Wastewater treatment Removal of dissolved inorganic substances Associate

Wastewater treatment. Removal of dissolved inorganic substances Associate Prof. Tatyana Solodovnik Cherkasy State Technological University solodovniktetana@gmail. com

Removal of dissolved inorganic substances Contents Types of wastewater pollution Sources of contamination with dissolved inorganic substances Methods of sewage treatment from dissolved inorganic substances Methods for cleaning industrial wastewater from typical inorganic impurities Wastewater treatment 2

Types of wastewater pollution § Mechanical § Chemical: organic and inorganic solutes § Biological and bacteriological § Thermal - water from thermal power plants and nuclear power plants § Radioactive http: //rosao. ru/information/articles/205 Wastewater treatment 3
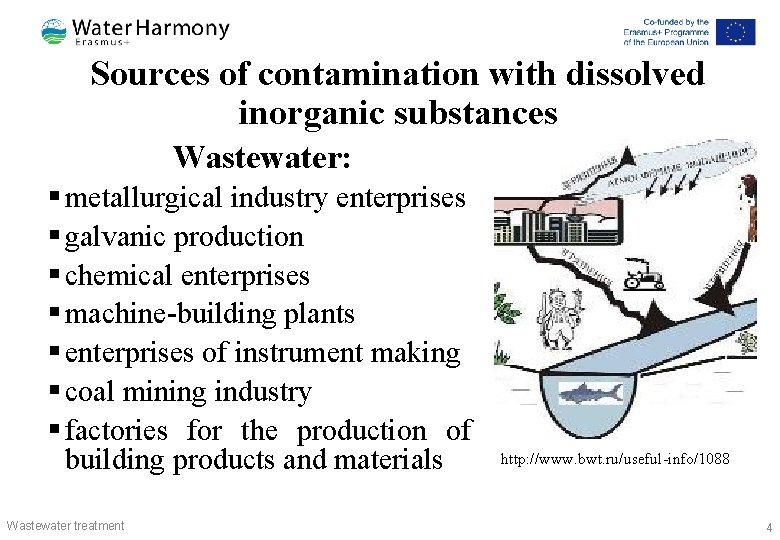
Sources of contamination with dissolved inorganic substances Wastewater: § metallurgical industry enterprises § galvanic production § chemical enterprises § machine-building plants § enterprises of