WARMUP Read the objectives and essential questions Can

WARM-UP § Read the objectives and essential questions. Can you answer the question? § DESCRIBE what must happen in order for a change of state to occur.

Warm up April 29, 2019 • Be ready for class seated, quiet. • Copy & answer the following questions in your note book: • What elements make up the compound table sugar C 12 H 22 O 11? • How many of each type of element can be found in the compound table sugar?


Vocabulary review… The temperature at which a solid turns into a liquid is known as its… ____. The temperature at which a liquid boils is known as its…_______. The ability of one substance to dissolve into another is known as its…______.
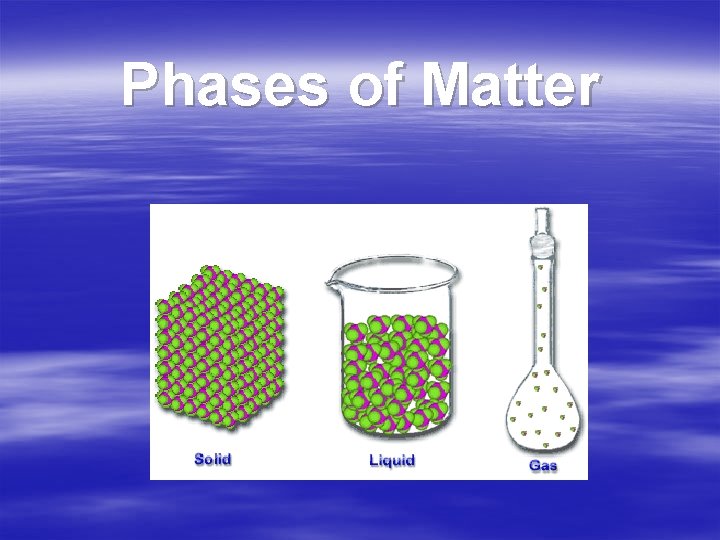
Phases of Matter

Objectives § Explain the effect of heat on the motion atoms § Describe what happens to particles (atoms) during a phase change

Essential Questions § How does heat affect the motion of atoms? § How do atoms react during a change of phase?

Frontloading quiz Shape and volume of matter in the ______ state do not change. The particles of a _____ are very far apart and move independently of one another. A liquid’s atoms have more energy and _______ than a solid. Word bank: movement, solid, gas

Warm-up § Listen to the states of matter song… § What does the song tell you about the properties of a Gas, a liquid, and a solid? song

Warm-up § Describe how atoms behave as far as movement, structure, and energy in each phase of matter (solid, liquid, and gas).

Warm-up § Draw a picture of how atoms “look” as far as movement, attraction, and energy in each phase of matter (solid, liquid, and gas). Hint: draw each one in a container or a jar.

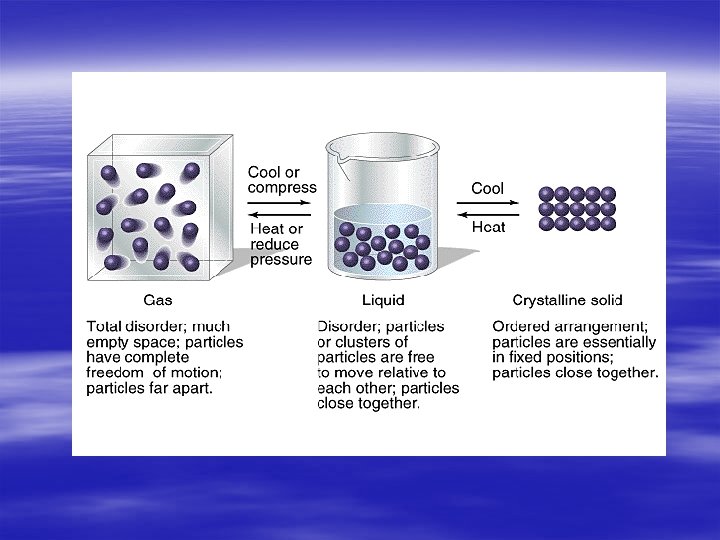
§ Physical forms in What are which substances exist phases § Solid, Liquid, and of matter? Gas states video clip § All matter is made of particles atoms and molecules


§ Anything that has a definite shape and volume § Particles are very close together § Particles have a very strong attraction to § https: //phet. colora each other do. edu/en/simulati § Particles vibrate in on/states-of-matter place § Particles have low energy § What are solids?


