Water and Ocean Structure Chapter 6 The Water





















- Slides: 33

Water and Ocean Structure Chapter 6

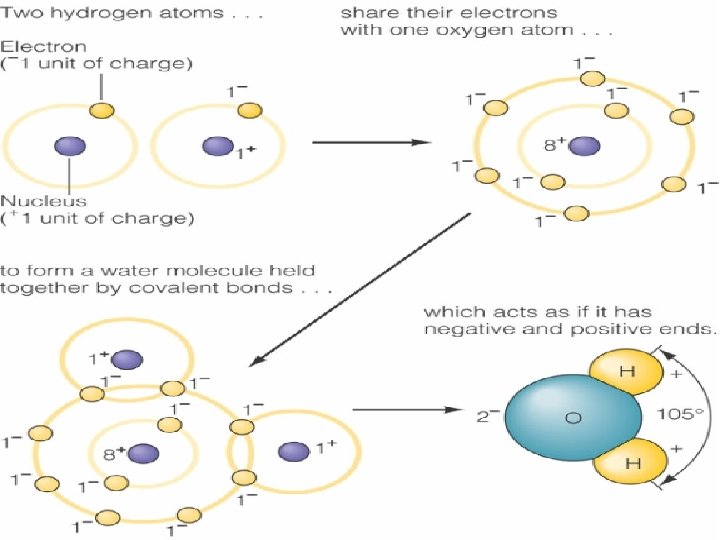
The Water Molecule • Molecule – Group of atoms held together by chemical bonds • Covalent bonds – Pairs of electrons are shared – Sharing leads to an unequal distribution of charge o v 104. 5 bond angle (H-O-H)


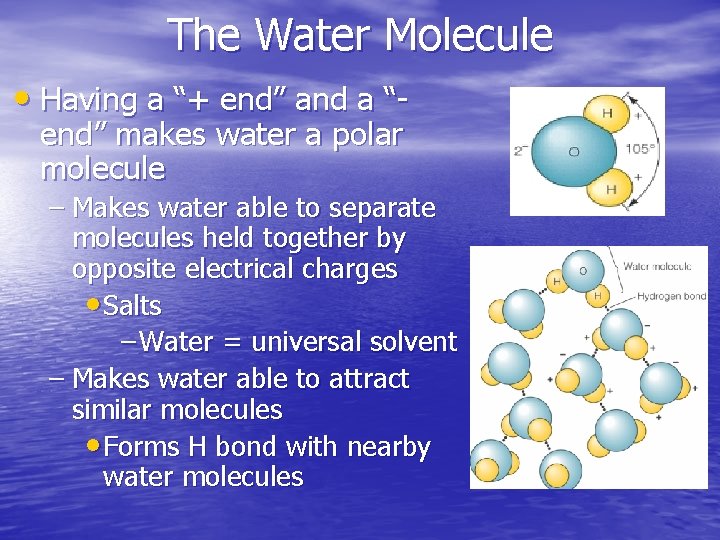
The Water Molecule • Having a “+ end” and a “end” makes water a polar molecule – Makes water able to separate molecules held together by opposite electrical charges • Salts – Water = universal solvent – Makes water able to attract similar molecules • Forms H bond with nearby water molecules

The Water Molecule • Cohesion – The sticking together of individual water molecules • Surface tension • Adhesion – The sticking of water to solids • Makes thing wet • Both lead to capillary action


Water and Heat • Heat – The energy produced by the random vibration of atoms or molecules • How many and how rapidly • Temperature – An object’s response to and input (or removal) of heat • Different substances = different responses • How rapidly • More heat energy – Hot bath or candle flame?

• Degrees – Temperature measurement • Celsius vs. Fahrenheit – 1 o. C = 1. 8 o. F • Conversions – o C o. F o vo F = C x (9/5) + 32 vo F = 20 o. C x (9/5) + 32 = 68 o. F – o o F C vo C = (o. F – 32) x 5/9 vo C = (60 o. F – 32) x 5/9 = 15. 6 o. C

Water and Heat • Heat Capacity – A measure of the heat required to raise the temperature of 1 g of a substance by 1 o. C • Calories per gram – Calorie v. The amount of heat required to raise the temperature of 1 g of water by 1 o. C – Heat capacity of water one of the highest of all substances • Can absorb (or release) large amounts of heat without large changes in temperature

