WarmUp Drops On A Penny Complete the minilab

Warm-Up: Drops On A Penny Complete the mini-lab on your desk. Write your answers on the paper provided and glue sideways into your composition book.

Biology Notes Biology & Biochemistry Part 3 Pages 40 -43

Standard 2. 1 Discuss the properties of water that ____ contribute to Earth’s suitability as an environment for life: cohesive behavior, expansion upon freezing, and versatility as a solvent Essential Question How do the unique properties of water allow life to exist on Earth?

I. Water is a molecule which means its atoms share _____ electrons • This makes water a _______ covalent molecule • In water, there is one atom of ______ oxygen and hydrogen two atoms of _____

Remember…. . Compounds are made of atoms. Water has 3 atoms total. This makes water a compound. We know that the atoms share electrons but WHY? ? ? ? Compounds are formed so that atoms can get full outer energy shells What is a full outer energy shell? 2 electrons on the first shell 8 electrons on the second shell

• Although the electrons in water are _____, they are not shared _____ shared equally between the oxygen atom and the ____ 2 hydrogen atoms vpolar molecule = a group of atoms that _____ electrons unequally resulting share in charges on the atoms Øwater is a _______ polar covalent molecule

• Due to it’s polarity, (unequal sharing of electrons) the oxygen atom in water has a negative charge and each of the small ____ hydrogen atoms has a small _____ positive charge
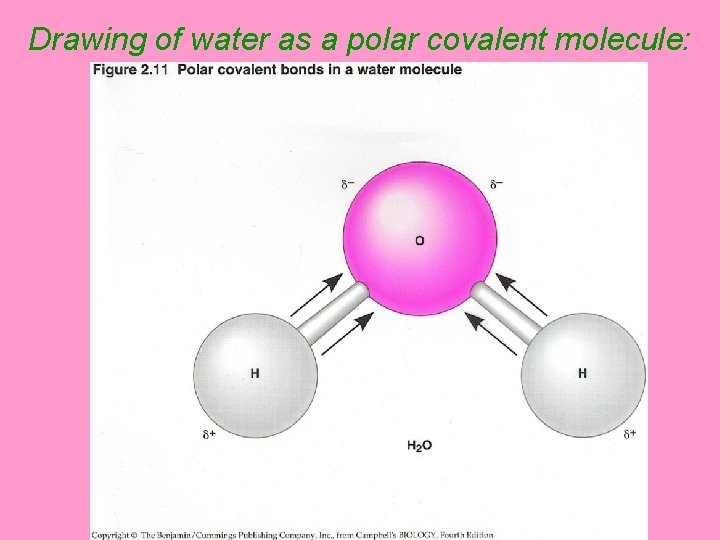
Drawing of water as a polar covalent molecule:

• The charges on a water _____ molecule allow bond it to be attracted to and ____ with other water molecules v. Hydrogen Bond = the bond that forms between hydrogen and another ____ polar covalent molecule Øhydrogen bonds are considered to be weak and not nearly as strong very ______ covalent or ionic bonds as _____
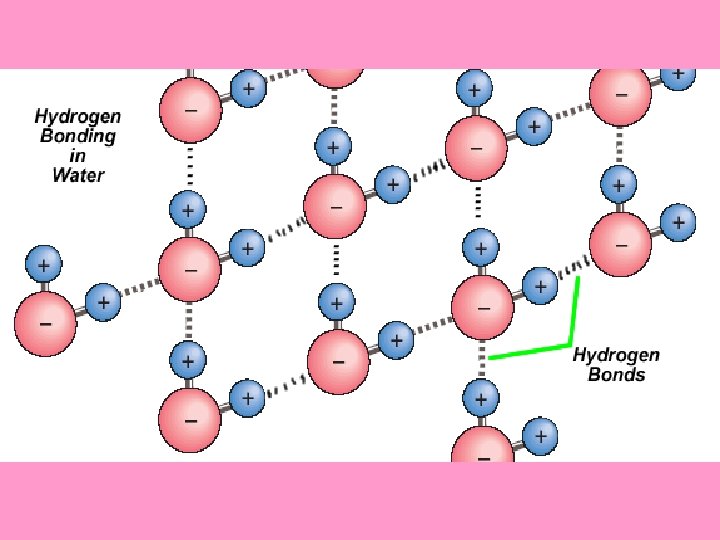

polarity and the resulting • Water’s _____ hydrogen bonds that water can form _____ with itself as well as other _______ polar covalent molecules allows water to have very special properties _____ v. These properties explain why water is life on _______ Earth important to _______

