WarmUp 11316 SWBAT explain the difference between physical

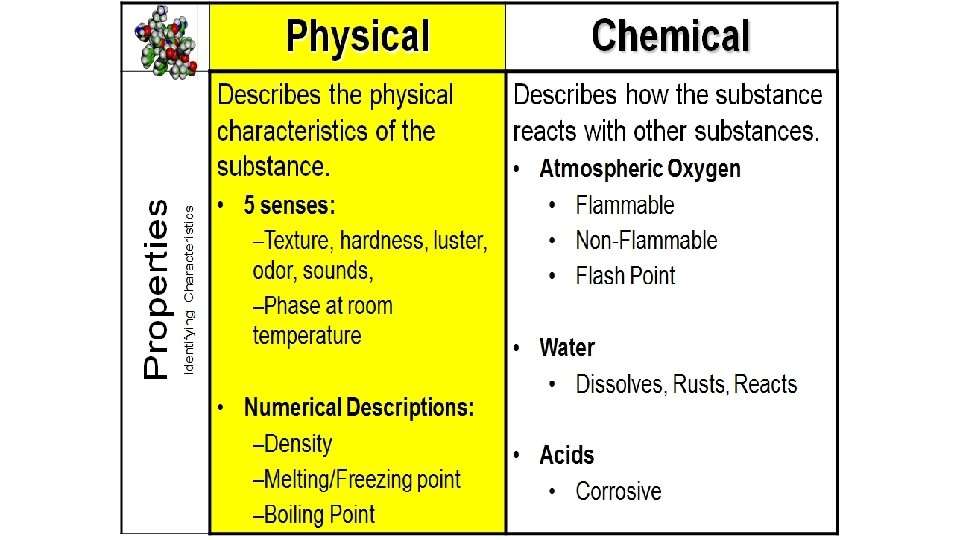
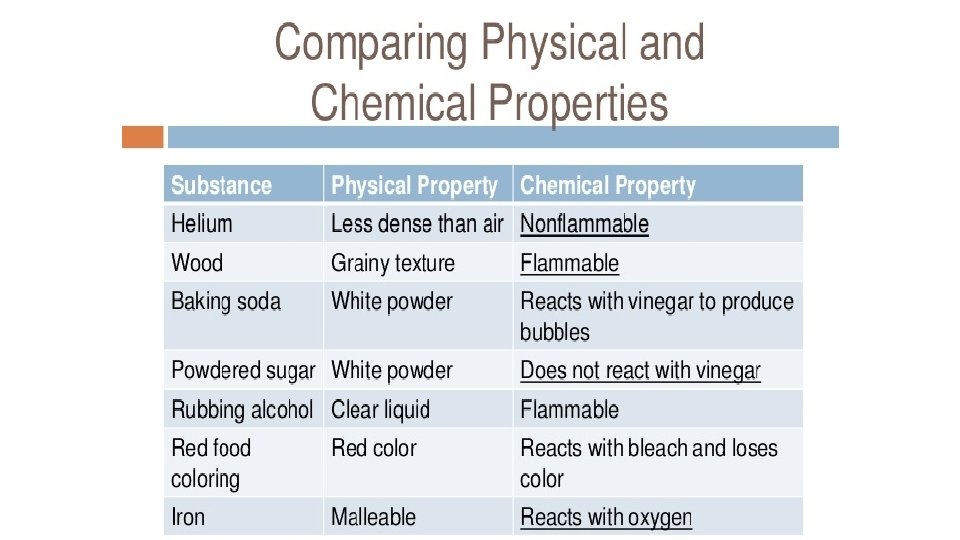
Warm-Up 11/3/16: SWBAT explain the difference between physical and chemical properties. • Think of all of the properties that we have talked about this year, make a list.





Is it a chemical or physical change? Is it reversible? Yes No Physical Change Chemical Change



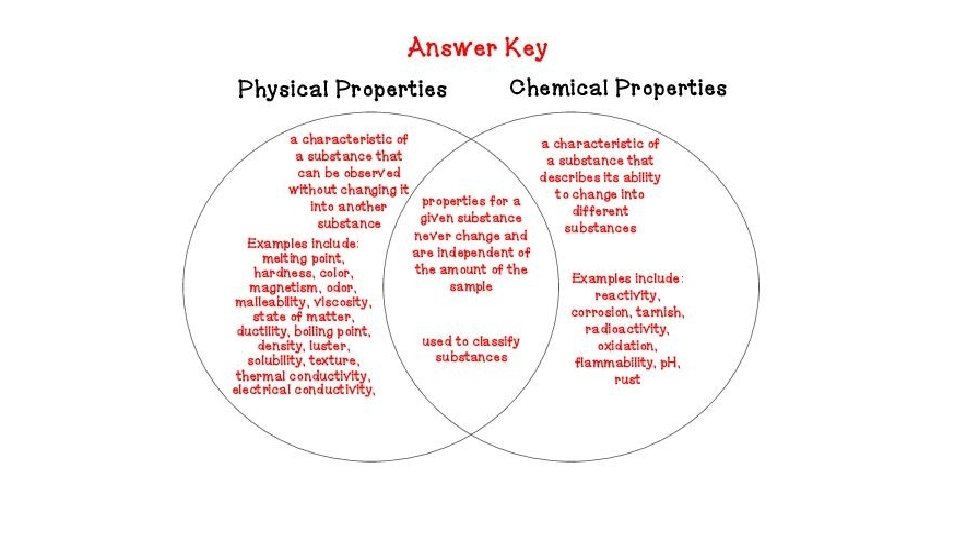
Make a Venn Diagram Physical Properties Chemical Properties

