Warmup February Tuesday Please get out your notes

Warm-up February Tuesday Please get out your notes and lab paper. Read the lab Background section for part II and write down two questions you have about the lab.

Bellwork February Wednesday : Smarties! n Table 1: Just put smartie in your mouth Table 2: Swirl smartie, but don’t chew Table 3: Chew smartie, then let dissolve n Questions n n q q What are three things that can speed up the dissolution of the smartie? Will temperature always affect how substances dissolve? Why or why not?

Chemistry I Chapter 16 - Solution Formation

16. 1 properties of solutions Factors affecting rate of n Agitation solution: q q brings fresh solvent in contact w/ solute does not affect amount the amount of substance that dissolves

Factors affecting rate of solution: n Temperature q heat increases rate of solution by increasing kinetic energy and motion of particles
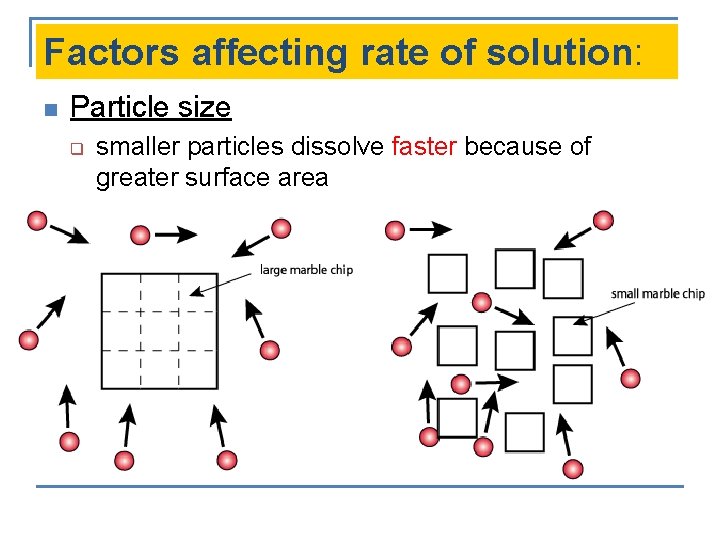
Factors affecting rate of solution: n Particle size q smaller particles dissolve faster because of greater surface area

Question n What would dissolve faster? q q A cube of sugar Granulated sugar Powdered sugar Why?
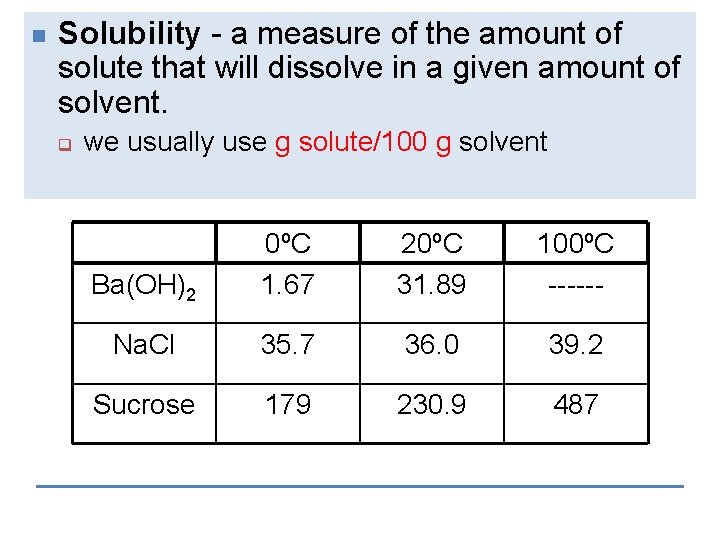
n Solubility - a measure of the amount of solute that will dissolve in a given amount of solvent. q we usually use g solute/100 g solvent Ba(OH)2 0ºC 1. 67 20ºC 31. 89 100ºC ------ Na. Cl 35. 7 36. 0 39. 2 Sucrose 179 230. 9 487