What is a liquid? § substance that has a definite volume but not a definite shape § Particles are not very close together slide past each other § Particles have a semi – attraction to each other § Particles move around more than solids § Particles have medium energy

Liquid characteristics § Surface Tension - a force that causes particles on the surface to bond tighter - causes droplets § Viscosity - resistance or non - resistance to flow § High viscosity - flows slowly § Low viscosity - flows freely

Surface Tension
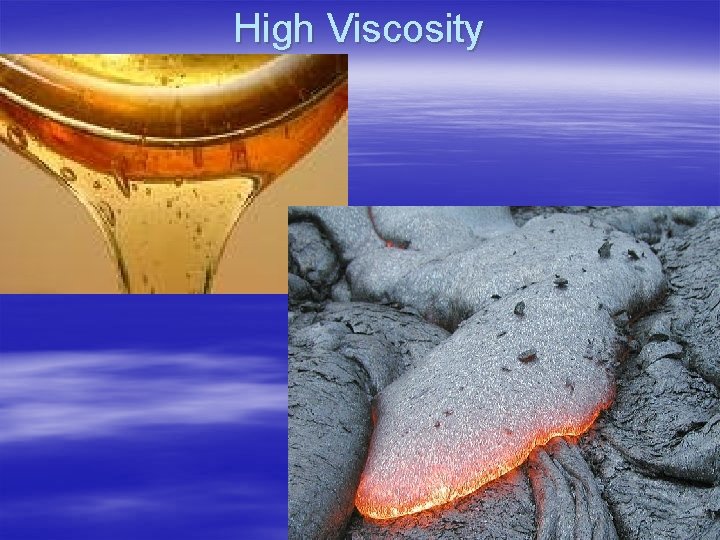
High Viscosity

Liquid Trivia § Water is the only substance that can be found as a solid, liquid, and a gas at normal surface temperatures and pressures on Earth.

§ Gases have no definite shape What are and no definite volume gases? § Gases can be https: //phet. colorado. edu/en/ compressed or simulation/legacy/gasspread way apart properties § Particles move very fast § Particles have a lot of energy

Gases released from a volcano
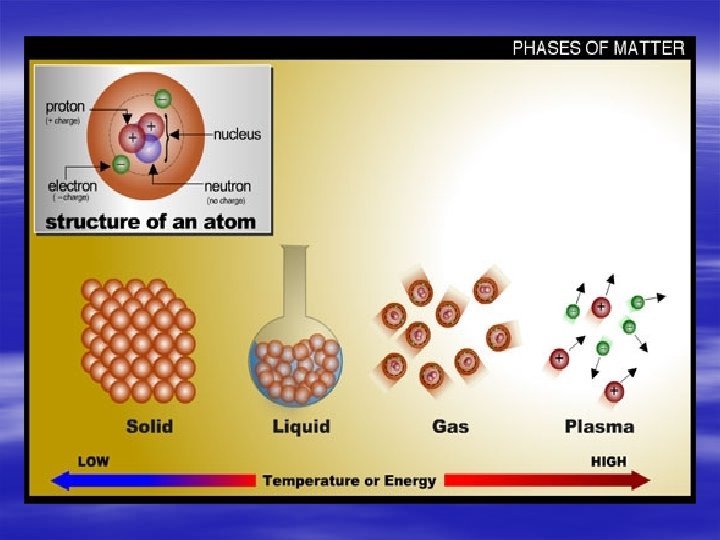

Plasma has been called the "fourth state" of matter, after solids, liquids and gases. Most of the matter in the universe is in the form of plasma. A plasma is formed if some of the negatively charged electrons are separated from their host atoms in a gas, leaving the atoms with a positive charge. The negatively charged electrons, and the positively charged atoms (known as positive ions) are then free to move separately under the influence of an applied voltage or magnetic field.

