Water Bonding Water has unusual properties Polar Covalent

Water Bonding Water has unusual properties
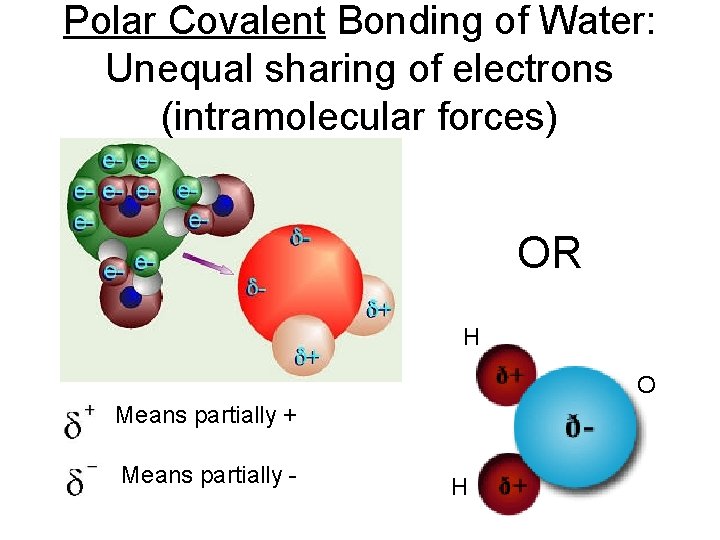
Polar Covalent Bonding of Water: Unequal sharing of electrons (intramolecular forces) OR H O Means partially + Means partially - H


Hydrogen Bonds hydrogen bond = is the attractive force between the hydrogen attached to an electronegative atom (tendency to attract electrons) of another molecule (usually O, N, F) Hydrogen Bonding Dance (1 min)
• Youtube Polarity and Hydrogen Bonding • (3: 52)
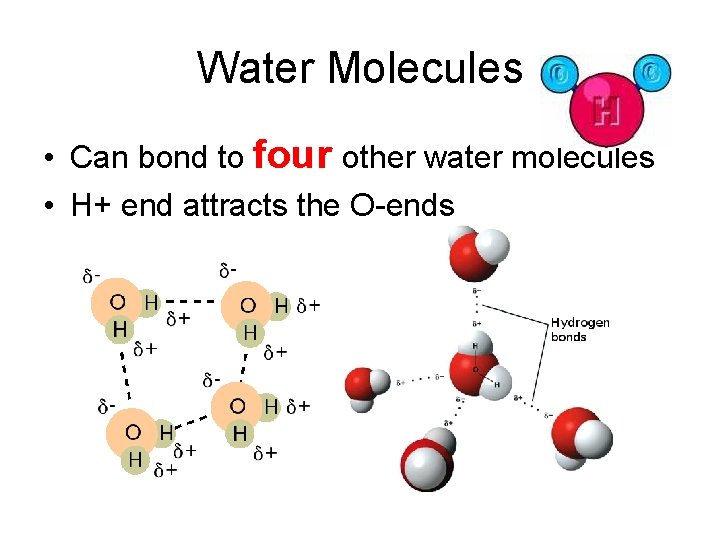
Water Molecules • Can bond to four other water molecules • H+ end attracts the O-ends
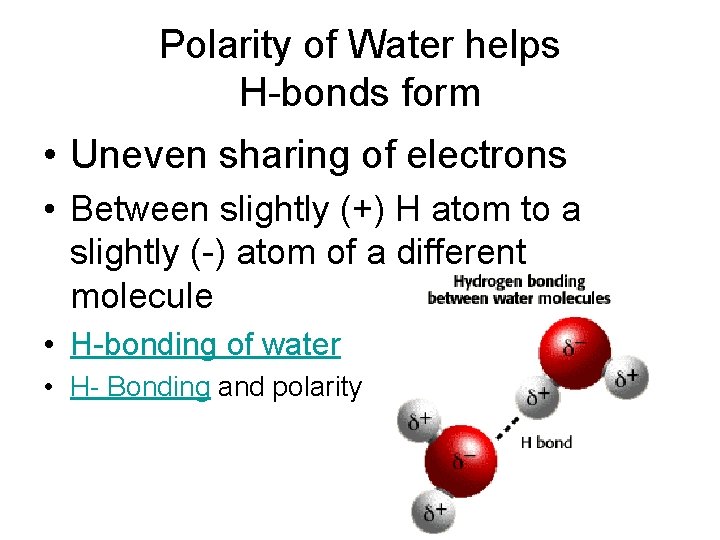
Polarity of Water helps H-bonds form • Uneven sharing of electrons • Between slightly (+) H atom to a slightly (-) atom of a different molecule • H-bonding of water • H- Bonding and polarity
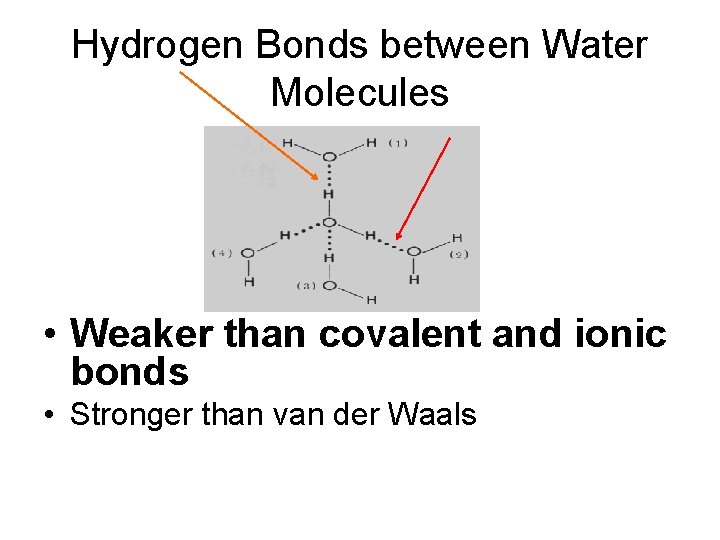
Hydrogen Bonds between Water Molecules • Weaker than covalent and ionic bonds • Stronger than van der Waals