Factors affecting solubility: n The nature of the solute and the solvent q “like dissolves like”
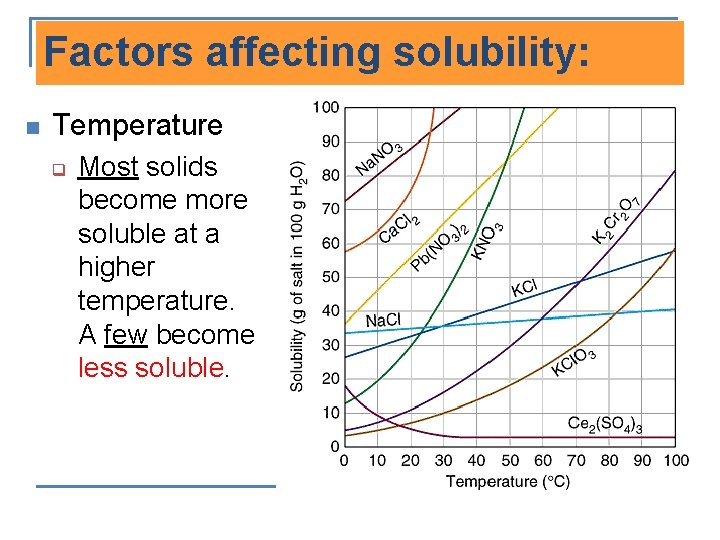
Factors affecting solubility: n Temperature q Most solids become more soluble at a higher temperature. A few become less soluble.
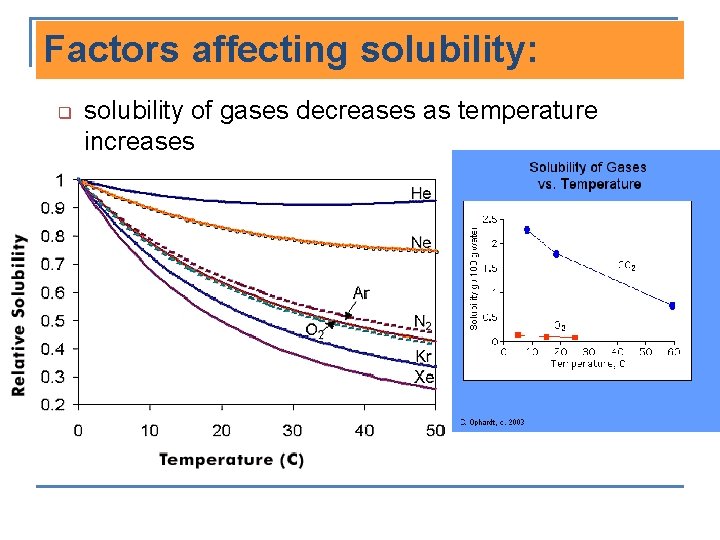
Factors affecting solubility: q solubility of gases decreases as temperature increases
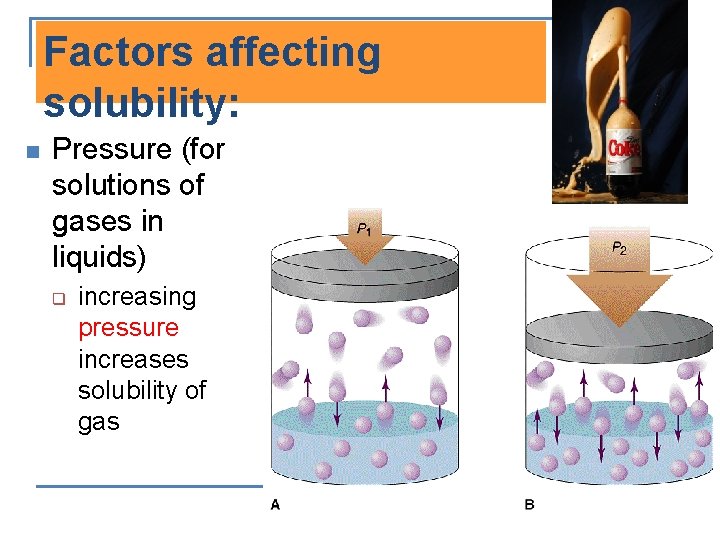
Factors affecting solubility: n Pressure (for solutions of gases in liquids) q increasing pressure increases solubility of gas

Question n Pressurized CO 2 is pumped into three cans of soda. The first can is at 10ºC, the second is at 20ºC and the third is at 50ºC. In which can will the most gas dissolve and why?

