Watch Video and Answer Questions https www youtube

Watch Video and Answer Questions https: //www. youtube. com/watch? v=59 m-0 ZGk. Bn. Q&list=UUM 1 rx 9 Lq. Piv 3 LDn. Jmw 6 -Rp. A
The Building Blocks of Matter: Atoms - ++ + + + - -
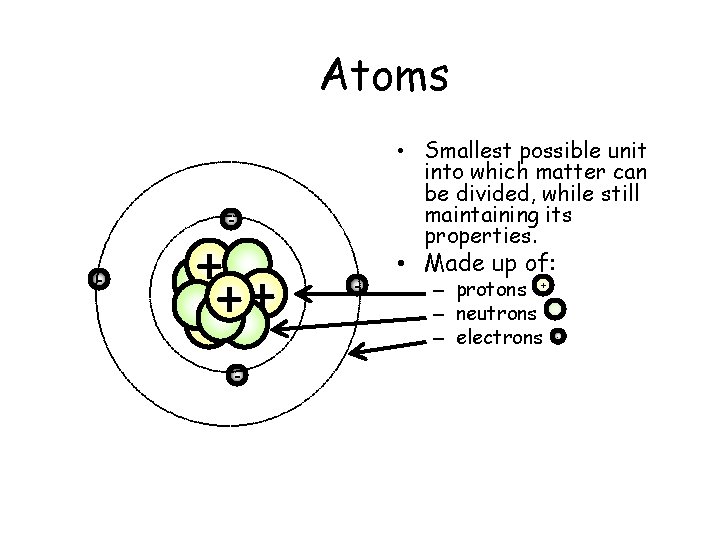
Atoms • Smallest possible unit into which matter can be divided, while still maintaining its properties. - - + + - - • Made up of: – protons + – neutrons – electrons -

Element • An element is a substance that is made entirely from one type of atom. • For example, the element hydrogen is made from atoms containing a single proton and a single electron. • If you change the number of protons an atom has, you change the type of element it is.

Element vs Atom Imagine going to an ice cream store. • Let's say that they have 30 different flavors of ice cream. – Those are elements, the things that I have available to build my dessert from. • The smallest amount of ice cream that the store will sell to me is a scoop. – This is an atom.

WHAT IS AN ATOM? o The smallest unit of matter that retains the identity of the substance o Consists of a central nucleus surrounded by one or more electrons.
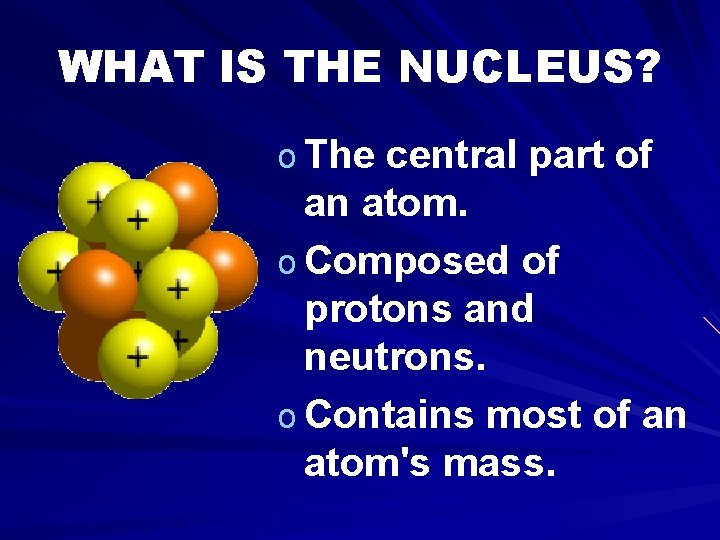
WHAT IS THE NUCLEUS? o The central part of an atom. o Composed of protons and neutrons. o Contains most of an atom's mass.

WHAT IS A PROTON? o Positively charged particle. o Found within an atomic nucleus.

WHAT IS A NEUTRON? o Uncharged particle. o Found within an atomic nucleus.

WHAT IS AN ELECTRON? o Negatively charged particle. o Located around the nucleus in the electron orbit or cloud.

How do these particles interact? Protons and neutrons live compacted in the tiny positively charged nucleus accounting for most of the mass of the atom The negatively charged electrons are small and have a relatively small mass but occupy a large volume of space outside the nucleus

How do the subatomic particles balance each other? – The protons = the electrons If 20 protons are present in an atom then 20 electrons are there to balance the overall charge of the atom—atoms are neutral – The neutrons have no charge; therefore they do not have to equal the number of protons or electrons

Basic Atomic Structure Video and ws https: //www. youtube. com/watch? v=eq. TNNxt. Gyz. E&list=UUM 1 rx 9 Lq. Piv 3 LDn. Jmw 6 -Rp. A

Read “An Atom Apart” and answer questions Read “Splitting the Atom” and Answer questions

• In Workbook do: p. 67 # 6, 7 : p. 68 #10 p. 73 #8, 9 : p. 77 # 8
- Slides: 15