instrument making § coal mining industry § factories for the production of building products and materials Wastewater treatment http: //www. bwt. ru/useful-info/1088 4
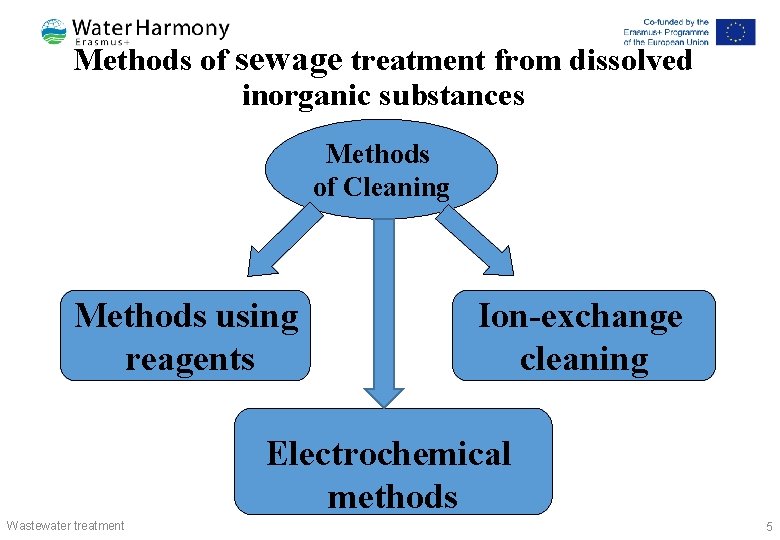
Methods of sewage treatment from dissolved inorganic substances Methods of Cleaning Methods using reagents Ion-exchange cleaning Electrochemical methods Wastewater treatment 5
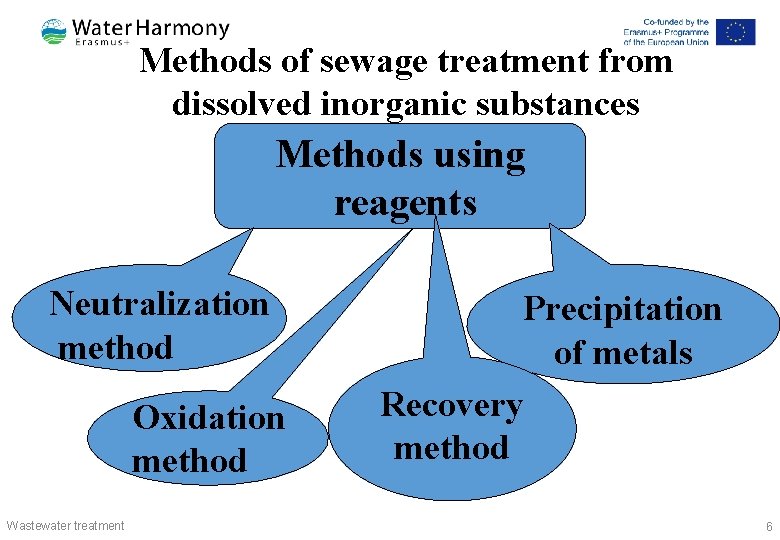
Methods of sewage treatment from 4 CH 3 dissolved inorganic substances COOH + 4 H 2 → 5 CH 4 + 2 H 2 O + 3 CO 2 4 H 2 + СО 2 → CH 4 + 2 H 2 O Methods using reagents Neutralization method Oxidation method Wastewater treatment Precipitation of metals Recovery method 6
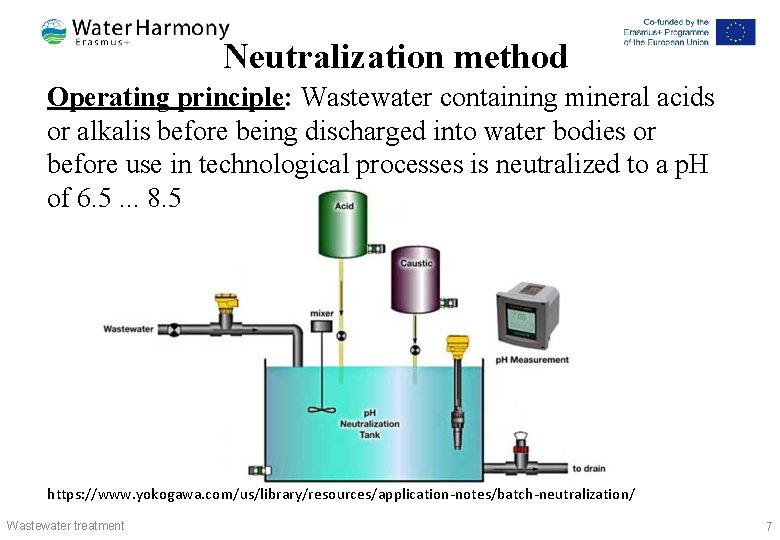
Neutralization method Operating principle: Wastewater containing mineral acids or alkalis before being discharged into water bodies or before use in technological processes is neutralized to a p. H of 6. 5. . . 8. 5 https: //www. yokogawa. com/us/library/resources/application-notes/batch-neutralization/ Wastewater treatment 7