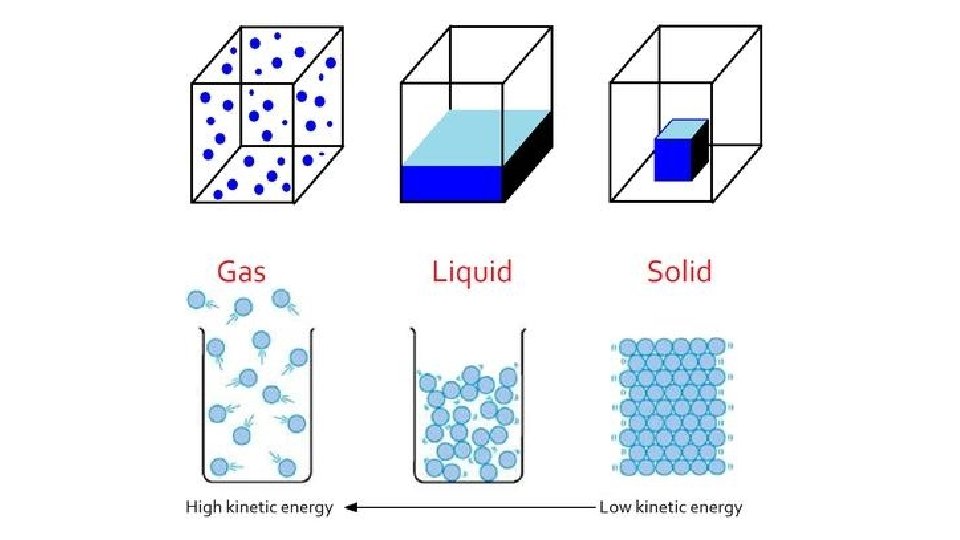
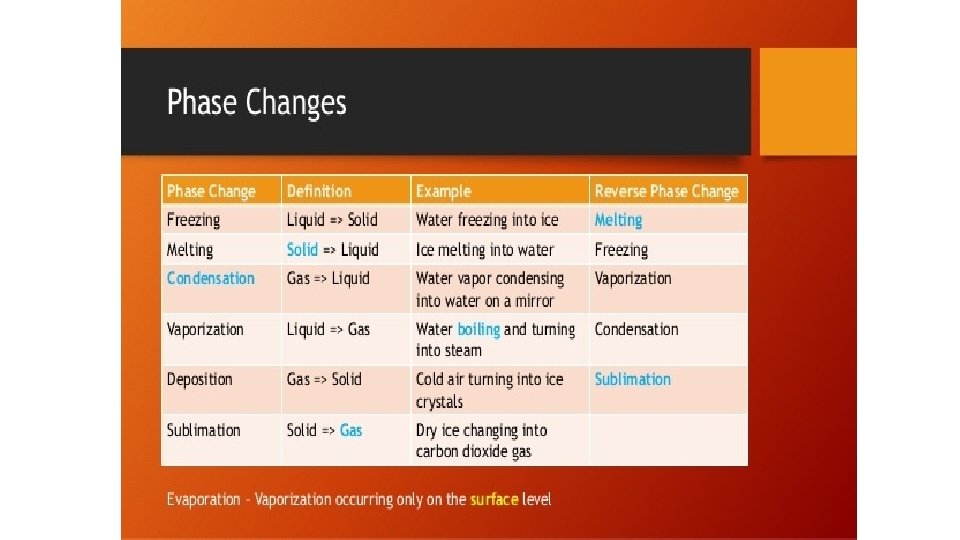
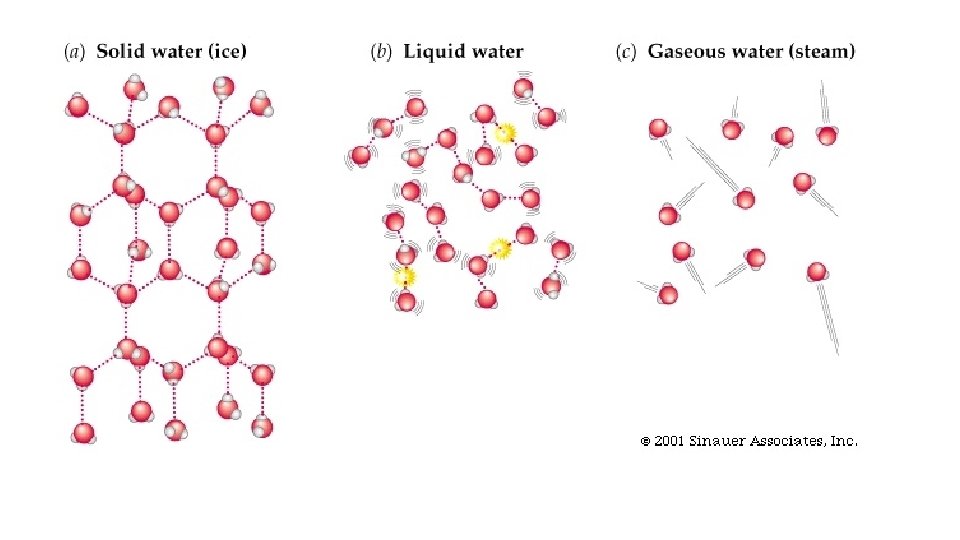
Warm-up 11/22/16 SWBAT describe three states of matter. • What are three states of matter? These are also known as phases. Do you know the six phase changes? � Hint: one is freezing, another is evaporation. • Draw the three phases as best you can, then draw and arrow to show the direction of the phase change and label it.

Quiz Decide whether each change is physical or chemical. 1. Phase Change 2. Boiling Point 3. Capacity to corrode 4. Combustibility 5. Freezing Point 6. Density 7. Electrical Conductivity 8. Viscosity 9. Melting Point 10. Capacity to Oxidize 11. Solubility