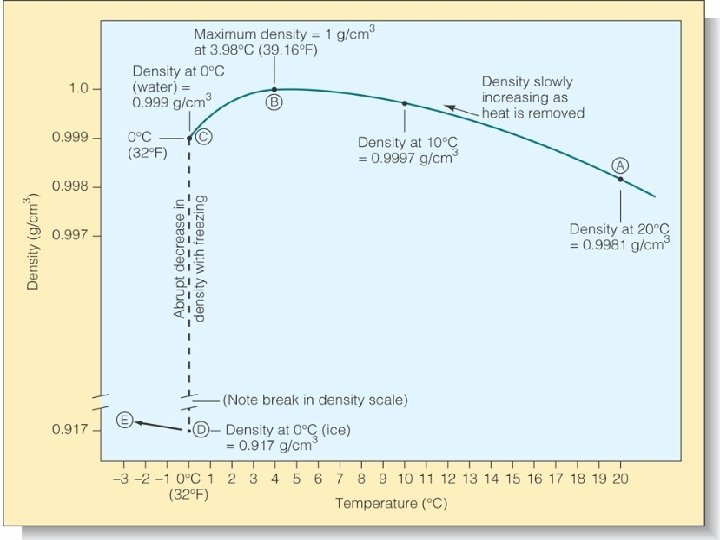
Water Temp. and Density • Density = mass per unit volume – Most substances become denser as they get colder • Water becomes more dense as heat is removed…


Water Temp. and Density …until it approaches its freezing point • Density quickly decreases as it turns from water to ice – Freezing o v. Bond angle changes from 104. 5 to 109 o v. Forms regular hexagons v. Water expands by ~9%

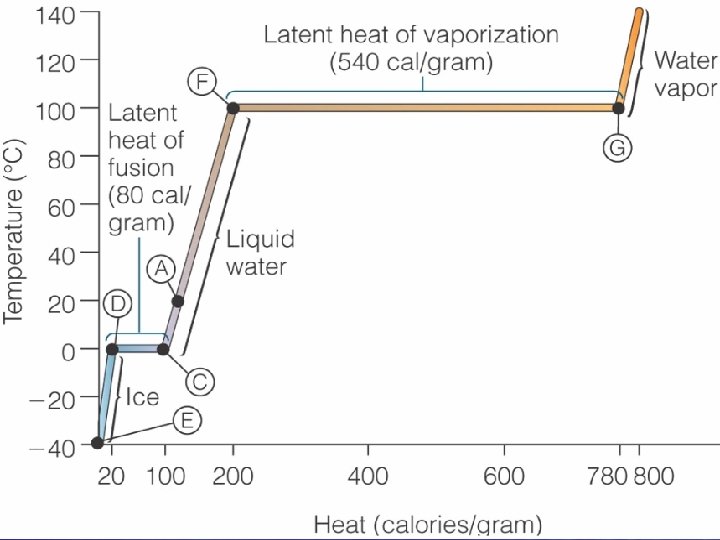
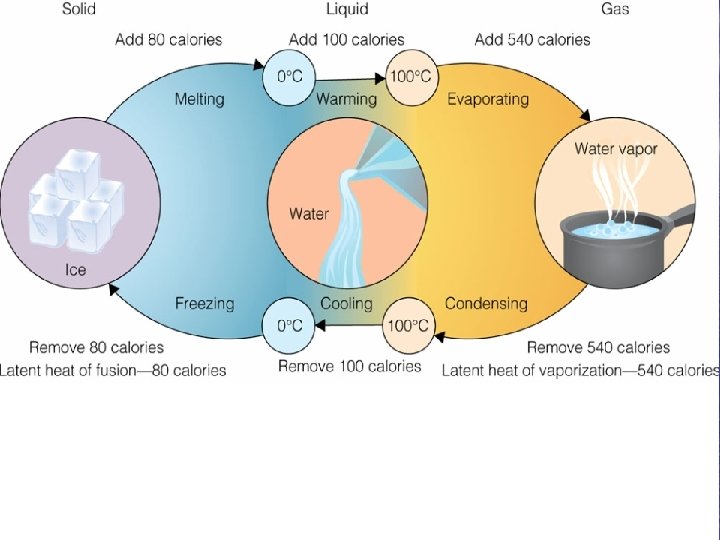
Latent Heats • Latent heat of fusion – Heat removed during freezing that does not change the temperature • 80 calories of heat per 1 g of water at 0 o. C to form ice • Latent heat of vaporization – Heat added during evaporation that produces a change in state but does not cause a change in temperature • 540 calories per 1 g of water at 20 o. C – Highest on Earth



Seawater vs. Pure Water • Seawater – 96. 5% pure water – 3. 5% dissolved solids and gasses • Lower its latent heat by ~4% • Act as a natural antifreeze – Salinity goes up freezing point goes down o v. Freezing point of seawater = -1. 9 C (28. 6 o. F)

Seawater vs. Pure Water • Both freezing and evaporating seawater leaves salt behind – Leftover dense water sinks to the bottom • Seawater evaporates more slowly than freshwater – 3. 3 cubic feet of the sea evaporates per year • Left over heat powers wind waves, storms, and currents