Neutralization method Operating mode: § Mixing of acidic and alkaline wastewater § Addition of reagents (solutions of acids, quicklime Ca. O, Ca (OH)2, ammonia solution, etc. ) § Filtering of acid wastewater through neutralizing materials § absorption of acid gases by alkaline water The choice of neutralization method depends on the volume and concentration of wastewater, the availability and cost of reagents. Wastewater treatment 8

Oxidation method Operating principle: As a result of the chemical oxidation reaction, toxic contaminants become less toxic and are removed from the water. Operating mode: For purification of waste water, oxidizing agents such as gaseous and liquefied chlorine, chlorine dioxide, calcium chlorate, calcium and sodium hypochlorite, potassium permanganate, potassium dichromate, hydrogen peroxide, oxygen (technical or oxygen of air), ozone are used. Wastewater treatment 9
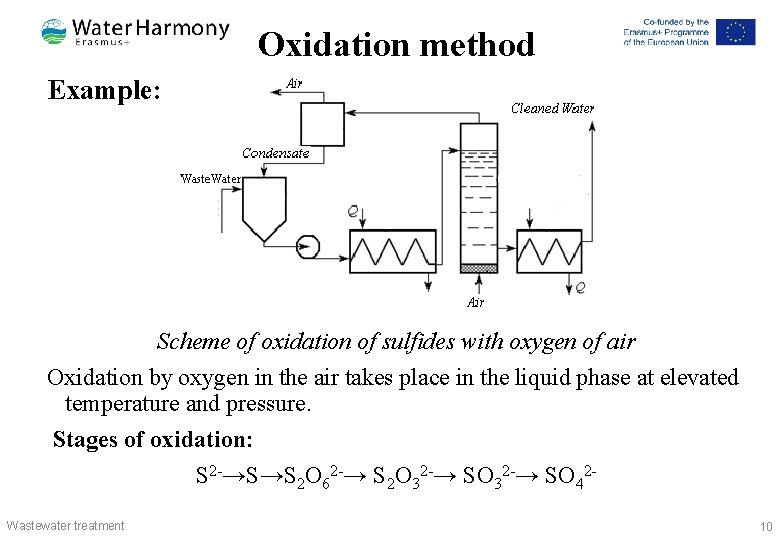
Oxidation method Example: Scheme of oxidation of sulfides with oxygen of air Oxidation by oxygen in the air takes place in the liquid phase at elevated temperature and pressure. Stages of oxidation: S 2 -→S→S 2 O 62 -→ S 2 O 32 -→ SO 42 - Wastewater treatment 10

Oxidation method Typical applications: For the neutralization of industrial wastewater, which contain: § toxic impurities (cyanides, complex copper and zinc cyanides) § chrome-containing effluent § compounds that are inexpedient to remove from sewage § compounds that are impractical to purify by other methods (hydrogen sulphide, sulfides) Wastewater treatment 11

Reduction method Operating principle: As a result of the chemical reaction of the reduction, toxic contaminants become less toxic, and the recovered substances are removed from the water by mechanical methods: filtration, flotation or sedimentation Operating mode: For the reduction reaction of inorganic compounds is used: Fe. S, Na. BH 4, Na. HSO 3, N 2 H 4, iron powder, aluminum powder, hydrazine, iron sulfate, hydrogen Wastewater treatment http: //vodakanazer. ru/kanalizaciya/xi micheskaya-ochistka-stochnyxvod. html 12

Reduction method Typical applications: The reduction method is used to remove mercury, chromium and arsenic compounds from wastewater. Example: solutions of sodium hydrosulfite for the reduction of substances containing Cr 6+: 4 H 2 Cr. O 4 + 6 Na. HSO 3 + 3 H 2 SO 4→ 2 Cr 2(SO 4)3 + 3 Na 2 SO 4 + 10 H 2 O Then Cr 3+ is precipitated with solutions of hydroxides Ca(OH)2, и Na. OH: Cr 3+ +3 OH-= Cr(OH)3↓ Example: A solution of hydrazine is used for the reduction of Cr 6+ at p. H = 7 -8: 4 K 2 Cr. O 4 + 3 N 2 H 4 → 4 Cr(OH)3↓ + 3 N 2↑ + 8 KOH Wastewater treatment 13

Precipitation of heavy metals Operating principle: Wastewater treatment from heavy metal ions is carried out by precipitation of heavy metals in the form of sparingly soluble compounds. The precipitation of metals occurs when various reagents are added: § hydroxides of calcium and sodium, § sodium carbonate, § sulfides of sodium, § various wastes, for example, ferrochrome slag. Wastewater treatment http: //srjcstaff. santarosa. edu/~oraola/j artest. html 14

Precipitation of heavy metals Operating mode: Precipitation of insoluble compounds is carried out in sedimentation tanks: § vertical with a descendingascending movement of water; § thin-layer shelf clarifiers http: //www. tecnoidea. it/eng/linea_acque. html Wastewater treatment 15

Precipitation of heavy metals Typical applications: Wastewater treatment of heavy industry and galvanic industries from heavy metal ions Example: Lead ions are precipitated as one of three hardly soluble compounds: Pb 2++OH-→Pb(OH)2↓ 2 Pb 2++2 OH- + CO→ (Pb. OH)2 CO 3↓ Pb 2+ + CO 32 -→ Pb. CO 3↓ p. H = 6. 0 corresponds to the beginning of the formation of lead hydroxide precipitate Wastewater treatment 16