Quiz Answers
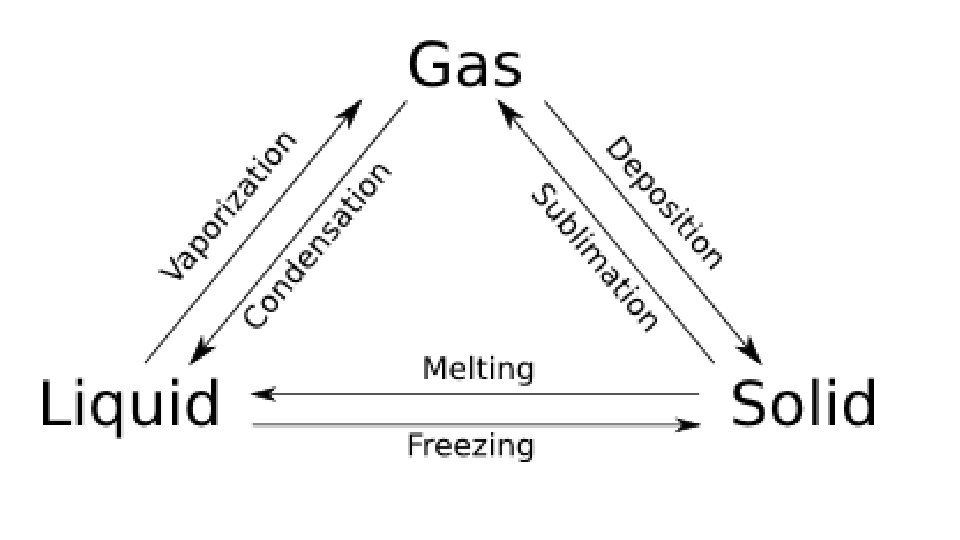
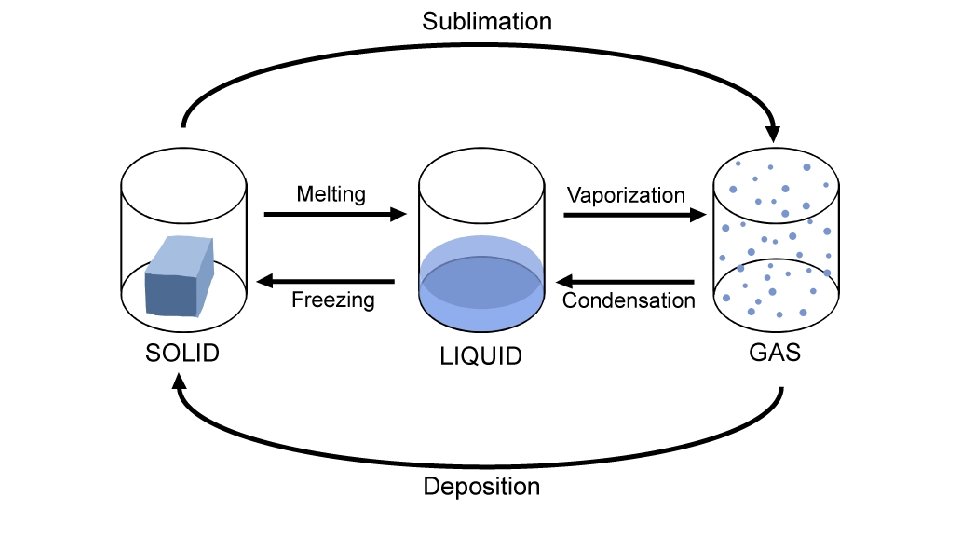
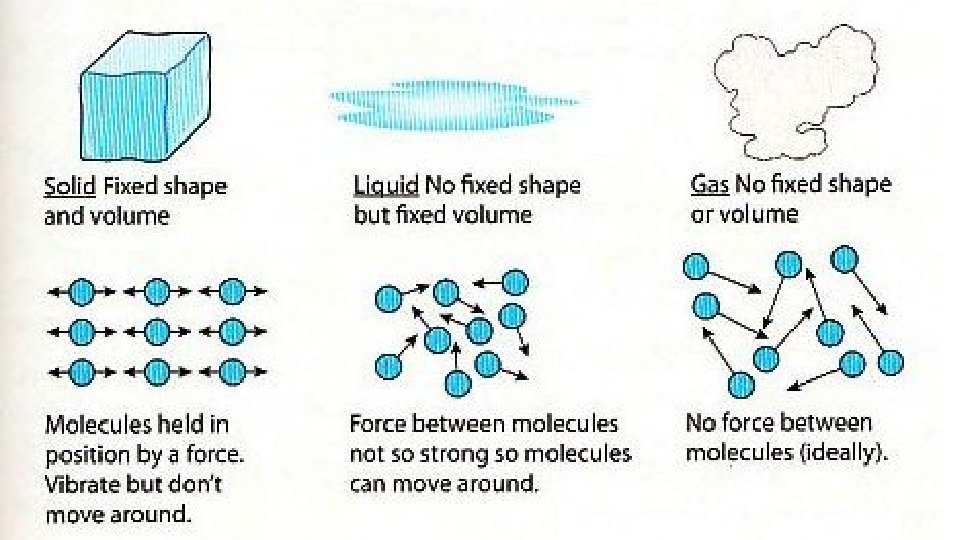
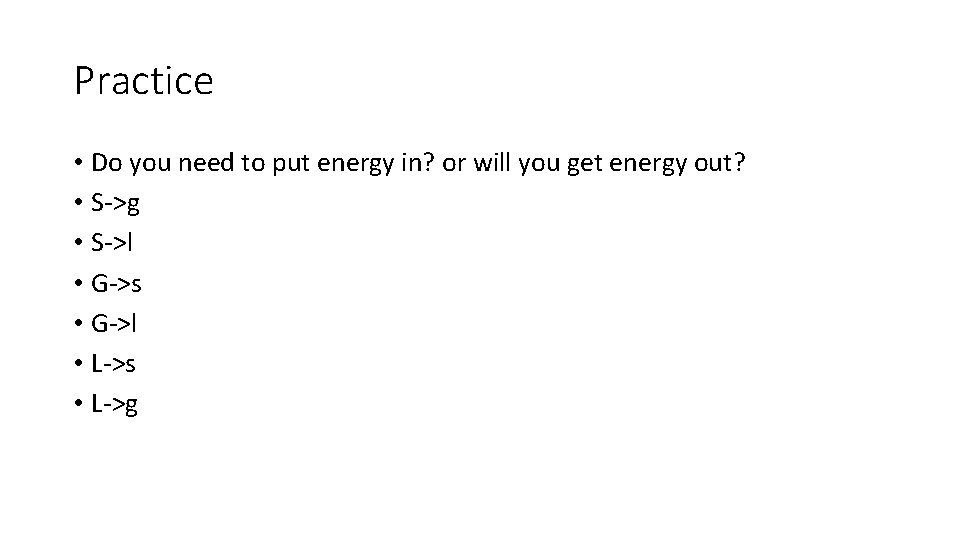
Practice • Do you need to put energy in? or will you get energy out? • S->g • S->l • G->s • G->l • L->s • L->g
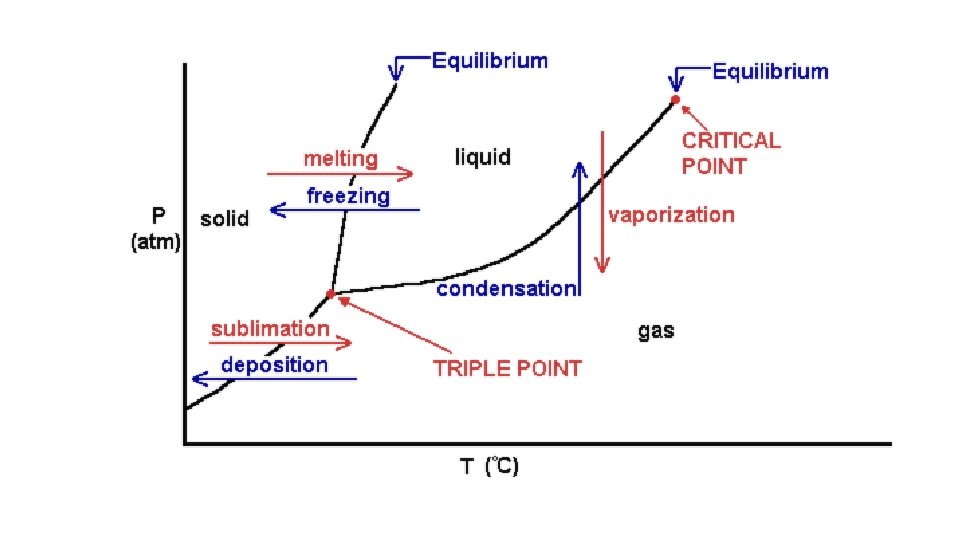
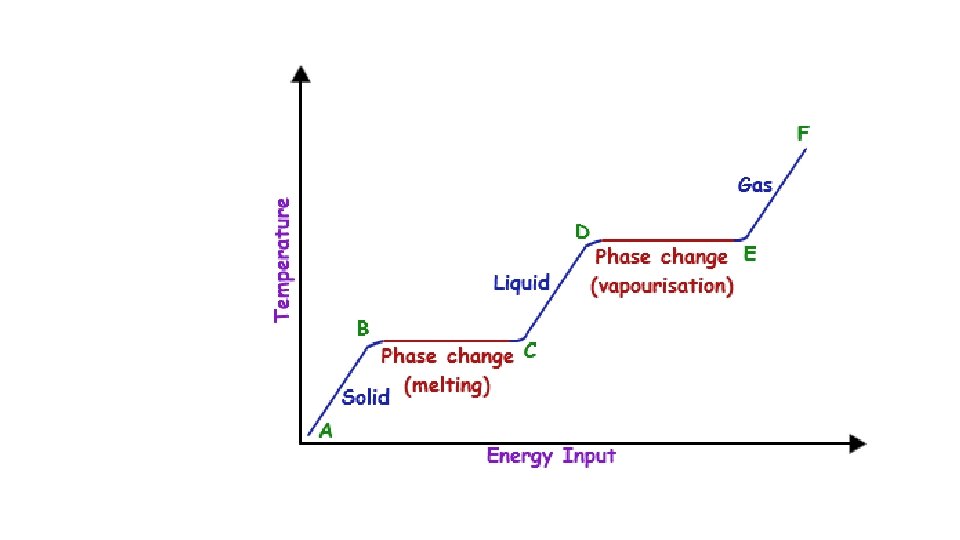

