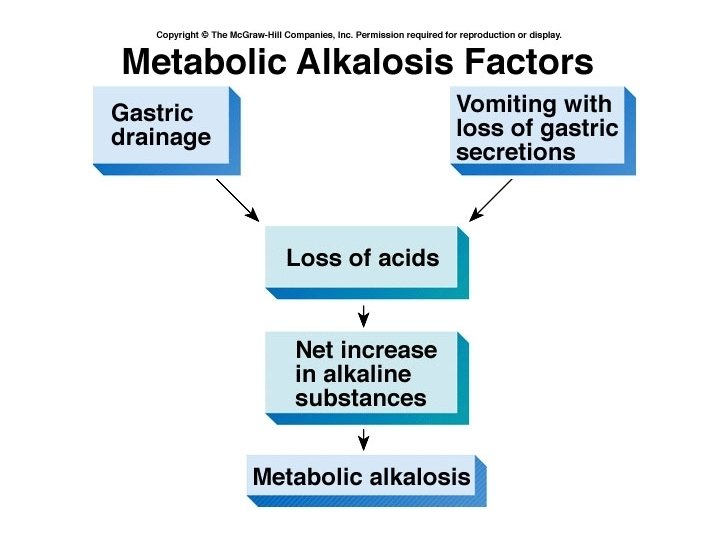
Metabolic problems • Metabolic acidosis is a bicarbonate deficit • Metabolic alkalosis is a bicarbonate excess • Compensation would occur through changes in the depth and rate of respiration.
- Slides: 72