Methods using reagents Benefits of methods neutralization, oxidation, reduction, precipitation: § A wide range of initial concentrations § Versatility § Easy operation § Conversion of toxic pollutants into less toxic or non-toxic forms. Disadvantages of methods neutralization, oxidation, reduction, precipitation: § High consumption of reagents § Additional wastewater pollution § Difficulty of removing heavy metals from the sludge for disposal § The need for significant sludge storage areas § Loss of valuable substances with precipitation Wastewater treatment 17

Ion-exchange cleaning Principle of operation: the process of exchange between ions that are in solution, and of ions that are on the surface porous resin beads. Operating mode: Initially, anions or cations are processed from wastewater on ion exchangers (sorption stage), and then the process of desorption of these ions from the resin (regeneration stage). Two ion-cleaning technologies are used: single-flow and counter-current Wastewater treatment http: //www. globalspec. com/learnmore/man ufacturing_process_equipment/filtration_se paration_products/ion_exchange_resins 18

Ion-exchange cleaning Typical applications: § Wastewater treatment from heavy metal ions: zinc, copper, chromium, nickel, lead, mercury, cadmium, vanadium, manganese § For extraction of valuable or toxic components from wastewater, and also neutralization of these waters § Purification of industrial wastewater from compounds of arsenic, phosphorus, as well as cyanide compounds and radioactive substances Wastewater treatment http: //www. unechabros. com/Ion%20 E xchange%20 Plants. html 19

Benefits: Ion-exchange cleaning § High cleaning efficiency § Obtaining, isolated from the waste water of metals, in the form of relatively pure and concentrated salts § The possibility of selective isolation of metals Disadvantages: § The need for preliminary treatment of waste water from oils, solvents, organic compounds § A large consumption of reagents for the regeneration of ion exchangers and treatment of resins § The need for preliminary separation of washing water from concentrates § Bulkiness of equipment, high cost of resins § The formation of secondary waste-eluents, which require additional processing Wastewater treatment 20
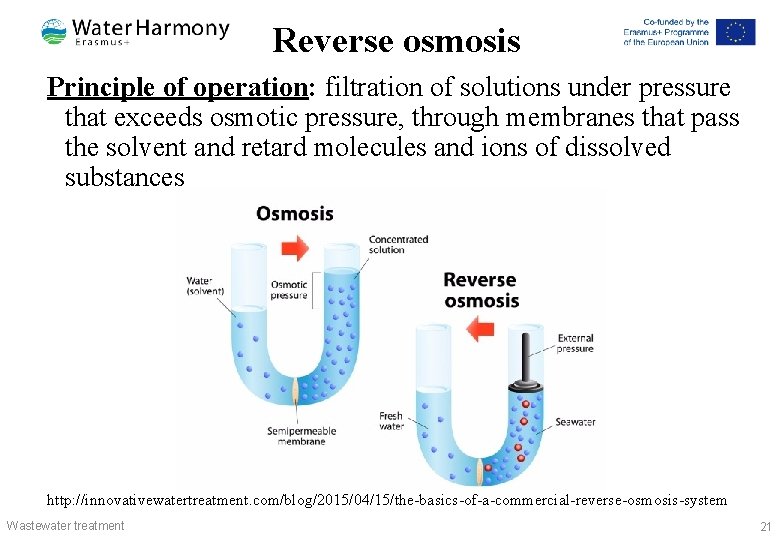
Reverse osmosis Principle of operation: filtration of solutions under pressure that exceeds osmotic pressure, through membranes that pass the solvent and retard molecules and ions of dissolved substances http: //innovativewatertreatment. com/blog/2015/04/15/the-basics-of-a-commercial-reverse-osmosis-system Wastewater treatment 21

Reverse osmosis Operating mode: Installation of reverse osmosis is able to remove particles from the water with a size of 1 -0, 1 nm, working pressure of 10200 bar. Typical applications: Desalination of treated wastewater: § in galvanic production § in the production of printed circuit boards http: //www. degremont-technologies. com/~degremon/Reverse -Osmosis-Skids-458 Wastewater treatment 22