Cool-down • Describe how motion of particles is related to the state of matter. • Make up your own example of each of the phase changes. • How will you study these three states of matter and phase changes for the test? • Videos: • Sublimation and deposition • https: //www. youtube. com/watch? v=Ft 2 Kgtl. P 8 Lk • Triple Point of Cyclohexane • https: //www. youtube. com/watch? v=XEb. MHm. Dhq 2 I • Supercritical Fluids • https: //www. youtube. com/watch? v=y. BRd. Brn. Il. TQ

1 st period took ELA pre-test 11/23 Warm Up 11/23/16 2 nd & 3 rd period only Warm Up 11/28/16 1 st period only SWBAT identify standard units. • Which units (feet, pounds, inches) do we use to measure stuff, imperial or standard? • The units that the rest of the world uses have a name, The International System of Units. Or “SI units” for short, often called the metric system. • Take 3 minutes to list all of the units that you know of and then we will categorize them as SI units or non-SI units. • Today we will be taking notes on a short lecture on standard units(SI units). Have your notes paper ready.

Warm-Up 11/28 2 nd and 3 rd period only SWBAT use dimensional analysis to solve conversion problems. 1. How many milligrams are there in a gram? 2. How many meters are there in a centimeter? 3. How many kilograms are there in a gram? 4. How many decigrams are there in a gram? 5. What are the six phase changes we have covered? 6. What is the difference between physical and chemical properties?

Quiz time 1. List three physical properties and three chemical properties. 2. Draw the phase change diagram. Include as much detail as possible.
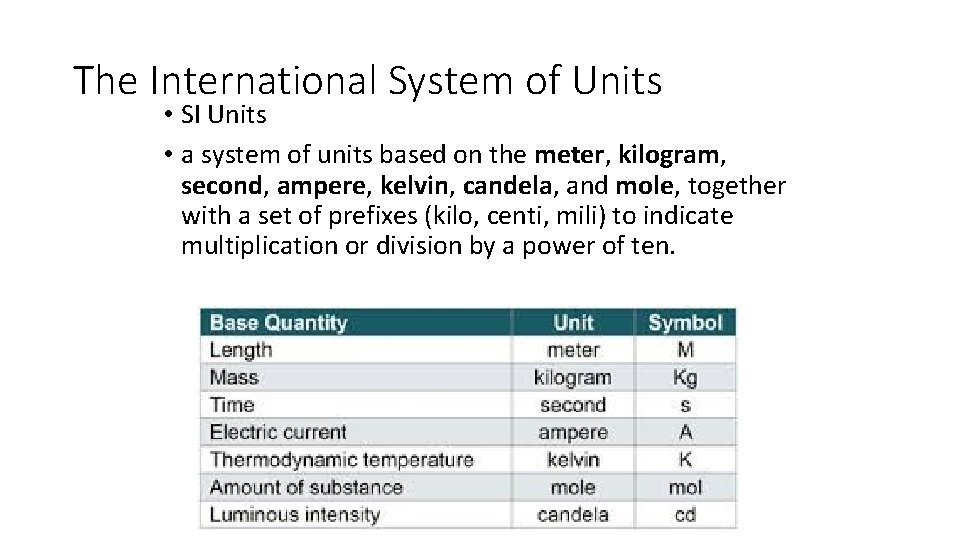
The International System of Units • SI Units • a system of units based on the meter, kilogram, second, ampere, kelvin, candela, and mole, together with a set of prefixes (kilo, centi, mili) to indicate multiplication or division by a power of ten.
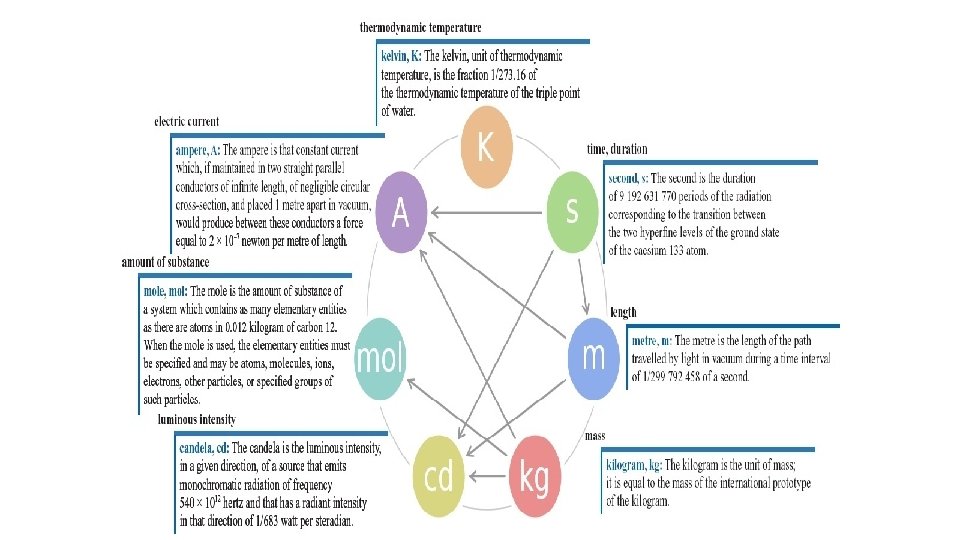

