Water and Its Properties Honors Biology Water Its

Water and It’s Properties Honors Biology

Water It’s 45 – 50 % of the weight of any functioning living system. n It’s 70 – 95 % of the human cell. n It’s the most common liquid on Earth. n 75% of the Earth’s surface is covered by it. n

States of Water n Solid = Ice n Liquid = Water n Gas = Steam
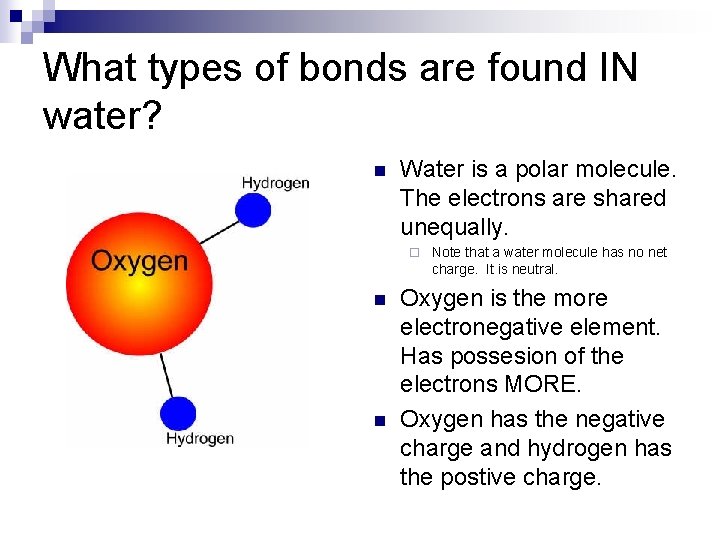
What types of bonds are found IN water? n Water is a polar molecule. The electrons are shared unequally. ¨ n n Note that a water molecule has no net charge. It is neutral. Oxygen is the more electronegative element. Has possesion of the electrons MORE. Oxygen has the negative charge and hydrogen has the postive charge.

Hydrophobic vs. Hydrophilic Hydro = water n Phobic = fearing n Philic = loving n Hydrophobic substances are nonpolar n Hydrophilic substances are either polar or ionic. n
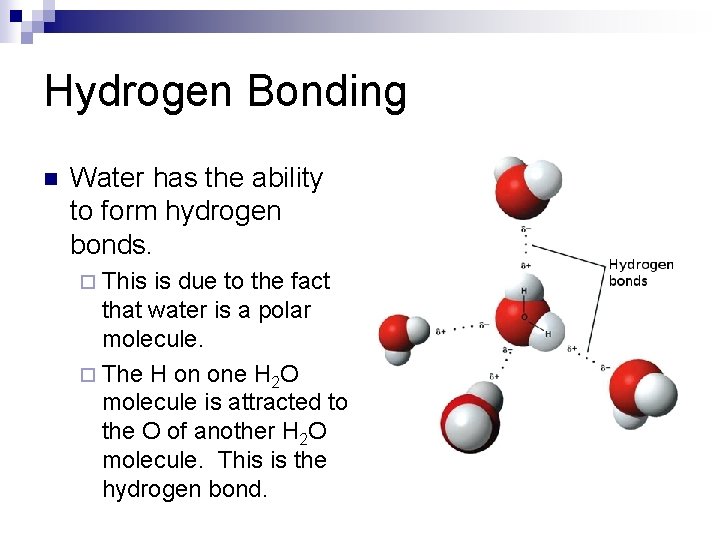
Hydrogen Bonding n Water has the ability to form hydrogen bonds. ¨ This is due to the fact that water is a polar molecule. ¨ The H on one H 2 O molecule is attracted to the O of another H 2 O molecule. This is the hydrogen bond.