Reverse osmosis Benefits: § High degree of purification § The possibility of utilization of heavy metals § Low energy costs § Simplicity and compactness of installations § Possibility of full automation of the process Disadvantages: § The need for preliminary treatment of waste water from oils, solvents, organic compounds § High cost of membranes § The complexity of operation, high requirements for the tightness of installations § Sensitivity of membranes to changes in the parameters of treated wastewater Wastewater treatment 23
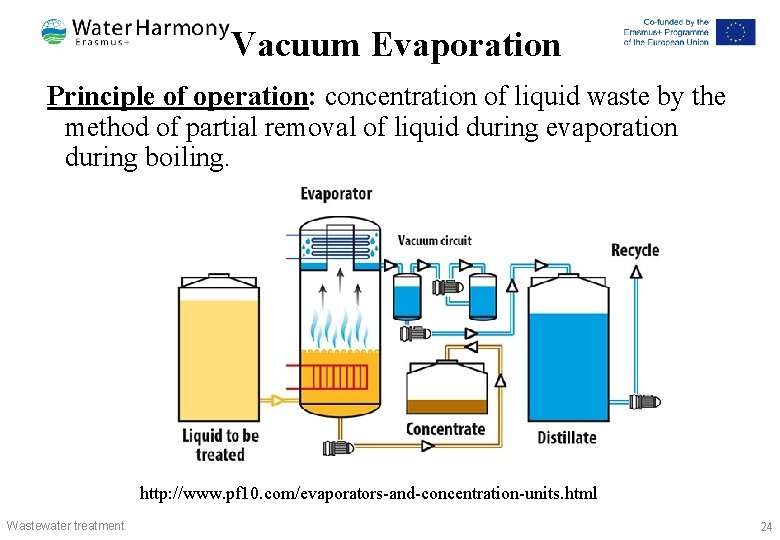
Vacuum Evaporation Principle of operation: concentration of liquid waste by the method of partial removal of liquid during evaporation during boiling. http: //www. pf 10. com/evaporators-and-concentration-units. html Wastewater treatment 24

Vacuum Evaporation Operating mode: Evaporation processes occur in a vacuum and the choice of pressure is determined by the properties of the waste water and the possibility of using the heat of the secondary steam. Typical applications: for wastewater treatment of heavy and chemical industry enterprises containing toxic compounds of heavy metals: copper, zinc, nickel, chromium, lead. Wastewater treatment http: //galvan. ru/? q=node/303 25

Vacuum Evaporation Benefits: § Simultaneous cleaning of various effluents with a high concentration of pollutants and containing aggressive liquids § High degree of purification § Return of salts and water to production § The possibility of organizing a closed cycle without discharges of harmful substances into the environment Disadvantages: § High energy intensity § High capital costs § The need for multi-stage wash tanks Wastewater treatment 26
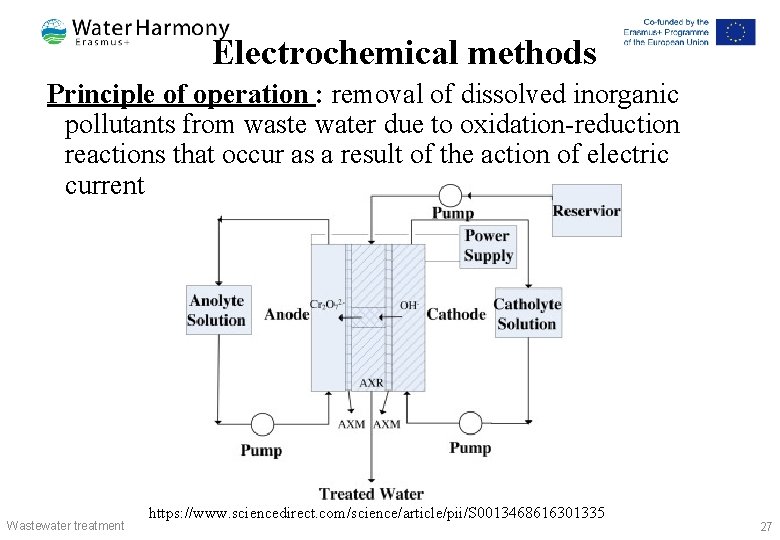
Electrochemical methods Principle of operation : removal of dissolved inorganic pollutants from waste water due to oxidation-reduction reactions that occur as a result of the action of electric current Wastewater treatment https: //www. sciencedirect. com/science/article/pii/S 0013468616301335 27

