Warmup Write down your homework Be ready for

Warm-up • Write down your homework • Be ready for a quiz over volume.

Warm-up Calculate the volume of the following regularly shaped objects (using V = length x width x height). #1: A book has a length of 25 cm, a width of 10 cm, and a height of 5 cm. What is the volume? #2: What is the volume of a suitcase that has a length of 50 cm, a width of 10 cm, and a height of 30 cm? What are the units for the volume of a solid? Units for the volume of a liquid? TAKE OUT THE MATTER READING PASSAGE FROM YESTERDAY OR TAKE IT FROM THE FRONT TABLE IF YOU DON’T HAVE IT. Also take out your matter notes to finish.

Warm-up 1. What are the units for the volume of a solid? 2. Units for the volume of a liquid? 3. TAKE OUT THE MATTER READING PASSAGE FROM YESTERDAY OR TAKE IT FROM THE FRONT TABLE IF YOU DON’T HAVE IT. Also take out your matter notes to finish.

Answers… • • 15, 000 cm 3 • SOLID OBJECT: cm 3 • LIQUID: m. L 3 1, 250 cm

Warm-up SHARE YOUR PHYSICAL PROPERTY DESCRIPTION FROM THE HOMEWORK WITH YOUR TABLE PARTNER AND HAVE THEM GUESS WHAT IT IS (IF YOU DON’T HAVE A TABLE PARTNER SHARE WITH ANYONE IN CLASS).

Jump Start-physical properties *If you were asked to describe a piece of fruit to a person who has never seen this type of fruit before, what would you tell the person? *Be descriptive in your language. *Please DO NOT tell your classmates which fruit you have chosen to describe, because you will be sharing your response later. *Use both qualitative and quantitative data. *Any volunteers want to read their fruit description to the class? (Please do not tell us what your fruit is before, we want to guess).

Secret Sack • PROCEDURE: – Each Lab Group will receive a sealed paper sack containing a mystery object. Do not open the sack! (unless it says you can) – For about 1 minute, make as many observations about the object as you can without opening the sack. You may touch, shake, or listen to the object through the sack. Record your observations. Be gentle!!!!

Secret Sack Continued DO NOT LOOK IN THE BAGs WE WILL DO IT TOGETHER AT THE END. • ANALYSIS in your notebook: – 1. At the end of 1 minute, discuss your findings with your partners. – 2. List the object’s properties that you can identify. – 3. Make another list of properties that you cannot identify. Then make a conclusion about the objects identity. – 4. Share your observations, your list of properties, and your conclusion with the class. Then, open the sack. – 5. Did you properly identify the object? If so, how? If not, why? Record your answers. ```

• • Bag #1 - Orange Bag #2 - Apple Bag #3 - Batteries Bag #4 -Grapes Bag #5 - Nails and screws Bag #6 -cotton balls Bag #7 -Beads

Warm-up Write out and answer the following questions in your Science notebook – Which of the following are examples of matter? • A. Dust C. Strand of hair • B. The moon D. All of the above – A graduated cylinder is used to measure • A. Volume C. Mass • B. Weight D. Inertia – The volume of a solid is measured in • A. Liters C. Cubic Centimeters • B. Grams D. All of the above – Mass is measured in • A. Liters B. Newtons C. Kilograms

Warm-up • Prepare for today, homework: • What does it mean to do a word wall picture? Term, definition and a visual example. • Predict whether a golf ball and a ping pong ball will float in water. • What does whether or not each ball floats have to do with density?

Warm-up • List the differences between 2 cans- Coke and diet coke. • How are they similar? • Predict whether they will float or sink in water.
Warm-up Per. 2 • Sign out a lap top and log on • List any questions you still have about matter, volume, mass, and density. • Next list the 4 states of matter.

Reminders for today… • Before you start anything… • Go to my weebly and read all information about the baby book project • If needed refer to the example Lithium PPT (you can NOT use Lithium in your book). • Remember to cite your sources and be sure to consider quality and neatness when creating your book.

Warm-up • What is solubility? • Take out your project rubric • Take the same laptop you had yesterday and continue working on your project.

Warm-up The density of water is 1 g/cm 3. Which of the following is correct? A. An object will float if the density is less than 1. B. An object will sink if the density is less than 1. Using the density of water, how can you tell if an object will float or sink?

Warm-up • With your partner(s), finish calculating the density of at least 7 of the cubes and complete questions from the density lab.

Warm-up • What is a solution? Predict what is a solute, and a solvent? (use your vocab. handout) • If you did not turn in your chem/phys. change handout put it in the tray for your class. • You will have a graded exit slip at the end of class.