Cohesion • Water is attracted to other water molecules (similar polar molecules) • H-bonding between water molecules hold them together Like in drops

Cohesion is why a waterfall, a stream, a water drop forms

Adhesion • Attraction of unlike substances • EX: water to the sides of a glass tube • Drop to leaf Strong adhesion to glass Not so much with plastic

Adhesion or Cohesion?

Surface Tension -the attraction of molecules to each other (cohesion) on a liquid's surface pull inward (due to H-bonds) to create a “film” A screen showing surface tension youtube Water molecules pull in all direction not on the surface

Surface tension is responsible for the shape of water droplets Surface Tension allows a Penny or Paper Clip to float ? Surface Tension and Capillary Action? ? - You. Tube H-bonds pull in water to create a “film”

Water Strider • Jesus Christ Lizard (natl geo) (1. 25 mins) • Can stand on top of water • Water Strider on Pond Surface (44 secs) See indentations of water strider’s feet in water.

High Specific Heat • = the amount of heat per unit mass required to raise the temperature by one degree Celsius. • The specific heat of water is higher than any other common substance. WHY? • Have to break/form those H-bonds.

Phases of Water • Describe how the molecules change (speed and spacing between molecules) as the temperature changes. • Water Phase Change Animation (keep clicking on the “increase heat”) • Did the water molecules break apart or did just the spacing between them change?
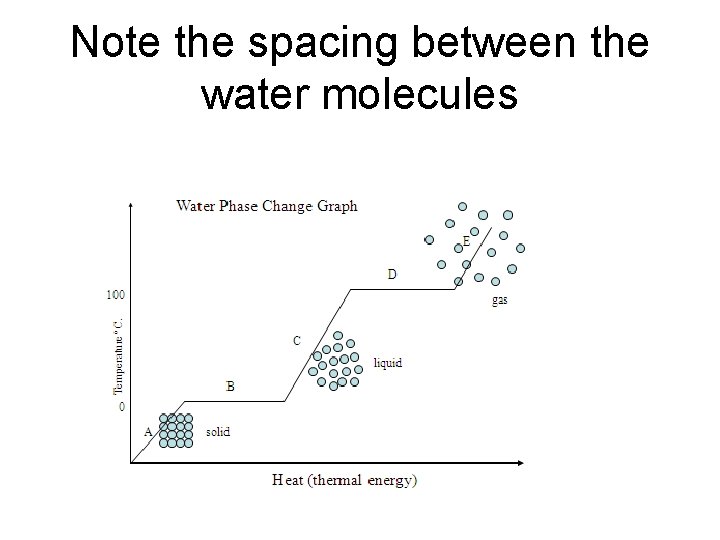
Note the spacing between the water molecules
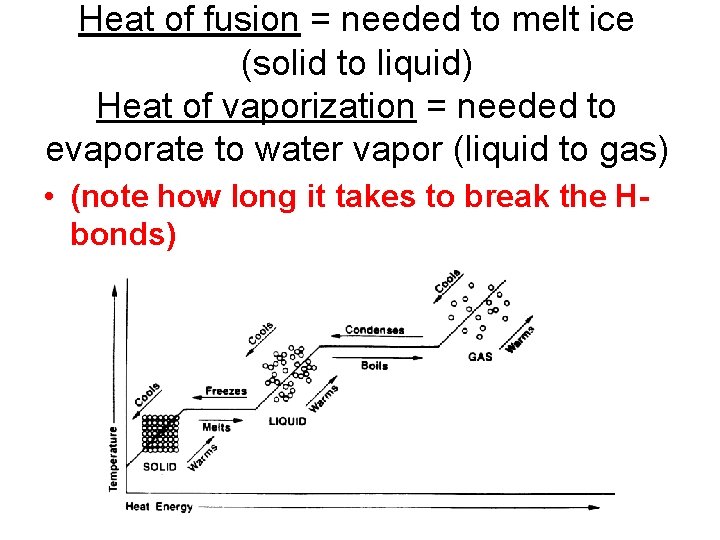
Heat of fusion = needed to melt ice (solid to liquid) Heat of vaporization = needed to evaporate to water vapor (liquid to gas) • (note how long it takes to break the Hbonds)
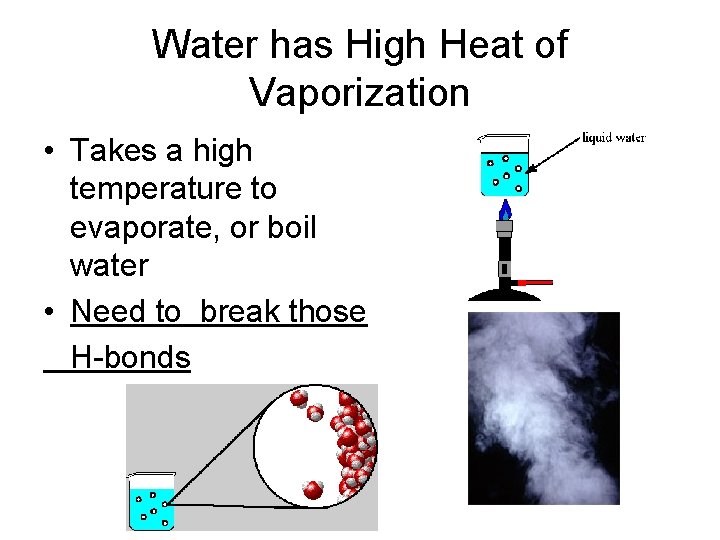
Water has High Heat of Vaporization • Takes a high temperature to evaporate, or boil water • Need to break those H-bonds
Water has High Heat of Fusion (Amount of heat to go from solid to liquid) • Takes a low temperature to freeze water (loses at lot of heat) • Those H-bonds must form