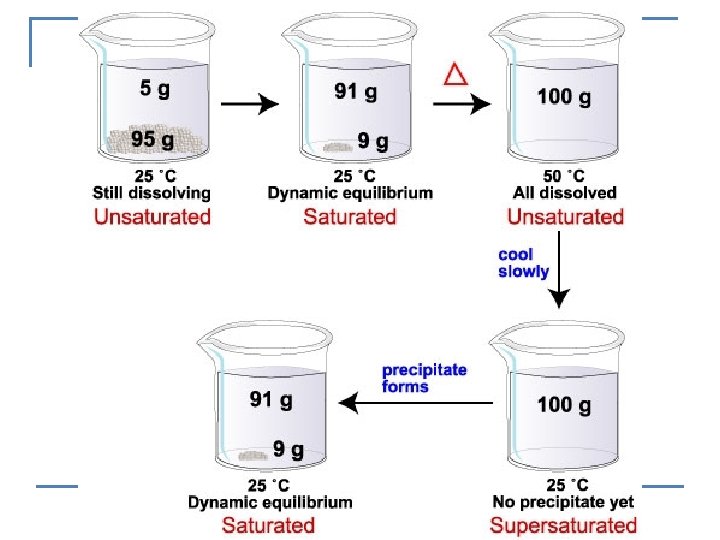
n Saturated solution -contains the maximum amount of solute that will dissolve in a given amount of solvent at a given temperature. n Unsaturated solution- more solute could be dissolved -indicated by no solid present n Supersaturated solution- more solute is dissolved than is theoretically possible at a given temperature.
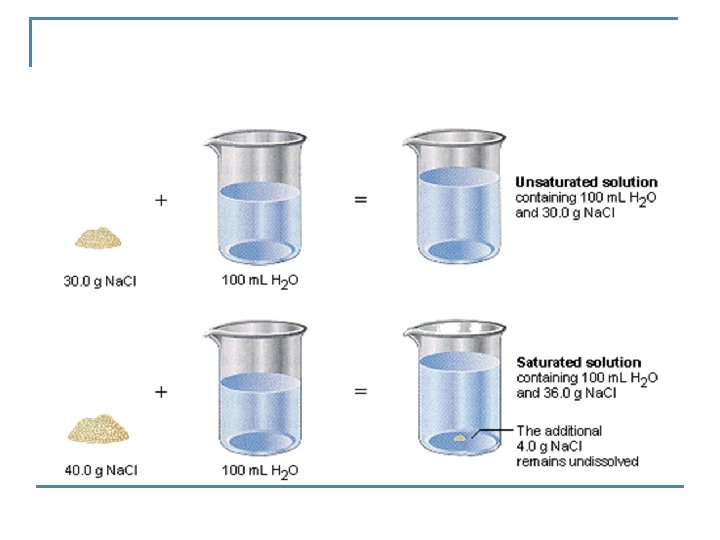