Stars are plasma

Phase Changes

Warm-up Describe what must happen in order for a change in state to occur. (think about the Bill Nye video). states of matter & liquid nitrogen

What are phase changes? § a physical change from one physical form to another From solid to liquid to gas & vice versa Requires adding or Removing energy
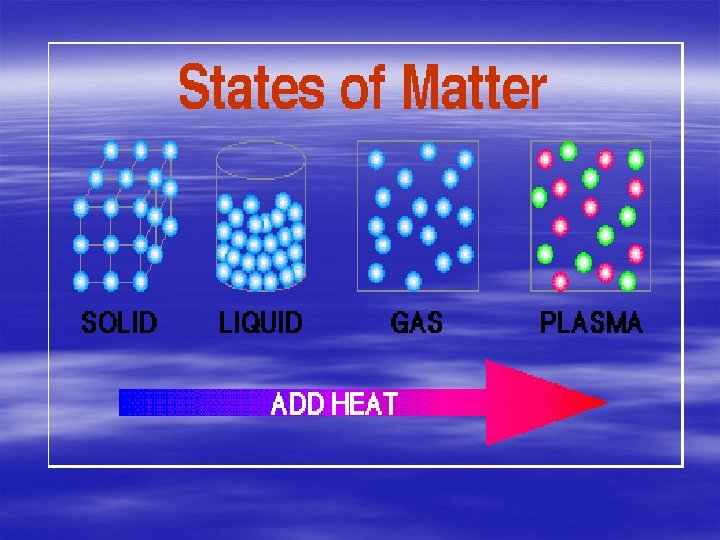

What are phase changes? 4 states of matter song § Melting Change of state from solid to liquid § Melting point/freezing point is the temperature that a substance either changes to a liquid or a solid.

What are phase changes? § Freezing- Change of state from liquid to solid § Melting and freezing temp for water is 0 degrees Celsius

What are phase changes? Evaporation- is a change of a Substance on its surface from a liquid to a gas

What are phase changes? § Boiling change of a liquid to a vapor. § boiling point is the temp. at which liquid turns to gas § Discovery ed boiling point

What are phase changes? Disc. ed. phase changes § Condensation change of a gas to a liquid § Condensation and evaporation/boiling point are the same temp. for a substance § When a gas like water condenses it takes up less space because the atoms slow down.

Can Crusher Demonstration § Hypothesize how I will crush the can. § From what you know about how atoms are effected by temperature why do you think the can was crushed?

§ When the can is put in the cold water upside down, the hot gas water molecules are very rapidly cooled. Some of the gas molecules are condensed back to the liquid water, so there are less gas molecules present. Cold water also cools the remaining gas molecules, which makes the molecules slow down, then there are fewer collisions with the walls of the can, which in turn causes less pressure inside of the can. The air pressure on the outside of the can is stronger. This causes the can to crush.

What are phase changes? § Sublimation change of a solid directly to a gas § Deposition – change of state from a gas directly to a solid

Condensation

Sublimation

This section is not part of the curriculum meant for extension only Behavior of Gases

Geologists sampling gases at the dome of a volcano- (90% of the gas is water vapor).

What are three things that affect the behavior of a gas? § 1. Temperature is a measure of how fast particles are moving. the faster they move the more energy (higher temp. ) they have and the farther they go, slower=less energy, lower temp


What are three things that affect the behavior of a gas? 2. Volume § it is the amount of space that an object takes up. § With a gas, volume depends on the container it is in. § Can be compressed

What are three things that affect the behavior of a gas? 3. Pressureamount of force exerted on a given area of space.