Look at the spacing and speed of molecules in each phase • Vision Learning (scroll down to Liquid Water and H-bonding and to Ice and H-bonding)

What’s the difference between… • Heat • Temperature • Total amount of energy of the atoms/molecules • A number of the average speed of molecules Which has more heat… 10 m. L of 50 o C water or 100 m. L of 50 o. C water?
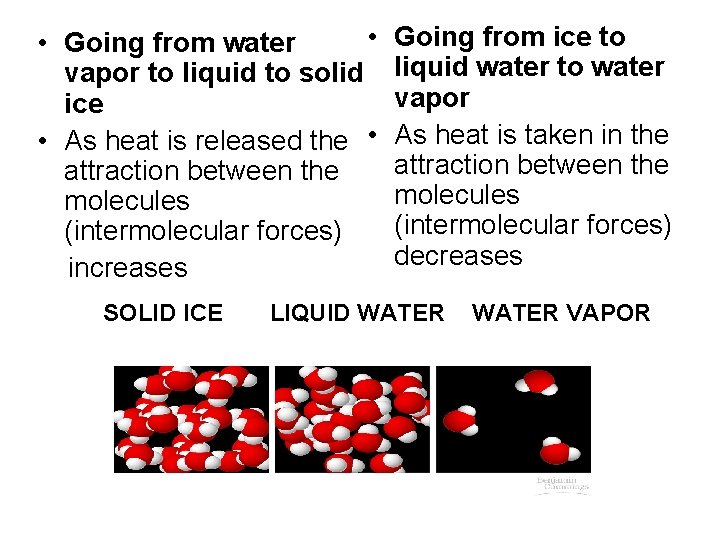
• • Going from water vapor to liquid to solid ice • As heat is released the • attraction between the molecules (intermolecular forces) increases SOLID ICE Going from ice to liquid water to water vapor As heat is taken in the attraction between the molecules (intermolecular forces) decreases LIQUID WATER VAPOR

Evaporative Cooling • The water of the surface of the skin absorbs heat to change water from liquid to gas. You feel cooler.

Water has High Specific Heat • Takes a lot of heat to raise the temperature of water 1 o. C(break the H -bonds) • This is why coastal areas have milder climates.

Kelley’s Island • Has the same weather as northern Ohio except… • “The still warm lake keeps us warmer in the fall, but the still cool lake gives us cold breezes in the spring. ”

Water expands upon freezing • If water worked like other liquids, then there would be no such thing as an ice berg, the ice cubes in your soft drink would sink to the bottom of the glass, and ponds would freeze from the bottom up!
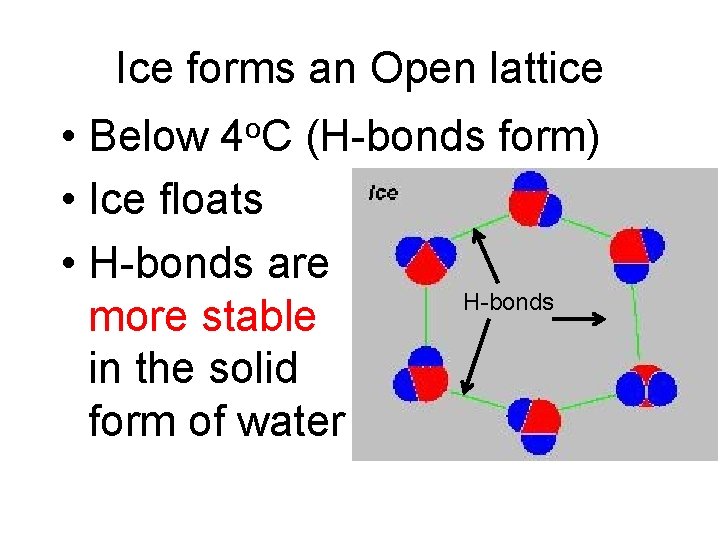
Ice forms an Open lattice • Below 4 o. C (H-bonds form) • Ice floats • H-bonds are H-bonds more stable in the solid form of water

Ice Expands and is Less Dense than Liquid Water 4 o. C Liquid Solid
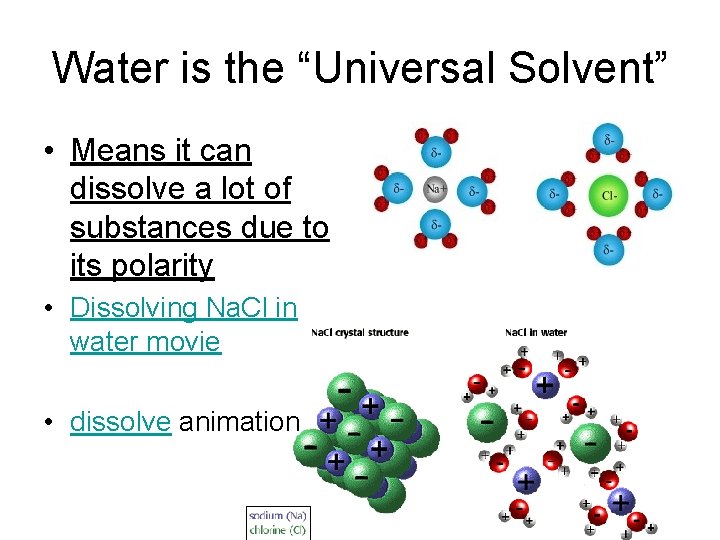
Water is the “Universal Solvent” • Means it can dissolve a lot of substances due to its polarity • Dissolving Na. Cl in water movie • dissolve animation