Global Thermostatic Effects • Thermostatic properties – Properties of water that act to moderate changes in temperature • Thermal inertia – Resisting temperature changes with the input or loss of heat – Temp differences • Land – 250 o. F • Ocean – 61 o. F


Effects of Water and Air Movement • Why doesn’t the water at the equator boil away or the oceans at the poles entirely freeze solid? – Currents move heat in water/air from tropics to poles • Equalizes polar-tropical heat imbalance

Effects of Air and Water Movement • Example – Gulf Stream – “outgoing” water is 10 o. C warmer than “incoming” water v 10 million calories/meter 3 v. Flow = 55 million meter 3/second v 550 trillion calories moved North/second – Energy used to evaporate ocean is released to make water vapor • Usually at a distance – Cools Cuba warms Canada


Density Structure of the Ocean • Liter of seawater – 2% to 3% more dense than pure water – Density of seawater = 1. 020 – 1. 030 g/cm 3 – Dissolved solids (“salt”) • Density goes up when the… – Temperature goes down – Pressure goes up – Salinity (how salty) goes up

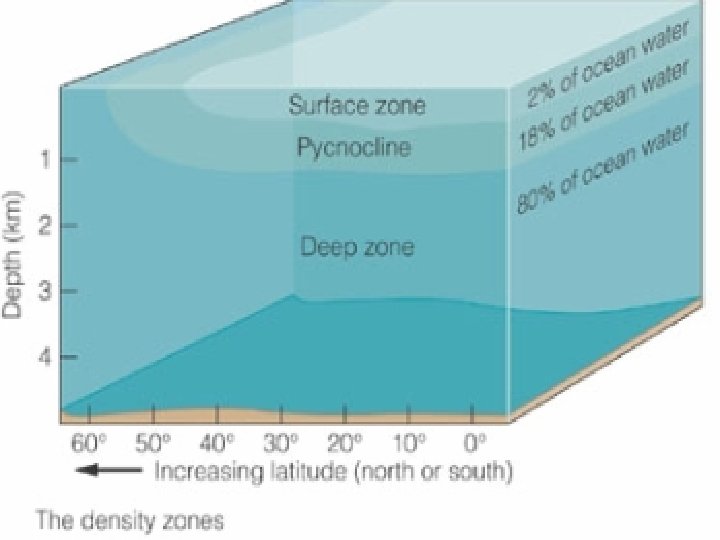
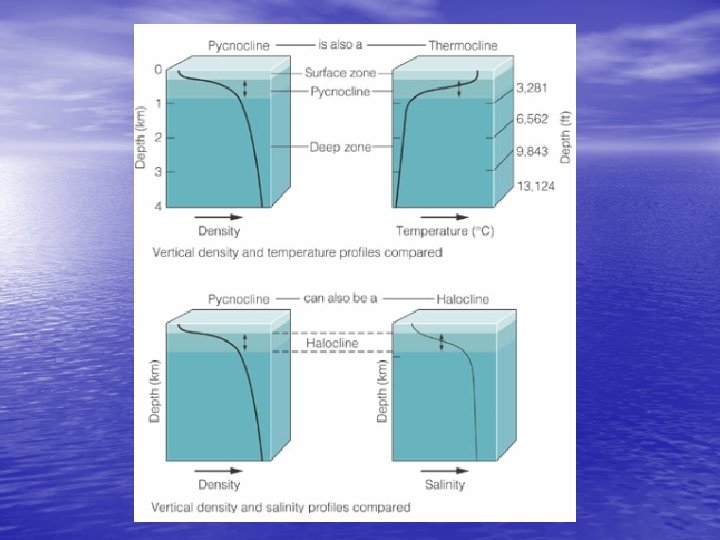
Density Structure of the Ocean • Density zones – Surface zone (mixed layer) (2%) • Least dense • Temp and salinity mostly constant throughout – Pycnocline (18%) • More dense • Density increases with depth – Deep zone (80%) • Most dense (below 1, 000 m) • Little change in density with depth