Special Properties of Water 1. 2. 3. 4. 5. Cohesion and Adhesion Good Solvent Lower Density as a Solid High Specific Heat High Heat of Vaporization

Cohesion and Adhesion n n Cohesion occurs when a water molecule bonds to another water molecule. It has the capacity to resist rupturing under tension. ¨ Surface tension ¨ Drinking straw Adhesion occurs when a water molecule bonds to another polar substance. ¨ Capillary action ¨ Meniscus

How does water get to the top of plants? n Transpiration built on adhesion and cohesion. The adhesion allows water to “bond” to the xylem. ¨ The cohesiveness keeps the water molecules together in a continuous “chain”. ¨

Universal Solvent n Why? Water is a polar substance. This gives water positive and negative ends. Most other substances are with polar or ionic giving them positive and negative ends. This makes water “attracted” to many different substances. The solution process is now able to occur.
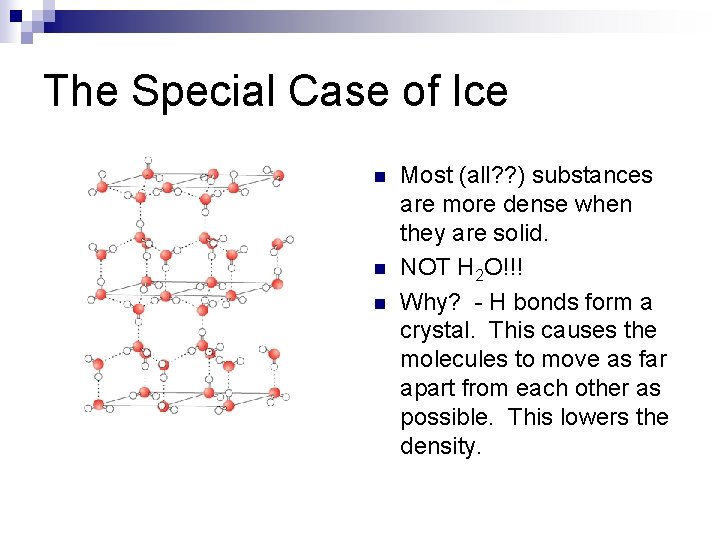
The Special Case of Ice n n n Most (all? ? ) substances are more dense when they are solid. NOT H 2 O!!! Why? - H bonds form a crystal. This causes the molecules to move as far apart from each other as possible. This lowers the density.

Why is “ice floats” important? n Oceans and lakes don’t freeze solid. ¨ Surface n ice insulates water below If ice sank…. ¨ Ponds, lakes and even oceans would freeze solid. ¨ In summer, only upper few inches would thaw. n Seasonal turnover of lakes. ¨ Cycling of nutrients in autumn

Specific Heat n n Specific heat = the amount of heat that must be absorbed or lost for 1 g to change its temperature by 1 C. Water has a high specific heat therefore resists changes in temperature. ¨ Takes a lot to heat it up. ¨ Takes a lot to cool it down.

Heat of Vaporization n Heat of Vaporization is the amount of heat a liquid most absorb for 1 g of it to be converted from a liquid to a gas. Organisms rely on heat of vaporization to remove body heat. Think about how cool the back of your neck feels when you sweat.

Why does water have all of these properties? ? n Water has all of these properties because of………. HYDROGEN BONDING
![p. H Scale Acid = p. H below 7 [H+] > [OH-] n Base p. H Scale Acid = p. H below 7 [H+] > [OH-] n Base](http://slidetodoc.com/presentation_image/3c7033d5395e58934665d060b4774071/image-16.jpg)
p. H Scale Acid = p. H below 7 [H+] > [OH-] n Base = p. H above 7 [H+] < [OH-] n Neutral [H+] = [OH-] Acid Ion = H+ or H 3 O+ Base Ion = OHn

More about p. H n n n Most biological fluids have a p. H between 6 and 8. The internal p. H of our cells is about 7. 4. Slight changes can be harmful. Drop in p. H is called acidosis. Increase in p. H is called alkalosis A person can not survive more than a few minutes if p. H drops to 7 or rises to 7. 8.

Acid Precipitation n Refers to rain, snow or fog with a p. H below 5. 6. Caused by the presence of sulfur oxides and nitrogen oxides in the atmosphere. Think about why we have acid rain.
- Slides: 18