Water is the Universal Solvent • What types of compounds are good at dissolving? – Polar – Ionic – WHY? – Opposite charges attract – Remember: Likes dissolve likes: Polar into polar/nonpolar into nonpolar

Aqueous Solution • Means a solution in which water is the solvent • Ag+(aq) + Cl-(aq) → Ag. Cl(s) • EXAMPLES: blood, Coke, Pepsi, urine, Tea, coffee

Mixtures-Solutions-Suspensions • Mixtures: 2 or more elements physically mixed (NOT CHEMICALLY BONDED) – A. Solutions: type of mixture with evenly distributed ions throughout – B. Suspensions: Mixture of water and undissolved material keeping the small materials suspended

Can you identify: Mixtures. Solutions-Suspensions? Oil and Water Sand Salt

What is the difference?

What’s the difference between… • Mixtures, solutions, suspensions, and COMPOUNDS? COMPOUNDS ARE CHEMICALLY JOINED…the others are physically mixed.

SOLUTIONS • When one substance dissolves another substance. • SOLVENT + SOLUTE = SOLUTION • Solvent=material doing the dissolving (surrounding the ions) • Solute=what is being dissolved

Salt Solution Which is the solvent? Which is the solute? Water is solvent. Salt is the solute.
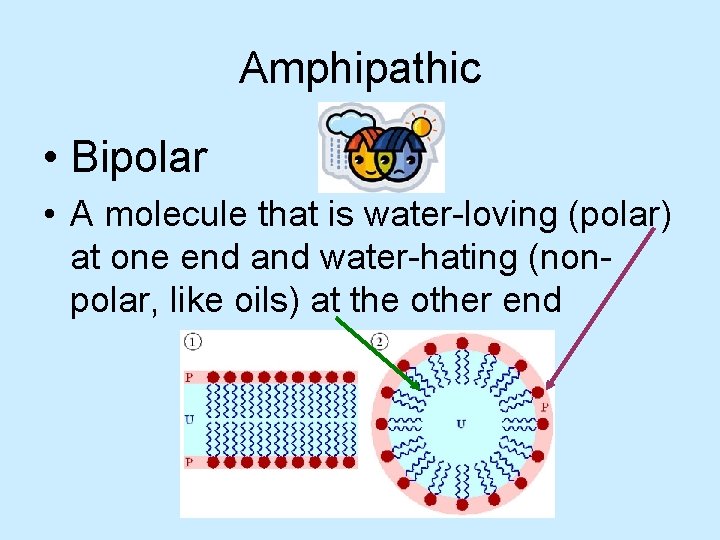
Amphipathic • Bipolar • A molecule that is water-loving (polar) at one end and water-hating (nonpolar, like oils) at the other end
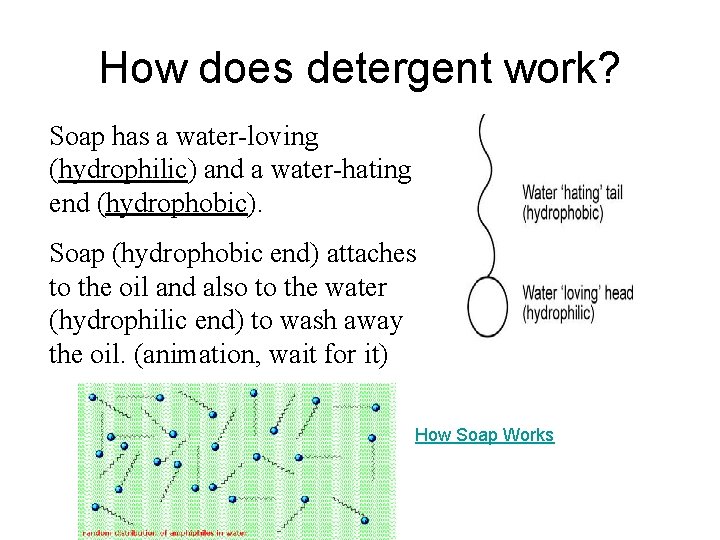
How does detergent work? Soap has a water-loving (hydrophilic) and a water-hating end (hydrophobic). Soap (hydrophobic end) attaches to the oil and also to the water (hydrophilic end) to wash away the oil. (animation, wait for it) How Soap Works

Capillary Action Capillary action – the narrower the tube the higher the water rises a. Adhesion - of water to tube b. Cohesion-water to water (column of water) Meniscuswater creeps up the sides of the glass tube