This is the “metric system”
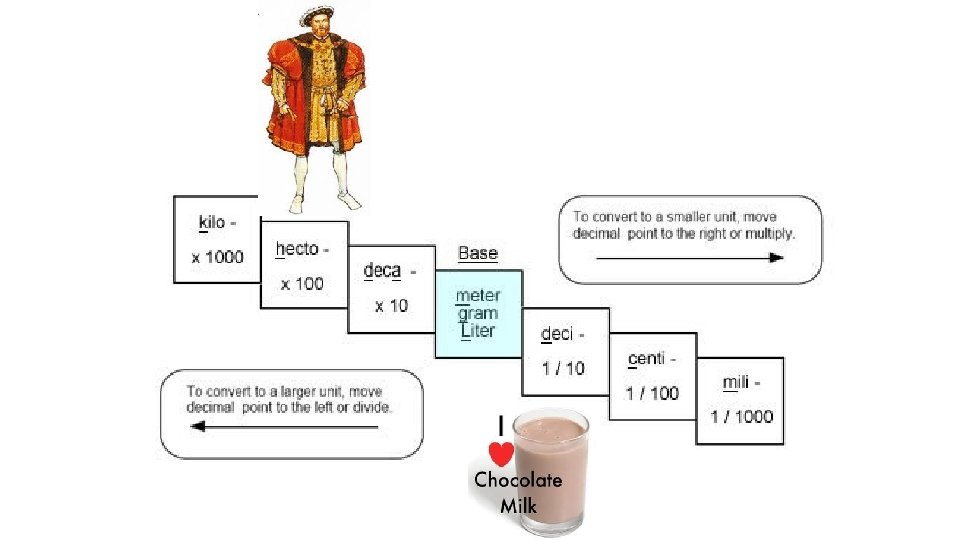


Let’s put this to use! • What is the speed of light? • Can you fathom what this means? • What units would make this number more meaningful? • How can you convert to those units?

Practice with Dimensional Analysis In labs, we will use graduated cylinders and beakers to measure volume, they are labeled with milliliters. Let’s practice some useful conversions. 1. How many liters in a milliliter? 2. How many milliliters in a liter? 3. How many milliliters in 10 L? 4. How many m. L in 11. 7 L? 5. How many L in 7, 892 m. L? 6. How many m. L in. 62 L?

Homework • Unit Conversions Worksheet – 6 problems, due Wednesday.

WARM-UP: 11/30/16: SWBAT calculate density. Which do you think would have the greater volume and mass? Why? • 1 lb of feathers • 1 lb of rock

Density is defined as mass per unit volume. It is a measure of how tightly packed and how heavy the molecules are in an object. Density is the amount of matter within a certain volume.
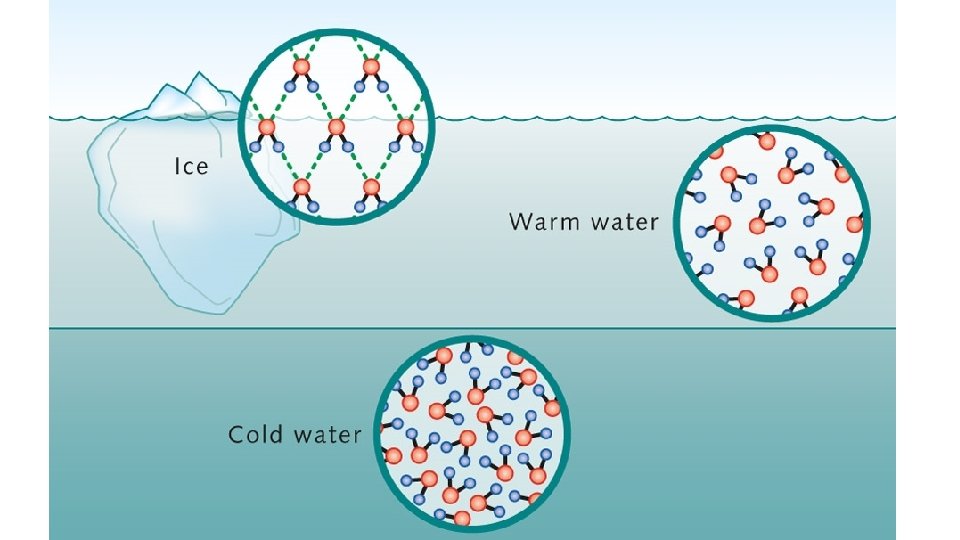

Proof that water and ice have different densities

To find the density 1 - Find the mass of the object 2 - Find the volume of the object 3 - Divide = Mass Volume Density

Units for density g/cm 3 or g/ml Formula: M = mass M=Dx. V V= volume D = density V=M/D D=M/V

Warm-Up 12/1/16 SWBAT calculate the density of various objects in the lab. 1. If the mass of an object is 35 grams and it takes up 7 cm calculate the density. 3 of space, To find density : 1) Find the mass of the object 2) Find the volume of the object 3) Divide : Density = Mass - Volume

To find density : 1) Find the mass of the object 2) Find the volume of the object 3) Divide : Density = Mass - Volume Ex. If the mass of an object is 35 grams and it takes up 7 cm space, calculate the density. 3 of Set up your density problems like this: Given: Mass = 35 grams Volume = 7 cm 3 Unknown: Density (g/ cm 3 ) Formula: D = M / V Solution: D = 35 g/7 cm D = 5 g/cm 3 3

Density Lab Find the density of each object in the bag, then predict whether each object would sink or float in each of the following liquids. MATERIAL DENSITY (g/cm 3 or g/m. L) Dawn dish soap 1. 06 Light corn syrup 1. 33 Maple Syrup 1. 37 Honey 1. 42

Warm-Up 12/2/16 SWBAT name the parts of a lab report and use data from the density lab to write a “results section” of a lab report. Use this website to find the parts of a lab report: https: //owl. english. purdue. edu/owl/resource/670/03/ Write only the results section of the lab report for the density lab.

The “Results” Section • Restate hypothesis • Most important goes first • Use charts, graphs, or tables to make data very easy to read • Equations used • Sample calculations • You should always have multiple trials in case or error

Warm-Up 12/5/16 SWBAT complete problems using dimensional analysis. 1. How many milliliters in 25. 7 liters? 2. How many milliliters in 12. 7 kiloliters? 3. How many grams in 15 decigrams? 4. How many decigrams in 2 kilograms? 5. How many centimeters in 5. 78 meters? 6. How many centimeters in 12. 223 decameters? 7. How would you find your own density? My gradebook closes this Friday, the 9 th for late work.

You will get your homework back and have a chance to ask questions.

Quiz 1. How many milliliters in 25. 7 liters? 2. How many milliliters in 7. 7 kiloliters? 3. How many grams in 0. 5 decigrams?

Quiz Answers • 25700 m. L • 7, 700, 000 m. L • 0. 05 g

Warm-up 12/6/16 SWBAT distinguish between physical and chemical changes. 1. What is the difference between physical and chemical changes?

