Water Covalent Bonds Each water molecule consists of
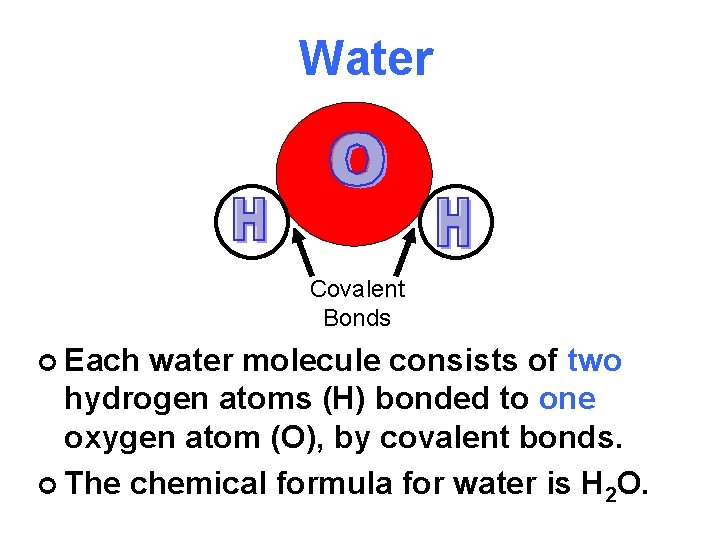
Water Covalent Bonds ¢ Each water molecule consists of two hydrogen atoms (H) bonded to one oxygen atom (O), by covalent bonds. ¢ The chemical formula for water is H 2 O.
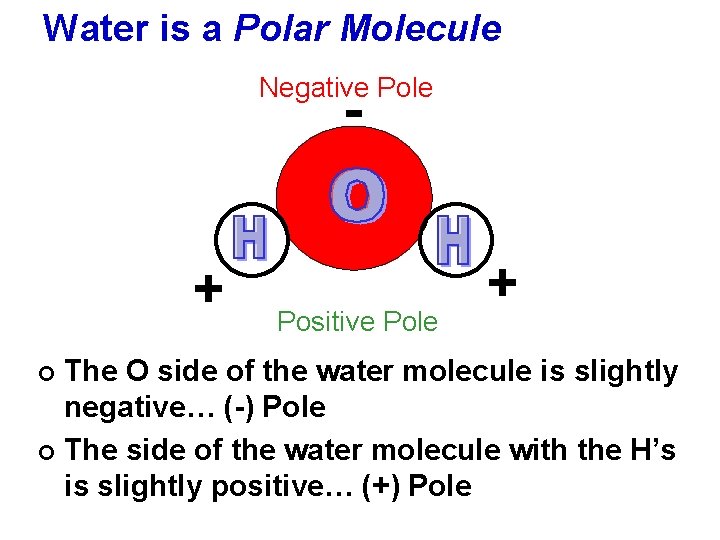
Water is a Polar Molecule Negative Pole - + Positive Pole + The O side of the water molecule is slightly negative… (-) Pole ¢ The side of the water molecule with the H’s is slightly positive… (+) Pole ¢

One water molecule is attracted to other water molecules by hydrogen bonds. + + - Hydrogen Bonds + - + + + +

Since water is polar and can form hydrogen bonds: 1. Water molecules are attracted to other water molecules by ¢ 2. COHESION – the attraction between molecules of the same substance Water molecules are attracted to other polar molecules by ¢ ADHESION – the attraction between molecules of different substances

Water has unique properties which make life on Earth possible.

1. Exhibits Surface Tension This thin film allows insects like the water strider to walk on water. Surface tension is created when water molecules stick to each other on the surface of water.

On the back of your sheet: ¢ Draw a picture representing surface tension.

On the front of your sheet: ¢ State which demonstration illustrated surface tension. ¢ State whether it was due to cohesion, adhesion, or both.
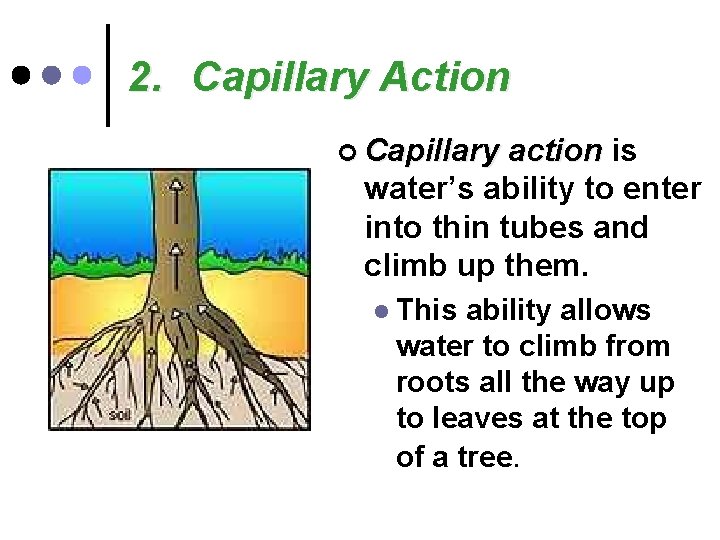
2. Capillary Action ¢ Capillary action is water’s ability to enter into thin tubes and climb up them. l This ability allows water to climb from roots all the way up to leaves at the top of a tree.