Note size of tube diameter and the height the liquid rises
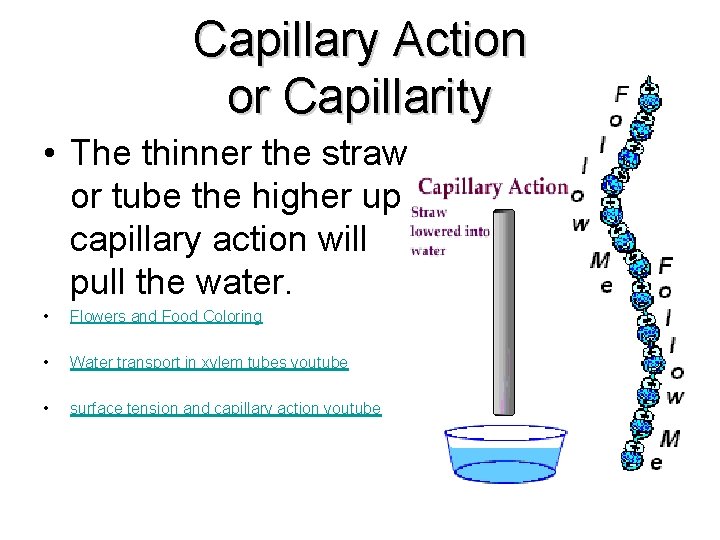
Capillary Action or Capillarity • The thinner the straw or tube the higher up capillary action will pull the water. • Flowers and Food Coloring • Water transport in xylem tubes youtube • surface tension and capillary action youtube
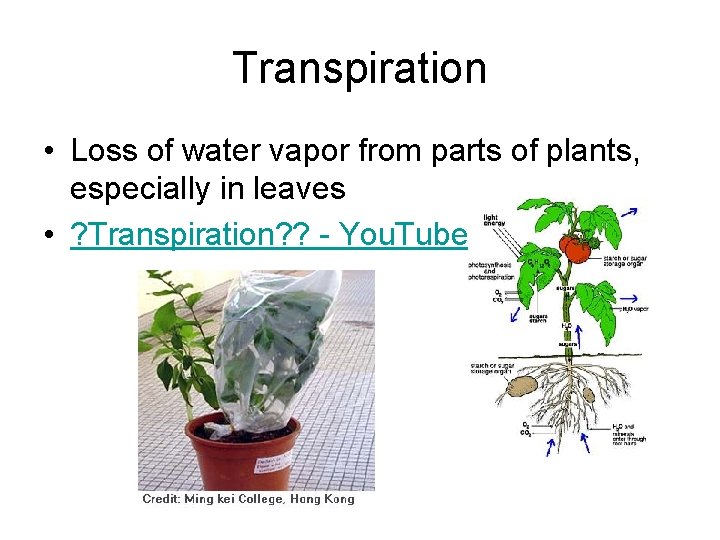
Transpiration • Loss of water vapor from parts of plants, especially in leaves • ? Transpiration? ? - You. Tube

Explain how each of these allow water to pull up a plant? • Cohesion • Adhesion • Transpiration ? Transpiration? ? - You. Tube • Can you explain what is going on in this video? • ? Flowers and Food Color Time Lapse? ? You. Tube

Capillarity in Plants • Surface Tension and Capillarity You Tube Video • Water Transport in Plants

Solutions • A SOLUTE is the substance to be dissolved (sugar). • The SOLVENT is the one doing the dissolving (water).

What is the difference?

SALT SOLUTION • Animation of Salt Dissolving in Water • Dissolve Another Nice Animation • When water is the solvent, the solution is called an aqueous solution.