Introduction to Matter Physical Properties: Density Solubility States of matter

Objectives • Analyze the physical properties of matter by discovering how physical properties such as density can help identify objects. • Identify and compare the physical properties of matter to include density, melting/boiling points, states of matter, and solubility. • Explain what happens to matter during a physical change.

Essential questions • How are the physical properties of matter used to compare pure substances? • What is density? • What are the 4 states of matter? • What is solubility?

• Something that can be observed • What are or measured Physical without Properties? changing the matter’s identity.

Name some physical properties of this apple

What are some specific Physical properties? Thermal conductivity- how well a substance transfers heat Phase of matter- solid, liquid or gas Density- how much mass is in a given space Density=mass/volume or D=m/v Solubility- how well a substance dissolves in H 2 O

Thermal conductivity
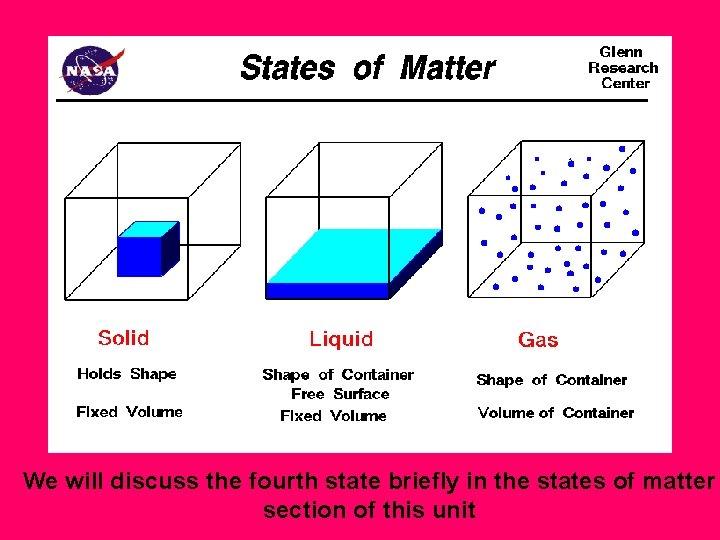
We will discuss the fourth state briefly in the states of matter section of this unit
Solubility

Solubility • COPY THESE DEFINITIONS UNDER THE QUESTION ON THE LEFT SIDE OF YOUR NOTES. • Solution= Solvent + Solute • Solvent: The liquid part of a solution • Solute: The solid/dry that is mixed and dissolved in the solution

Solubility Quick Lab (in your notebook) Materials: SOLUTES: Instant Coffee, Sugar, SOLVENT: water, 2 clear beakers, & spoons. Look at the two solids and hypothesize whether you think each one is soluble in water. Use you spoon to put 1 heaping spoonful coffee into 1 beaker and use the other spoon to put 1 heaping spoonful of sugar into the OTHER beaker. Mix thoroughly and record your results. Which mixture created a SOLUTION? Which one did not?

Ductility Ability to shape into wires

Malleability Ability to mold into flat sheets and Shapes.
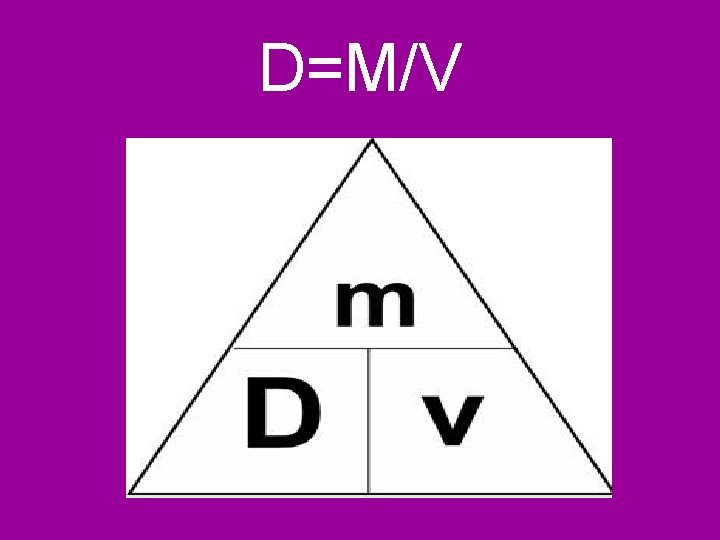
D=M/V

• In liquids, densest liquid sinks to the How will bottom, least dense density floats on top. help figure • Solids can be checked against out what a water’s density substance is? (1 g/cm 3), if solid floats it is less than 1 g/cm 3, if it sinks it is more than 1 g/cm 3

Density Demonstration What is the difference between these two golf balls in the water? mass, volume and density video density calculations (only play until 1: 36)

Density QUICK LAB – (1) Material Getter: • Graduated cylinder with 50 -60 m. L of Water, cup of marbles, & triple beam balance. • (2) Lab Initiator: • Drop one marble into the water and observe the displacement • Subtract to find the volume in cm 3 – (3) Data Collector: • Mass the marble on the balance and record the mass (in grams). – (4) Discussion Leader: • Predict: Will the marble density come out the same if we follow the same steps with 2 -8 marbles? • How do we represent the units for density, why? • What does the fraction bar mean?