On the back of your sheet: ¢ Draw a picture representing capillary action.

On the front of your sheet: ¢ State which demonstration illustrated capillary action. ¢ State whether it was due to cohesion, adhesion, or both.

3. High specific heat ¢ Water resists temperature changes l This means it can absorb a lot of energy without getting hot, and can lose a lot of energy without getting cold.
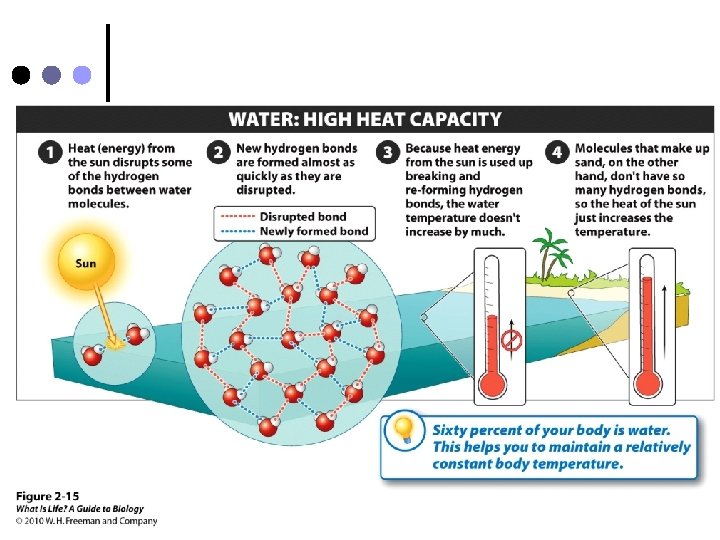

¢ Why is resistance to temperature change important? l l Remember – not only is about 2/3 of a human’s weight is water, but about 3/4 of Earth is covered with water Resistance to temperature change allows organisms to maintain constant body temperature, AND keeps the Earth at a fairly constant temperature.

On the back of your sheet: ¢ Draw a picture representing resistance to temperature change.

On the front of your sheet: ¢ State which demonstration illustrated resistance to temperature change. ¢ State whether it was cohesion, adhesion, or both.

4. High Heat of Vaporization ¢ ¢ High heat of vaporization means water evaporates slowly. It takes a lot of heat energy to change liquid water to water vapor. l When we sweat, heat from our body is used to evaporate the sweat, so we cool down.

On the back of your sheet: ¢ Draw a picture representing high heat of vaporization.

On the front of your sheet: ¢ State which demonstration illustrated high heat of vaporization. ¢ State whether high heat of vaporization was due to cohesion, adhesion, or both.

5. Expands as it freezes • Water expands as it freezes. This happens because as it reaches freezing temperature, the molecules push apart and lock into a lattice-like structure. • Ice is less dense than water.

Importance of Expansion upon Freezing a. Ice Floats! l Life at the bottom of lakes does not get crushed l Ice also serves as an insulator b. Ice breaks rocks into soil

On the back of your sheet: ¢ Draw a picture representing water’s expansion as it freezes.

On the front of your sheet: ¢ State which demonstration illustrated expansion of water as it freezes. ¢ State whether it was cohesion, adhesion, or both.

6. Is the Universal Solvent ¢ ¢ Water is called the universal solvent because it can dissolve many substances, like nutrients and wastes. This allows nutrients and wastes to be easily transported to and from aquatic organisms, and within land-based organisms.
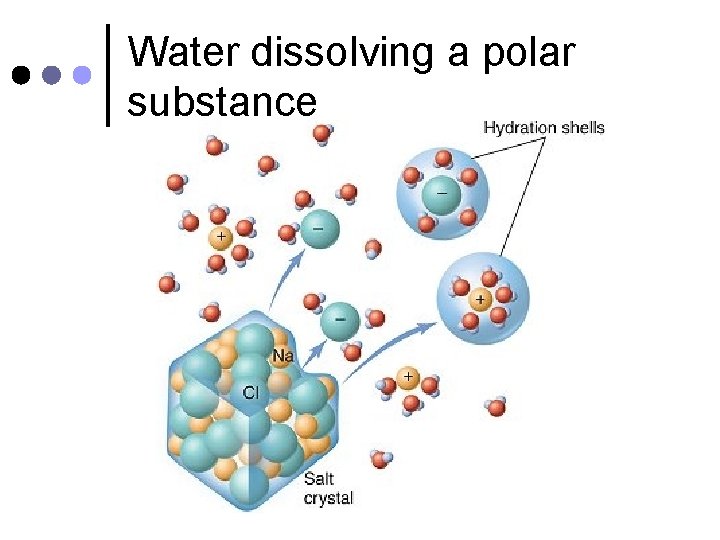
Water dissolving a polar substance

On the back of your sheet: ¢ Draw a picture representing water’s solvent capabilities.

On the front of your sheet: ¢ State which demonstration illustrated water’s solvent property. ¢ State whether it was cohesion, adhesion, or both.
- Slides: 27