II. The Properties Of Water A. Water is cohesive • Cohesion = an attraction between molecules of the same substance ______ • water is attracted and ______ bonds to itself with weak _____ hydrogen bonds e. g. water droplets, dew

B. Water has surface tension • Surface Tension = in bodies of water, the inward _____ pull of water molecules caused by cohesion _____ v surface tension is the reason why certain bugs can ____ walk on water
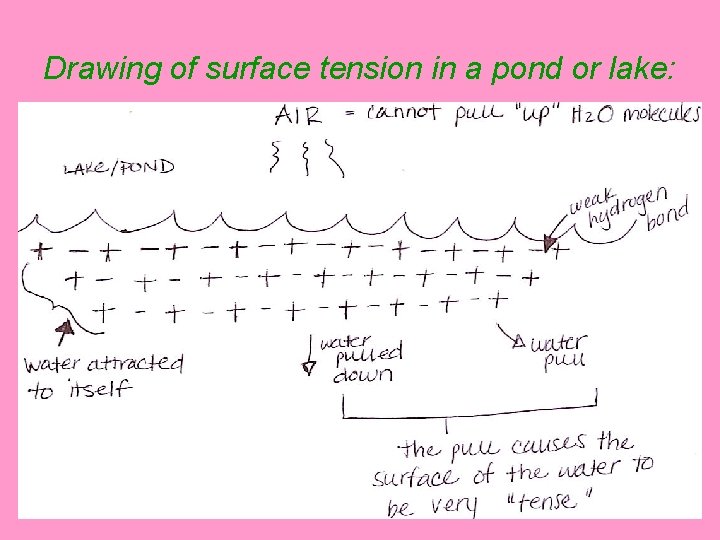
Drawing of surface tension in a pond or lake:

Think about it…… Why do oceans not exhibit surface tension? Imagine we are all form a circle and have a sheet pulled tight on the edge. Still holding on we lean back. What happens if one person lets go? Oceans do not exhibit surface tension because of currents, wind, and objects that break the hydrogen bonds between the water molecules.

C. Water is adhesive • Adhesion = an attraction between molecules of _____ different substances v water is attracted to other ______ polar molecules

Drawing of water in a glass showing adhesion:

D. Water can move by capillary action • Capillary Action = the ability of water to up thin tubes even defying move ___ gravity _____ v capillary action is due to ______ adhesion e. g. xylem in trees, paper towels

E. Water is a universal solvent • Solution = a mixture of two or more substances in which molecules are ______ evenly distributed _____ i. e. sugar water / salt water • Solvent = the substance in which the solute dissolves _____ polarity solutes that v. Due to water’s _____, are also ____ are attracted to water polar and tend to break apart and become water surrounded by the _____ e. g. see page 42
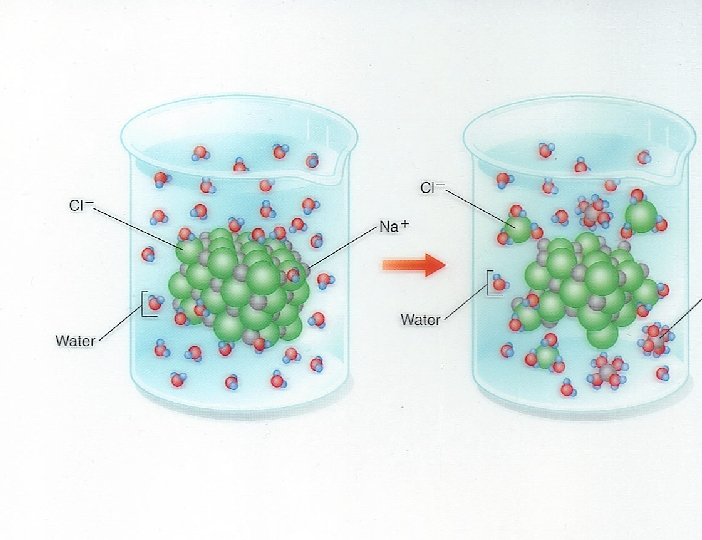

• In the body, water is valuable as a solvent vwater dissolves ( _____ ) breaks apart the foods we eat

Going back to oceans…. Another reason why oceans do not display surface tension is because of the salt…. WHY? Water is attracted to the salt more than itself so it’s water is less cohesive to itself. Without cohesion, there can be no surface tension.