FYI • The solvent is defined as the substance that exists in a greater quantity than the solute(s) in the solution. • Generally polar or ionic compounds will only dissolve in polar solvents.
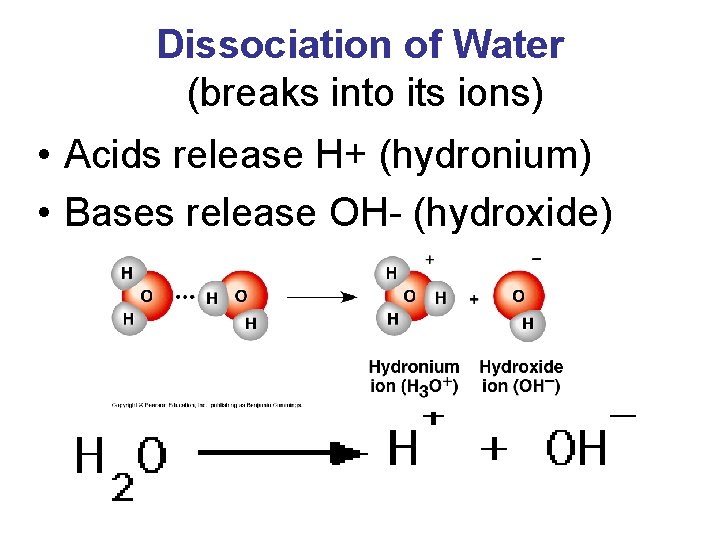
Dissociation of Water (breaks into its ions) • Acids release H+ (hydronium) • Bases release OH- (hydroxide)
Color Pencils and Handout • Animations of Transfer of H+ ions • Acid Strength Animations (contrast the H+ concentrations of HCl and HF)
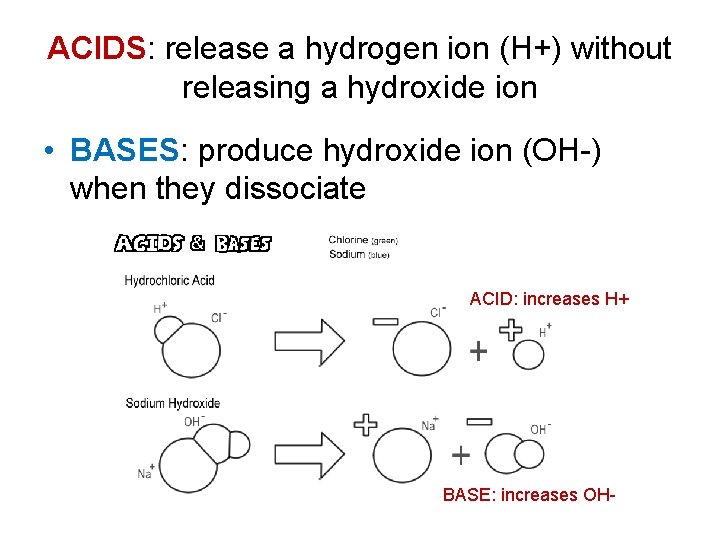
ACIDS: release a hydrogen ion (H+) without releasing a hydroxide ion • BASES: produce hydroxide ion (OH-) when they dissociate ACID: increases H+ BASE: increases OH-
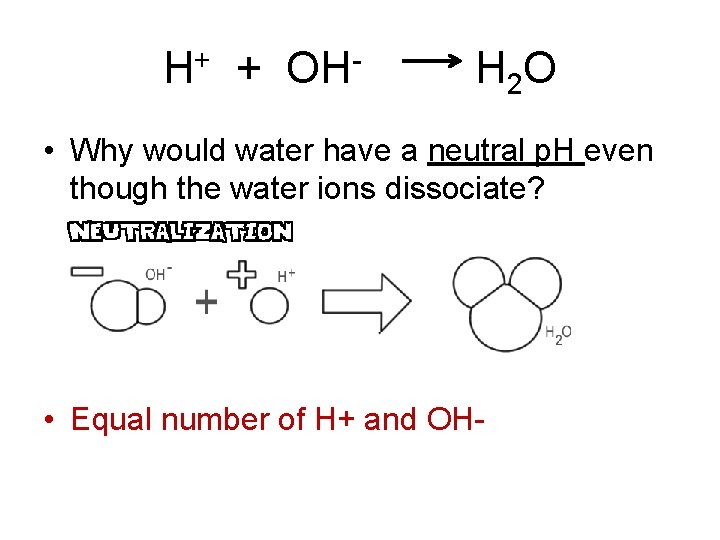
H+ + OH- H 2 O • Why would water have a neutral p. H even though the water ions dissociate? • Equal number of H+ and OH-
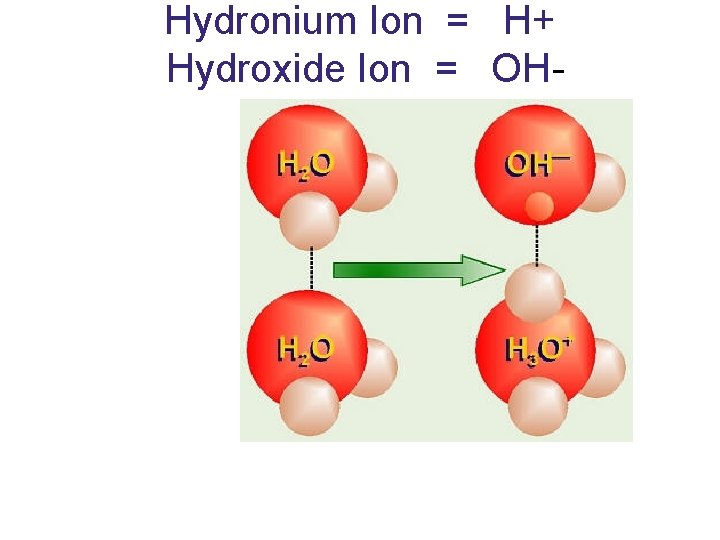
Hydronium Ion = H+ Hydroxide Ion = OH-

• Animations of Acids and Bases •

p. H at p. H 7. 0, a solution is neutral (H+ = OH-) • * at lower p. H (1 -6), a solution is acidic (H+ > OH-) • * at higher p. H (8 -14), a solution is basic (H+ < OH-) • *

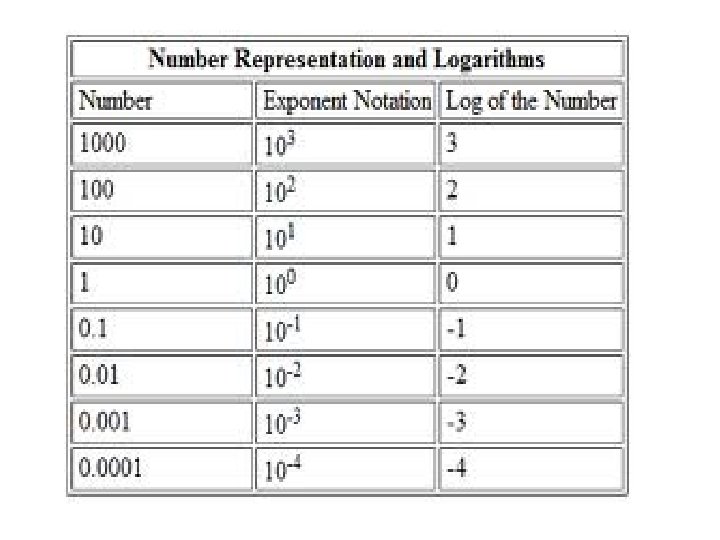
p. H • = the negative logarithm (base 10) of the molar concentration of dissolved hydronium ions (H 3 O+). SAY WHAT? • a low p. H indicates a high concentration of hydronium ions (acidic) • a high p. H indicates a low concentration of H+ ions (basic) • Bozeman: Acids, Bases, and p. H (13: 13)