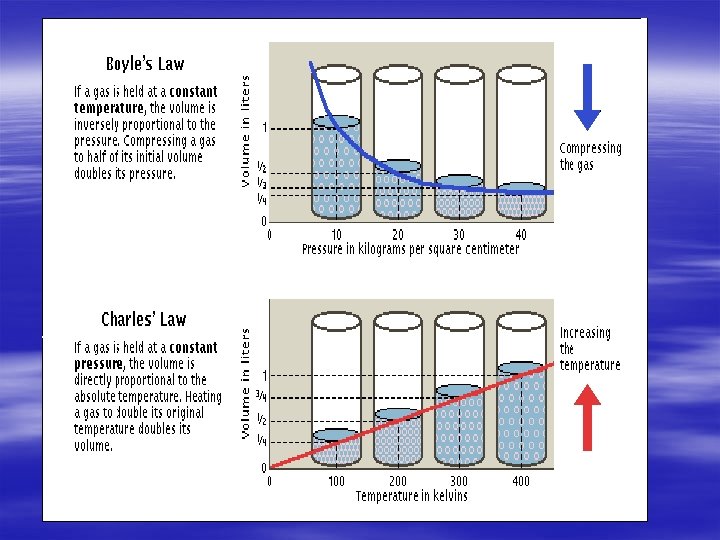
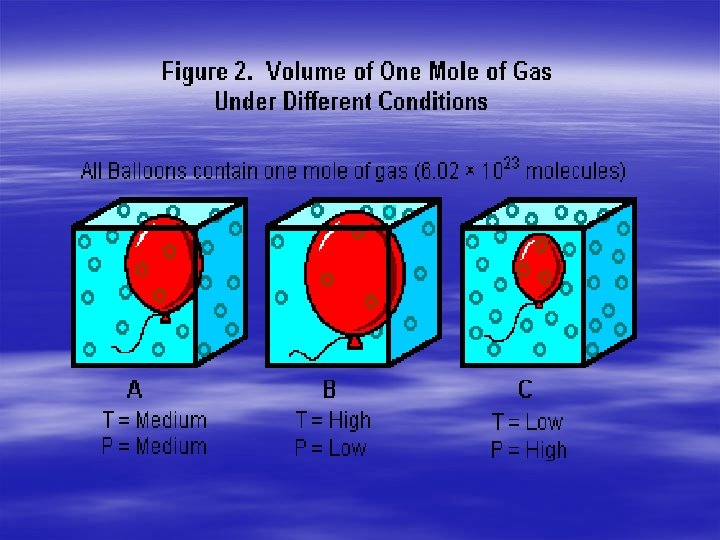
§ Charles law the What are the Gas Laws? volume of a gas is directly proportional to the temperature of a gas when the pressure is constant. § -as the temperature of a fixed amount of gas increases the volume increases.

What are the Gas Laws? § Boyles law the volume of a gas is inversely proportional to the pressure of the gas when the temperature is constant. -when the volume for a fixed amount of gas increases the pressure decreases
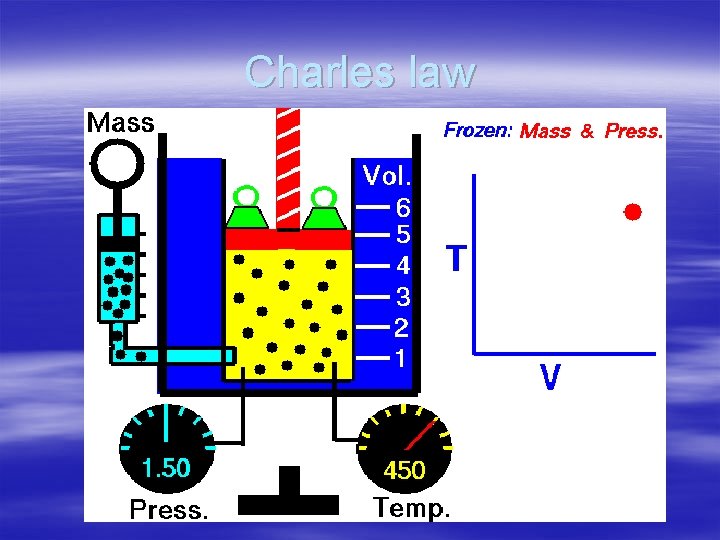
Charles law
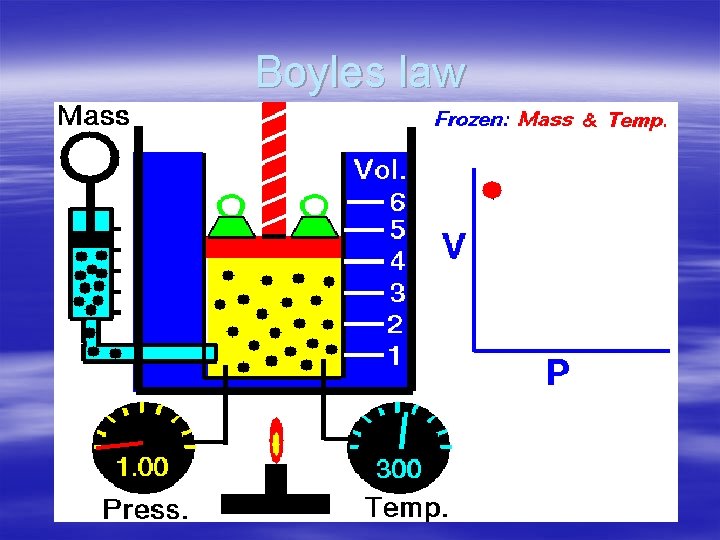
Boyles law

Charles law
- Slides: 53