Lab Notes Question: What type of change is occurring between the vinegar and the baking soda? Substance Litmus Paper Test Observations Vinegar and Sugar Vinegar and Baking Soda Leave room to record any other info, like the amount of each substance, or the type of sugar used, or % acidity of the vinegar, or materials. Write down EVERYTHING. Leave at least a quarter of a page.
1 st period Lab Groups Group 1 Group 2 Group 3 Group 4 Group 5 Group 6 Group 7 Group 8 Cassie Carlos Ethan Rachel Sieanna Jordana Taleah Riley Shaelee Olivia Talia Kyler Karlie Aidan Eddy Roman Liam Ella Hayden Chloe Ashton Tatianna Dylan Kari Luis Samantha Luke Quinn Dakota Tyler Ciara EVERYONE in the group must write down EVERYTHING that was done. EVERY measurement, EVERY calculation and EVERY observation in your notebook. Nice and neat. This is a part of your lab report. It is the original data and is always stapled to the lab report, in part to prove you actually did the lab. A lab report without data will earn a zero.
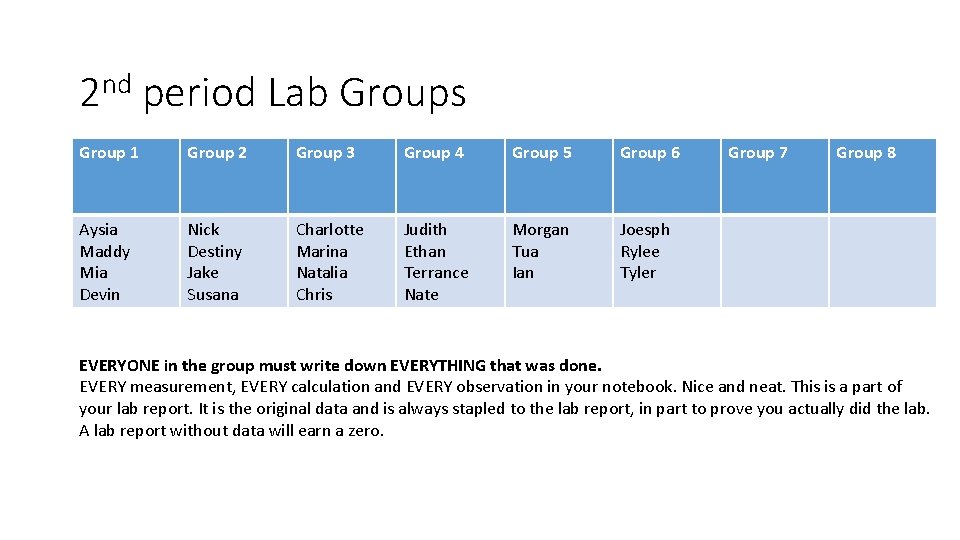
2 nd period Lab Groups Group 1 Group 2 Group 3 Group 4 Group 5 Group 6 Aysia Maddy Mia Devin Nick Destiny Jake Susana Charlotte Marina Natalia Chris Judith Ethan Terrance Nate Morgan Tua Ian Joesph Rylee Tyler Group 7 Group 8 EVERYONE in the group must write down EVERYTHING that was done. EVERY measurement, EVERY calculation and EVERY observation in your notebook. Nice and neat. This is a part of your lab report. It is the original data and is always stapled to the lab report, in part to prove you actually did the lab. A lab report without data will earn a zero.
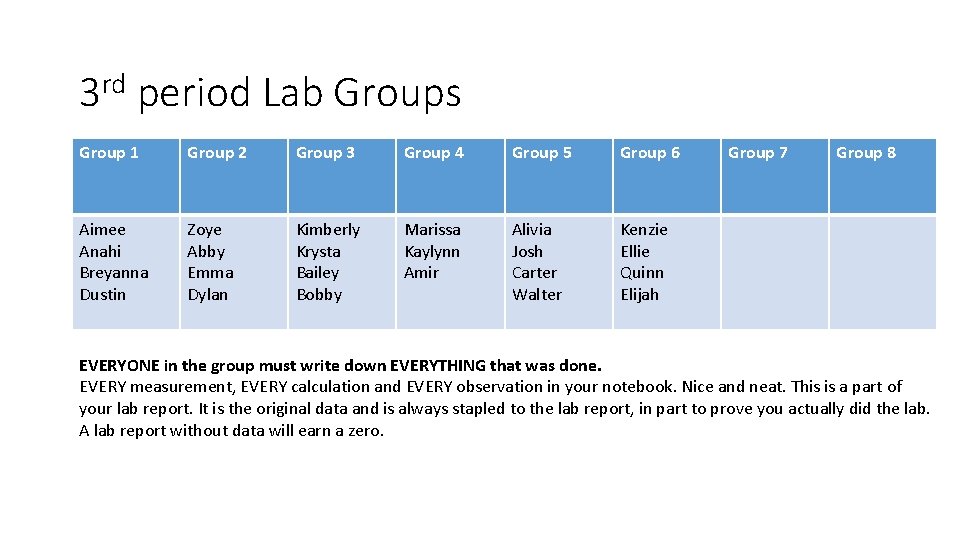
3 rd period Lab Groups Group 1 Group 2 Group 3 Group 4 Group 5 Group 6 Aimee Anahi Breyanna Dustin Zoye Abby Emma Dylan Kimberly Krysta Bailey Bobby Marissa Kaylynn Amir Alivia Josh Carter Walter Kenzie Ellie Quinn Elijah Group 7 Group 8 EVERYONE in the group must write down EVERYTHING that was done. EVERY measurement, EVERY calculation and EVERY observation in your notebook. Nice and neat. This is a part of your lab report. It is the original data and is always stapled to the lab report, in part to prove you actually did the lab. A lab report without data will earn a zero.

Litmus Paper • Litmus paper can be used to tell if a chemical change has occurred because it tells us if the p. H (p. H measures acidity) of a substance has changed by changing color. The litmus paper is made by taking dyes from a plant called lichen. Then a paper absorbs those dyes and has the property of being a “p. H indicator” meaning it tells you if the p. H of something changes.

C. E. R. Method A framework for shaping a scientific explanation. Claim: a statement that answers a question Evidence: qualitative or quantitative data/observations that support the claim Reasoning: scientific concept or explanation that links the evidence to the claim

Warm-UP 12/7/16: SWBAT write a chemical equation. 1. Define atom, molecule, and compound. 2. How many different elements are in the following equation? 3. Are elements or compounds formed in chemical reactions? 4. Define heterogeneous and homogeneous mixtures, give an example of each. Could your example be the other type of mixture under certain circumstances. EX: can of coke 5. What is inside the bubbles that form when you put hydrogen peroxide on a cut. 6. Finish your scientific explanation using CER method.

or ?