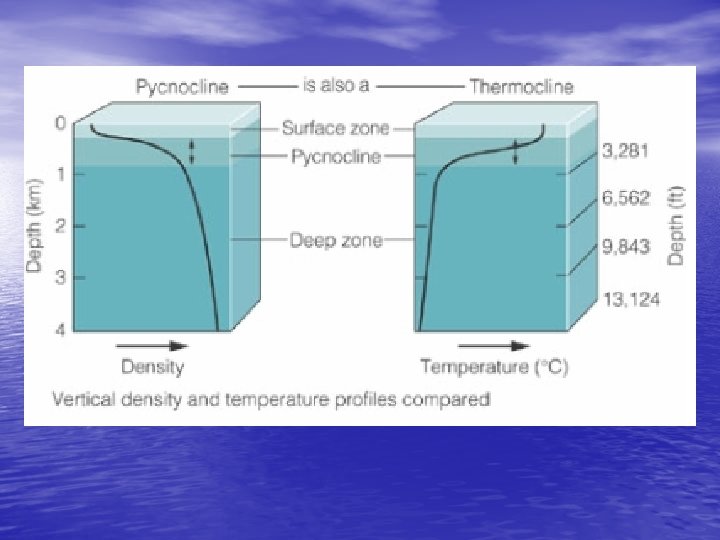
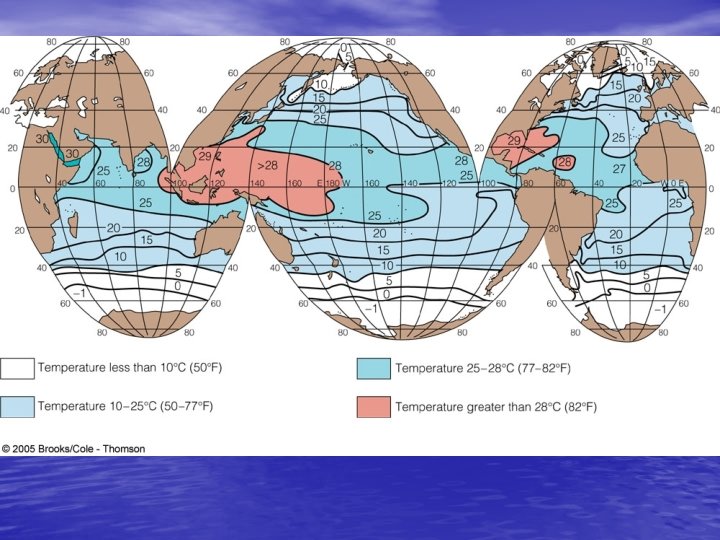
Density Structure of the Ocean • Temperature and stratification – Pycnocline changes density so rapidly due to temperature change • Thermocline – Regional differences? Factors? – Tropical regions – deep thermocline – Polar regions – no thermocline


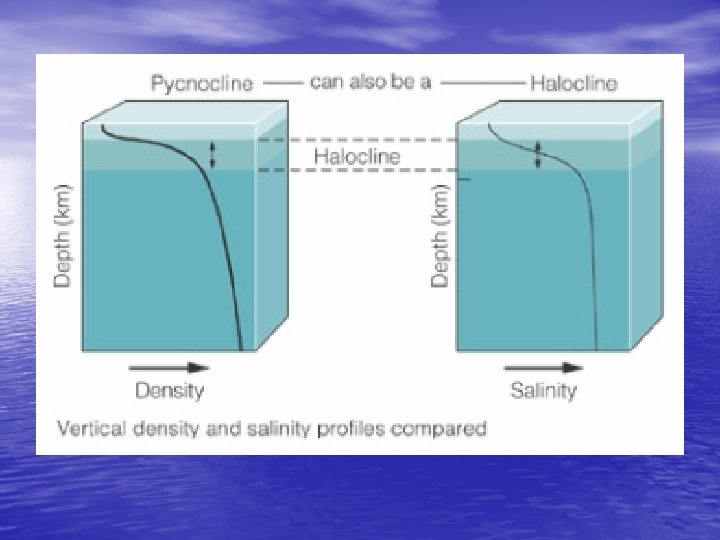
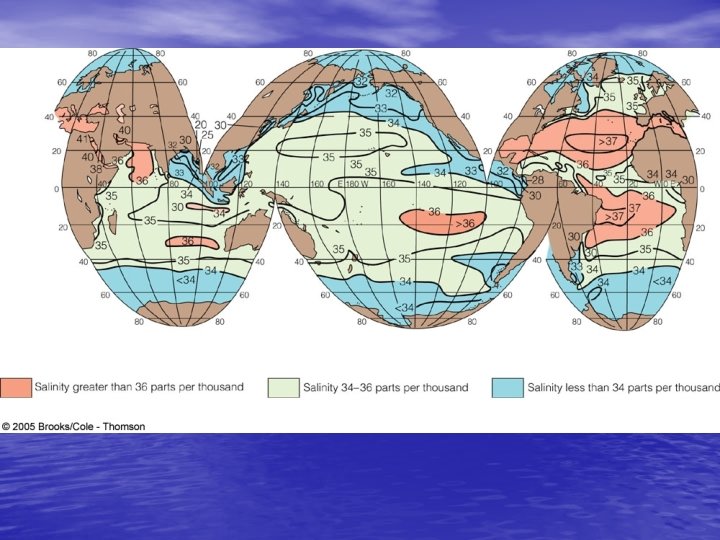
Density Structure of the Ocean • Salinity and stratification – Low salinity creates stratification • Wherever precipitation exceeds evaporation – Creates the halocline v. Zone of rapid salinity increase with depth • Thermocline (temp) + Halocline (salinity) = Pycnocline (density) – Water mass