EXAMPLE: n How could you determine if a solution is saturated, unsaturated or supersaturated? q q If solid is present – it is Saturated If no solid is present – it is unsaturated or supersaturated n n n To determine which, add a crystal Supersaturated will crystallize Unsaturated will dissolve the crystal DEMO
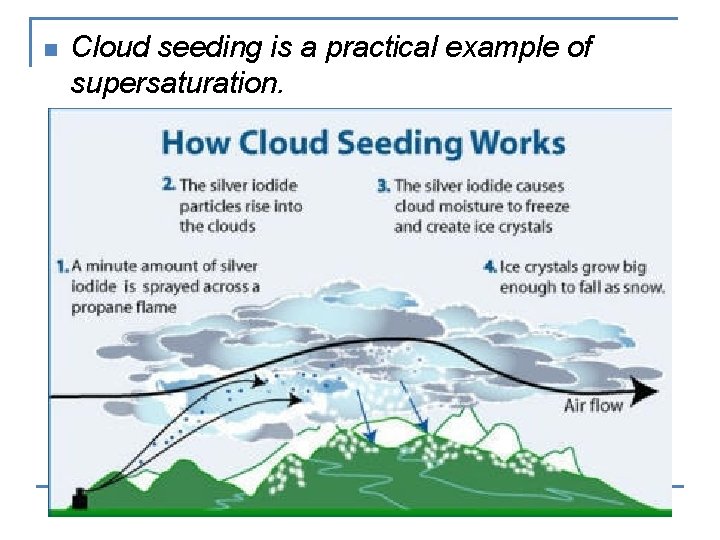
n Cloud seeding is a practical example of supersaturation.
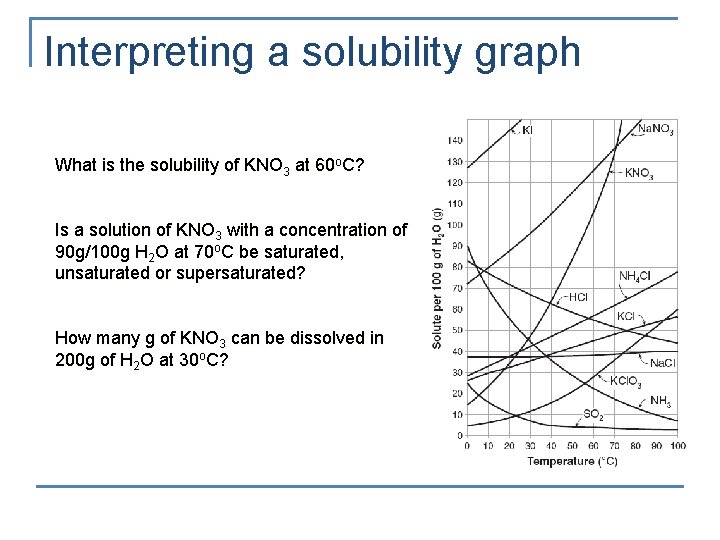
Interpreting a solubility graph What is the solubility of KNO 3 at 60 o. C? Is a solution of KNO 3 with a concentration of 90 g/100 g H 2 O at 70 o. C be saturated, unsaturated or supersaturated? How many g of KNO 3 can be dissolved in 200 g of H 2 O at 30 o. C?

n Miscible - liquids that are soluble in each other q n Ex. ethanol and water Immiscible- liquids that are not soluble in each other q Ex. oil and water

Question n Which of the following would be miscible in a polar solvent? q q q q HCl Na. OH Gasoline Mg. Br 2 Sugar Olive oil Li. Cl
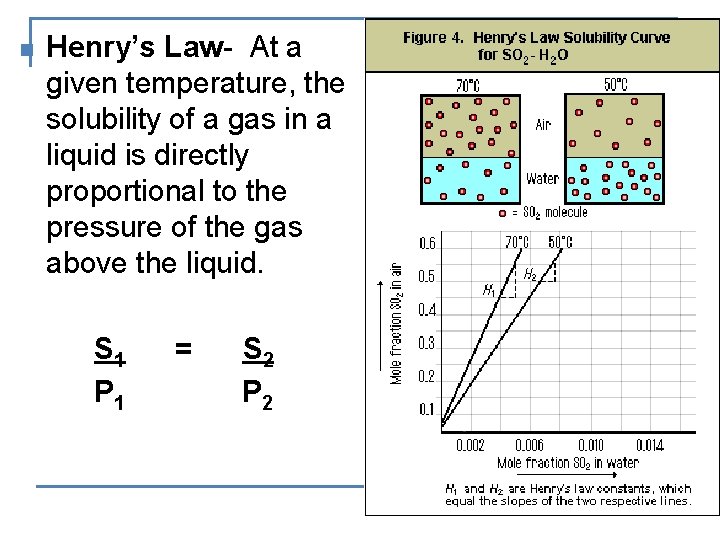
n Henry’s Law- At a given temperature, the solubility of a gas in a liquid is directly proportional to the pressure of the gas above the liquid. S 1 P 1 = S 2 P 2

EXAMPLE: At 25 o. C, the solubility of a certain gas in H 2 O is 0. 75 g/L of H 2 O at a pressure of 1. 0 atm. What is the solubility of the same gas at 10. 0 atm pressure? S 1 S 2 = S 1 = 0. 75 g/L P 1 P 2 P 1 = 1. 0 atm S 2 = ? 0. 75 x x = 7. 5 g/L = P 2 = 10. 0 atm 1. 0 10. 0 n
- Slides: 23