Electrochemical methods Operating mode: The process of electrochemical cleaning is carried out using soluble and insoluble electrodes: § Anodes made of graphite, magnetite, lead dioxide, manganese and ruthenium, which are applied to a titanium plate § Cathodes made of molybdenum, of an alloy of tungsten with iron or with nickel Main processes: § anodic oxidation § catholic reduction § electrocoagulation § electro flocculation § electro dialysis Wastewater treatment 28

Electrochemical methods Typical applications: § for wastewater treatment from dissolved impurities: cyanides, amines, sulfides, heavy metal ions § for wastewater treatment of galvanic plants § for the electrochemical extraction of copper and other non-ferrous metals, as well as precious metals from highly concentrated and slightly concentrated wastewater Wastewater treatment https: //wholesaler. alibaba. com/productdetail/Power-plant-electrocoagulationwastewater-treatment_60471907924. html 29
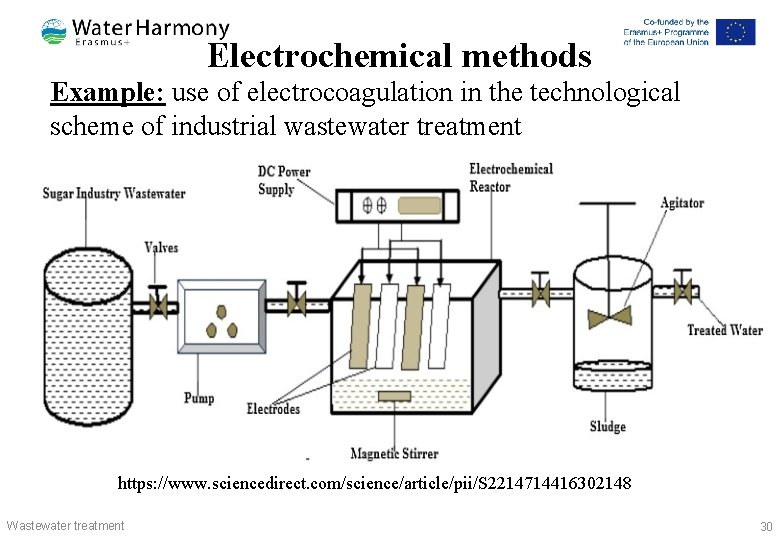
Electrochemical methods Example: use of electrocoagulation in the technological scheme of industrial wastewater treatment https: //www. sciencedirect. com/science/article/pii/S 2214714416302148 Wastewater treatment 30

Electrochemical methods Benefits: § Using a simple technological scheme of cleaning without the use of chemical reagents § Extraction of valuable metals from waste water § Easy operation and maintenance § High degree of purification Disadvantages: § High power consumption § Possibility of formation of collateral dangerous compounds Wastewater treatment 31

Methods for cleaning industrial wastewater from typical inorganic impurities Purification from heavy metal ions: electroflotation, sedimentation, electrocoagulation, electrodialysis, ultrafiltration, ion exchange. Purification from ions Chromium (III): electroflotation, ion exchange, precipitation, ultrafiltration, filtration. Purification from ions Chromium (VI): electroflotation, electrocoagulation, electrochemical reduction. Wastewater treatment 32

Methods for cleaning industrial wastewater from typical inorganic impurities Purification from cyanides: chemical oxidation, electroflotation, electrochemical oxidation. Purification from sulphates: sedimentation with reagents + filtration, vacuum evaporation, nanofiltration, reverse osmosis. Purification from chlorides: reverse osmosis, vacuum evaporation, electrodialysis. Cleaning from salts: nanofiltration, reverse osmosis, electrodialysis. Wastewater treatment 33

Reference literature § Physico-chemical methods of water treatment. Water resources management Edited by I. M. Astrelin and H. Ratnaweera 2015 § Selected publications from the Water Harmony Project: Water Research and Technology 2015, 323 pages Wastewater treatment 34
- Slides: 34