• This negative of the logarithm matches the number of places behind the decimal point. • For example, 0. 1 molar hydrochloric acid should be near p. H 1 and 0. 0001 molar HCl should be near p. H 4 (the base 10 logarithms of 0. 1 and 0. 0001 being 10− 1, and 10− 4, respectively). • What would be the p. H of 0. 0000001 M H+ or 10 -7? • p. H 7
![• • • • p. H [H+] [OH-] [H+] X [OH-] 0 1 • • • • p. H [H+] [OH-] [H+] X [OH-] 0 1](http://slidetodoc.com/presentation_image_h2/450d8e5a26f1d66430b2a3042ae54aa2/image-62.jpg)
• • • • p. H [H+] [OH-] [H+] X [OH-] 0 1 2 3 4 5 6 7 8 9 10 11 12 13 1 X 10 -0 1 X 10 -1 1 X 10 -2 1 X 10 -3 1 X 10 -4 1 X 10 -5 1 X 10 -6 1 X 10 -7 1 X 10 -8 1 X 10 -9 1 X 10 -10 1 X 10 -11 1 X 10 -12 1 X 10 -13 1 X 10 -14 1 X 10 -13 1 X 10 -12 1 X 10 -11 1 X 10 -10 1 X 10 -9 1 X 10 -8 1 X 10 -7 1 X 10 -6 1 X 10 -5 1 X 10 -4 1 X 10 -3 1 X 10 -2 1 X 10 -14 1 X 10 -14 1 X 10 -14 1 X 10 -14 • 14 1 X 10 -0 1 X 10 -14 acid neutral base
![p. H = -log[H+] • At equilibrium, the concentration of H+ is 1. 00 p. H = -log[H+] • At equilibrium, the concentration of H+ is 1. 00](http://slidetodoc.com/presentation_image_h2/450d8e5a26f1d66430b2a3042ae54aa2/image-63.jpg)
p. H = -log[H+] • At equilibrium, the concentration of H+ is 1. 00 × 10 -7, so we can calculate the p. H of water at equilibrium as: • p. H = -log[H+] • = -log[1. 00 × 10 -7] • So p. H = 7. 00
![p. OH = -log[OH-] • NEUTRAL • p. H+ + p. OH- = 14. p. OH = -log[OH-] • NEUTRAL • p. H+ + p. OH- = 14.](http://slidetodoc.com/presentation_image_h2/450d8e5a26f1d66430b2a3042ae54aa2/image-64.jpg)
p. OH = -log[OH-] • NEUTRAL • p. H+ + p. OH- = 14. 00 at 25°C

What is H+ when OH - = 1 x 10 – 9 M? • p. H + p. OH = 14. (p. H is the negative log of the concentration of H+ ions, while p. OH- is the negative log of the concentration of OH- ions. ) If the concentration of OH- is 10 -9 = p. H 9 Subtract that from 14 to get the p. H = 14 – 9 = p. H 5
![Two Ways to Contrast Acids and Bases • acid: generates [H+] in solution base: Two Ways to Contrast Acids and Bases • acid: generates [H+] in solution base:](http://slidetodoc.com/presentation_image_h2/450d8e5a26f1d66430b2a3042ae54aa2/image-66.jpg)
Two Ways to Contrast Acids and Bases • acid: generates [H+] in solution base: generates [OH-] in solution • acid: anything that donates a [H+] (proton donor) base: anything that accepts a [H+] (proton acceptor) • Good Activities on p. H understanding (The find the answers areas)
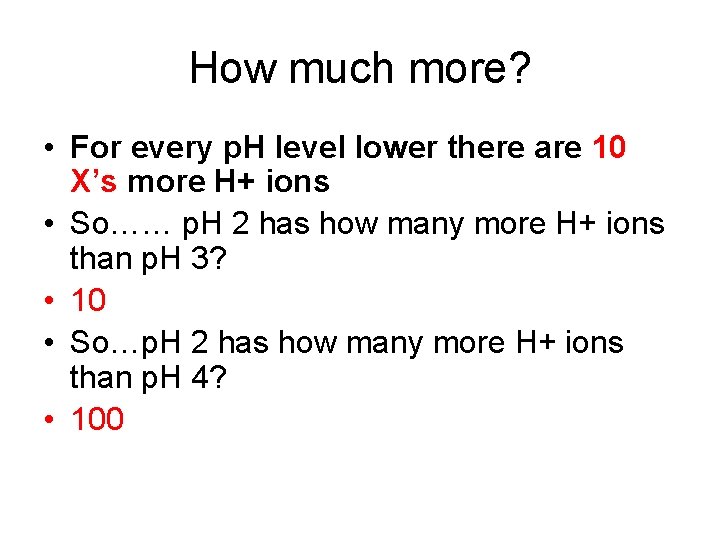
How much more? • For every p. H level lower there are 10 X’s more H+ ions • So…… p. H 2 has how many more H+ ions than p. H 3? • 10 • So…p. H 2 has how many more H+ ions than p. H 4? • 100
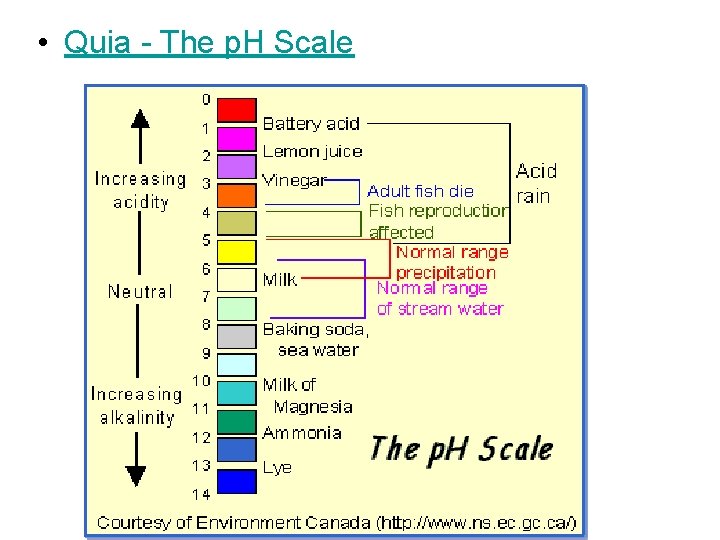
• Quia - The p. H Scale