Let’s take a quiz together… • http: //www. quia. com/quiz/303980. html
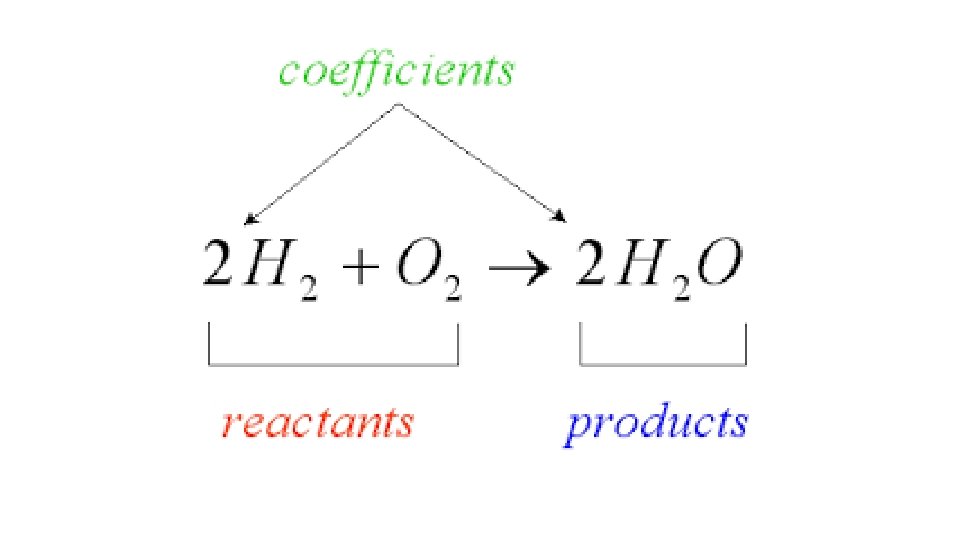
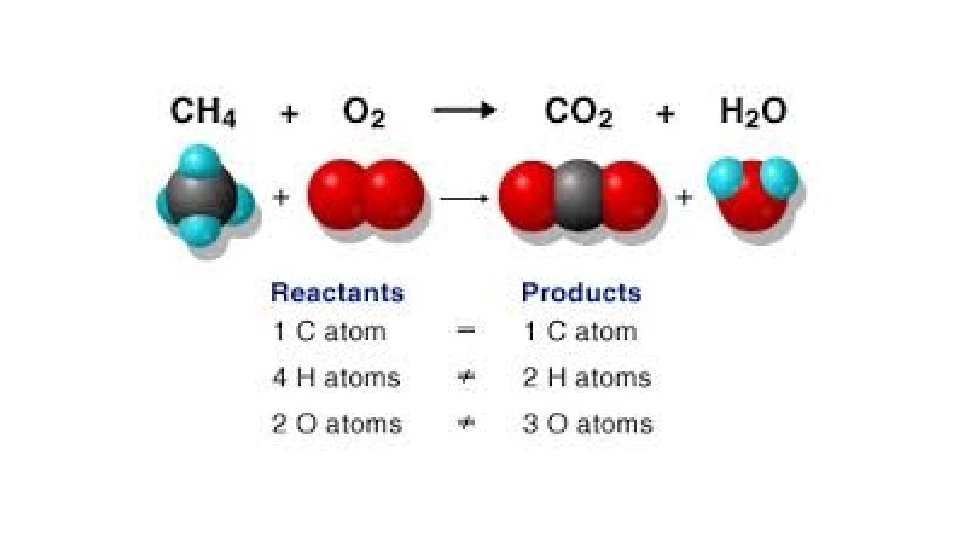
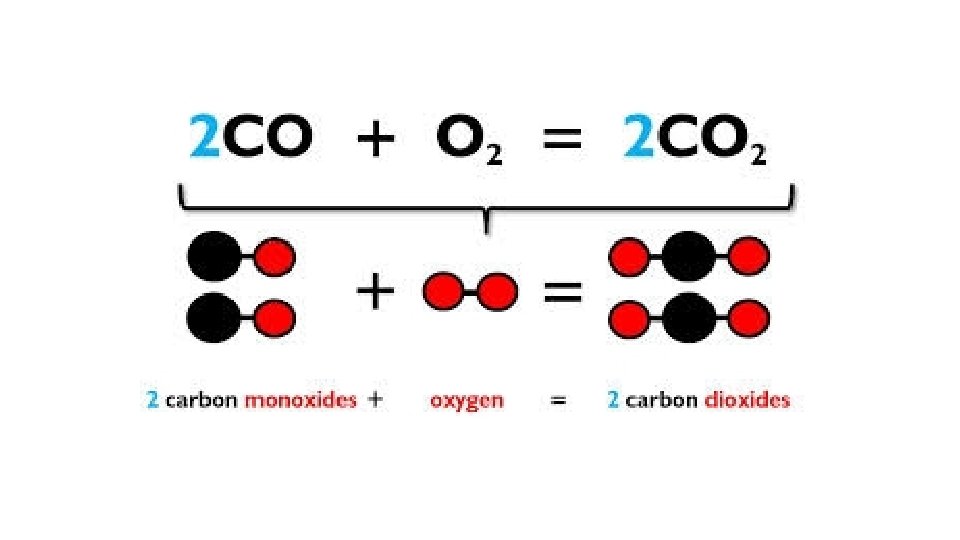
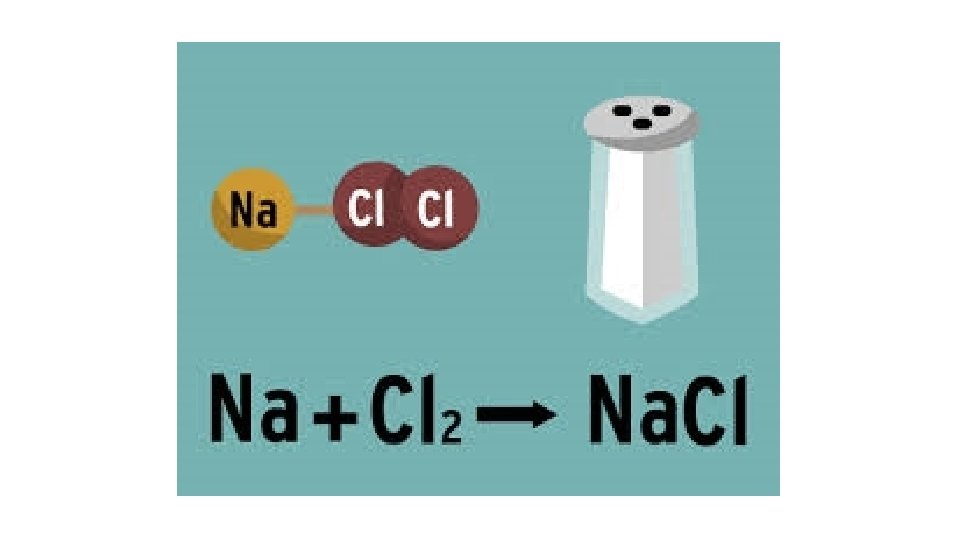
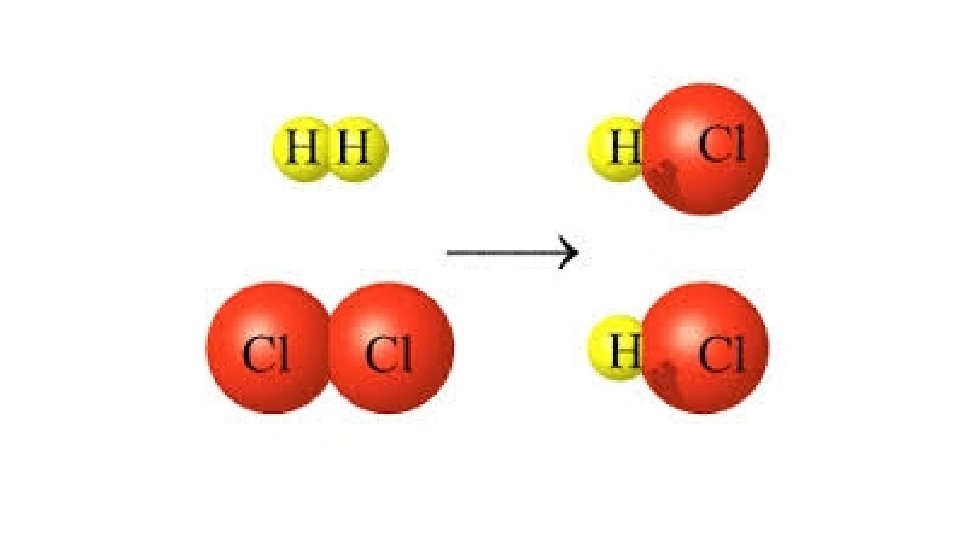