A. What you need 1. Mass=m grams 2. Volume=V cm 3, m. L, m 3, L How do you calculate Density? (the broken B. Formula m heart D= V equation) D=
![Density Demonstration: Coke vs Diet Coke • Coke-dietcokegr 6_sci_26_demo[1]. pdf • Demo with soda’s Density Demonstration: Coke vs Diet Coke • Coke-dietcokegr 6_sci_26_demo[1]. pdf • Demo with soda’s](http://slidetodoc.com/presentation_image_h/2148f4b56caede7d4bafd411b82883e3/image-37.jpg)
Density Demonstration: Coke vs Diet Coke • Coke-dietcokegr 6_sci_26_demo[1]. pdf • Demo with soda’s in fish tank. • List the differences between the 2 cans • Predict what the cans will do in the tank. • Students will calculate the density in notebooks/binders.

Post lab • Why does one float? • How is this related to density? • Look at the difference between the amount of sugar and aspartame (Nutra. Sweet) • D=m/v • Compare these densities to the density of water-1 gm per cm 3

Making Sense of Density Quick Lab (1)Does the density of a material vary with volume? (2)Material: • 1 crayon with paper removed, paper towel, balance graduated cylinder, approximately 50 m. L of water. (3) Procedures: • Create a small chart to record your data Item Whole Crayon Piece 1 Crayon Piece 2 Mass (g) Volume (cm 3 ) Density

Making Sense of Density Quick Lab • Use a balance to find the mass of the whole crayon • Add enough water to a graduated to cylinder to complete submerge the crayon • Use water displacement to measure the volume of the crayon. Record your results in the table. • Use the density formula to calculate the density. • Break the crayon into two pieces. • Repeat procedures for the other two crayon pieces. • Record your results in the table.

Data Analysis and Conclusion 1. For the object you tested, compare the density of the whole object with the densities of the pieces. 2. Use your results to explain how density can be used to identify a material.

Journal Activity In your journal complete the following activity Today I learned about__________ With my class. The first thing we learned was ______. Next, ___________. Then, ___________________. I also learned that _________. Next time we study ________________________, I want to learn more about _______.

What are physical changes? • When matter is changed from one form to another without changing its identity. • Example: water • Water to ice, water to steam, steam to water

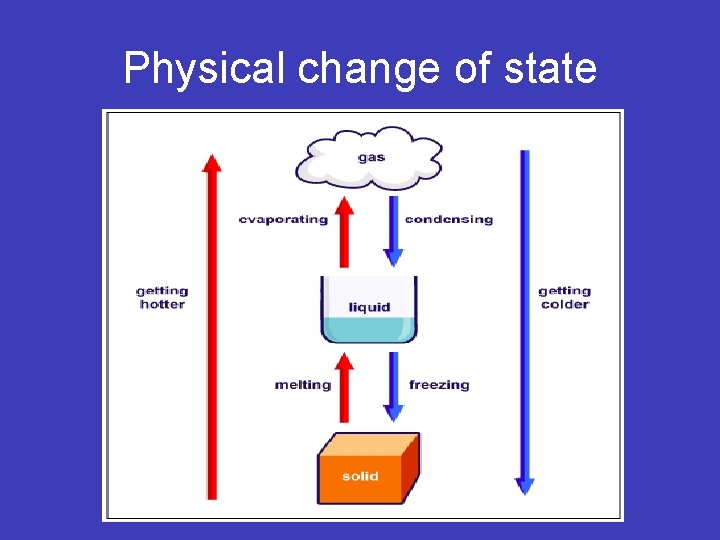
Physical change of state


• More Physical change examples: • Shape changes, like crushing a can, melting a popsicle, dissolving kool-aid, melting butter. These have changed shape, but are the same type of matter.

Journal Activity • In your journal complete the following. • Summarize today’s lesson in one or two sentences.

Objectives Review • Identify and compare the physical properties of matter to include density, melting/boiling points, states of matter, and solubility. • Density and melting/boiling point are the most important physical properties in identifying matter. • Explain what happens to matter during a physical change. – A substance undergoes a physical change, but its identity stays the same. Also…List the six examples of physical changes. – Breaking, melting, freezing, cutting, crushing, and dissolving.
WORD WALL Term Define • Physical Property • Physical Change • Mass • Matter • Malleability • Thermal conductility Picture It Density Solubility Volume Phase of matter Ductility
- Slides: 50