• When acids or bases are added to pure water, they immediately produce H 3 O+ or OH- ions that decrease or increase the p. H, respectively. • Buffer systems resist large p. H changes because added acids or bases are neutralized by the existing HA/A(acid/base) system (equilibrium).

Where are buffers important? • Keeping p. H in the blood constant (normal blood p. H is 7. 4)

Homeostasis: regulation of the internal environment and to maintain a stable, constant condition • The body is very sensitive to its p. H level, so strong mechanisms exist to maintain it. • Discovery Health "Homeostasis"

Kidneys regulating homeostasis • The body needs to have the consistent and proper amount of water, salt, and acid in the blood. The kidney keeps these things constant. • If there is too much water, the kidney puts more water in the urine. If there is not enough water, the kidney puts less water in the urine.

Where are buffers important? • Pharmaceuticals • Industry (cleaners, cosmetics, aquarium) • Working with enzymes

Get Extended Response Assignment • Choose two ways the body maintains homeostasis. DUE: _______ WORTH: _____
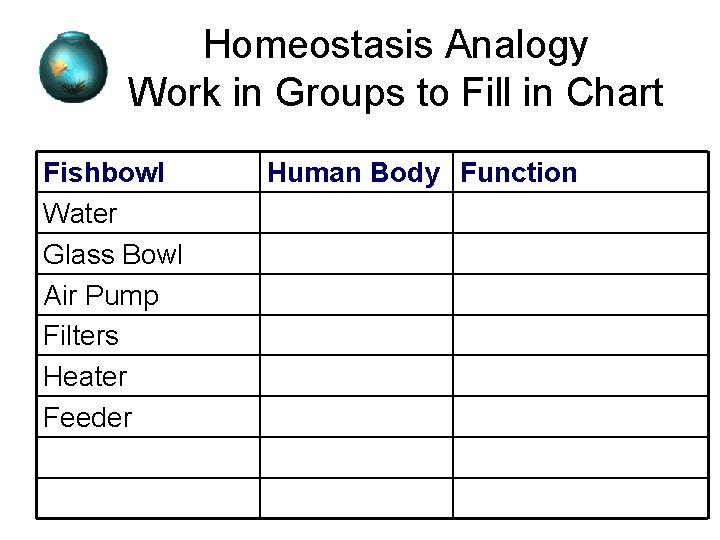
Homeostasis Analogy Work in Groups to Fill in Chart Fishbowl Water Glass Bowl Air Pump Filters Heater Feeder Human Body Function
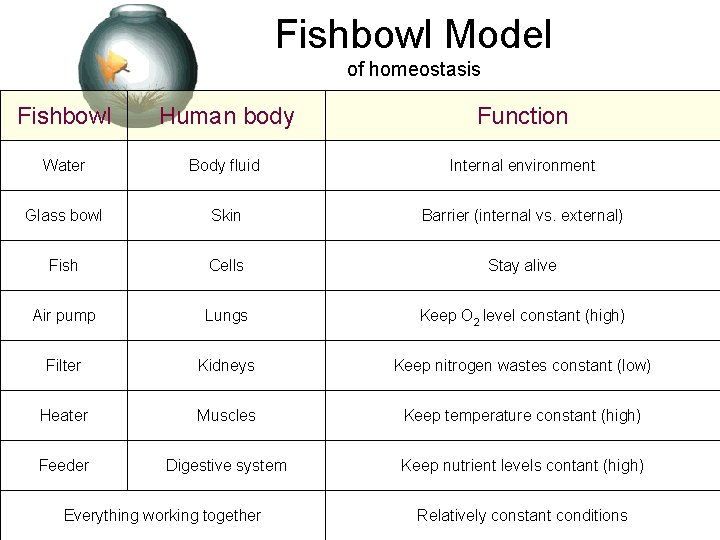
Fishbowl Model of homeostasis Fishbowl Human body Function Water Body fluid Internal environment Glass bowl Skin Barrier (internal vs. external) Fish Cells Stay alive Air pump Lungs Keep O 2 level constant (high) Filter Kidneys Keep nitrogen wastes constant (low) Heater Muscles Keep temperature constant (high) Feeder Digestive system Keep nutrient levels contant (high) Everything working together Relatively constant conditions

p. H Problems • Acid Precipitation: burning of fossil fuels (coal, oil, gas) alter p. H • Sulfur and nitrogen dioxides add to water and make sulfuric acid and nitric acid. • Map of US area acid precipitation p. H

p. H Problems • Ocean acidification = CO 2 dissolved in water lowers p. H (more acidic) to make carbonic acid • CO 2 + H 2 O H 2 CO 3 • Reduces carbonate ions to make shells, coral, and skeletons.

Properties of Water Lab

Overall Review • Bozeman Properties of Water (10 minutes)
- Slides: 81