F. Water has a high heat of vaporization • Heat of vaporization = the amount of heat it takes for a substance to _____ evaporate ______ vit takes a large amount of heat to break the hydrogen bonds _______ between water molecules

v this is valuable in the human body in the sweat form of _______ Ø when we exercise, heat accumulates body in the _______ evaporate the Øthe heat is used to ______ water in the body homeostasis is Øsweat results and _______ maintained for body temperature

III. Acids, Bases & p. H In some instances, when water is used to dissolve substances, some of the water molecules will break down to form ions (ion = an atom/compound that has gained or lost an electron forming an electrical charge on the atom/compound)
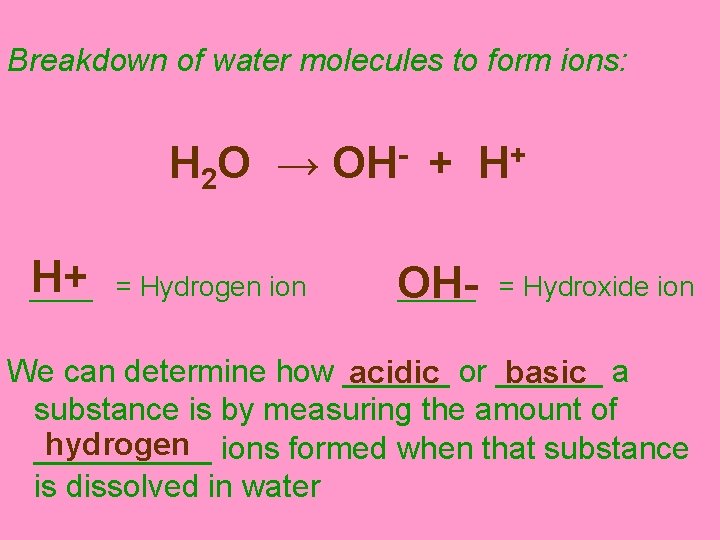
Breakdown of water molecules to form ions: H 2 O → OH- + H+ H+ = Hydrogen ion _____ OH- = Hydroxide ion We can determine how ______ acidic or ______ basic a substance is by measuring the amount of hydrogen ions formed when that substance _____ is dissolved in water

• p. H (potential of hydrogen) = the H+ concentration (strength of amount) of ___ ions in solution (when the substance is dissolved in ______) water vp. H is measured of a sale of _____ 0 - 14 v. A p. H of ____ 7 is considered neutral ØA neutral solution is one that has an OH- ions and ____ H+ equal amount of ____ ions e. g. distilled (pure) water
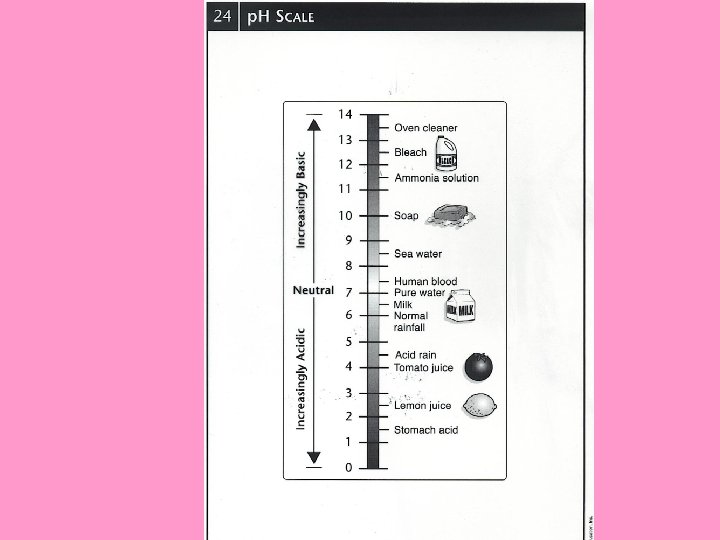

+ H • acid = a substance that forms ____ in 0 -6 solution and has a p. H of ______ vthis means there are more ____ H+ ions OH- ions than ______ e. g. acid rain tomato juice stomach acid

OH- in • base = a compound that forms ____ 8 -14 solution and has a p. H of ______ OH- ions v. This means there are more ____ H+ ions than ____ e. g. alkaline batteries detergents blood

6. 5 and • In the human body a p. H between ___ 7. 5 must be maintained as part of ___ homeostasis _______ vbuffer = a substance used to help maintain p. H within the body _____ e. g. Tums, calcium, pepcid AC, milk of magnesia

- Slides: 32