Warm-Up 12/8/16 SWBAT balance chemical equations. 1. H 2 O 2 decomposes to form water and oxygen gas. 2. Hydrogen gas combines with oxygen gas to form liquid water. 3. Magnesium reacts with oxygen gas to produce magnesium oxide. How many magnesium atoms do you need for each oxygen atom, keeping the octet rule in mind? Remember, magnesium is in group 2, so it has 2 valence electrons.

Homework • You have two worksheets. • On the first you only need to balance the equation by writing in the coefficient. You may add s, l, and g after each formula. • On the second sheet you must read the word problem, write the equation yourself, and then balance it. • Challenge: Whoever has the most correct at the beginning of class tomorrow wins a prize. (both worksheets) • Start your homework now so that I can solve the harder ones with you in class.

A few more word problems…. . 1. Sodium metal and chlorine gas react to form solid sodium chloride. 2. Lithium metal reacts with fluorine to produce solid lithium fluoride. 3. Liquid Ammonia (aka nitrogen trihydride, NH 3) decomposes to form nitrogen gas and hydrogen gas. 4. Liquid Hydrochloric acid (HCl) decomposes to form hydrogen gas and chlorine gas. 5. Sodium metal and water react to form solid sodium hydroxide (Na. OH) and hydrogen gas. 6. Methane gas(CH 4 ) and oxygen gas combine to form carbon dioxide and water. 7. Potassium and chlorine gas react to form solid potassium chloride. 8. Calcium and bromine react to form solid calcium bromide. 9. Calcium and oxygen react to form solid calcium oxide.

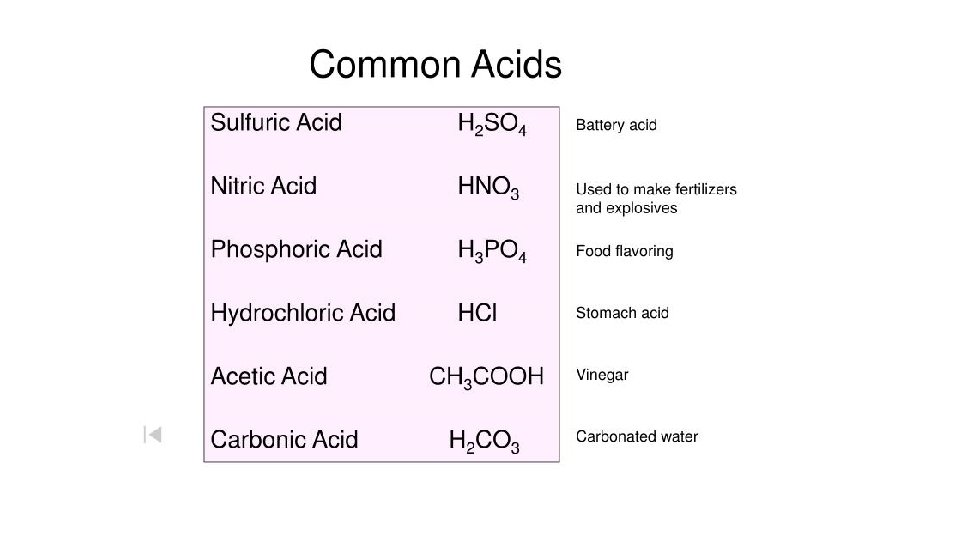
Warm-Up 12/9/16 SWBAT describe acids and bases. What do you know about acids and bases?

Words that describe acids and bases • Acids and bases are nouns, you can hold an acid, you can touch a base. • Acids are acidic and bases are basic, so acidic and basic and adjectives, that describe the property of being like an acid or like a base. You cannot hold acidic. • Acids have a certain level of acidic-ness, we call it acidity. We talk about the acidity of acids and basicity of bases. These words are actually nouns, you can point to a level of acidity, so acidity is a thing.
Acid and Bases
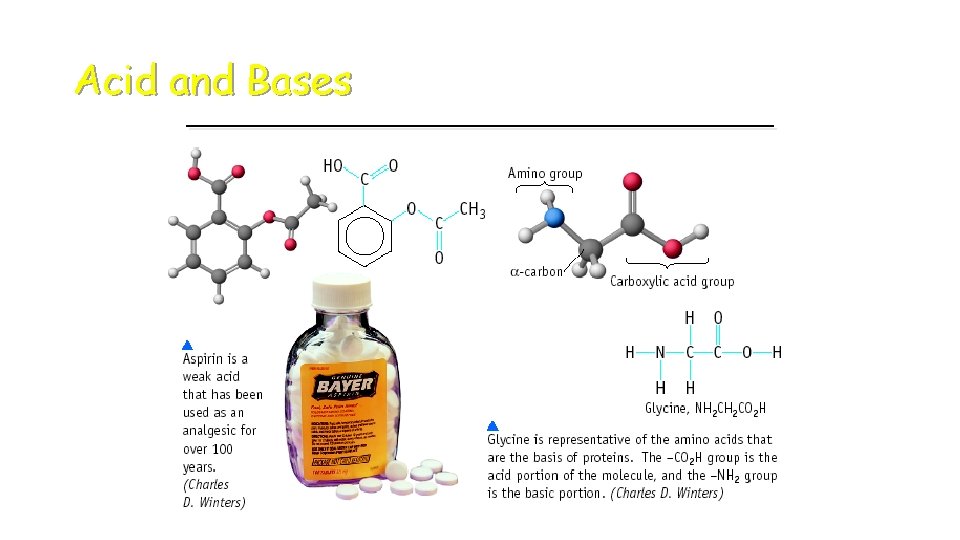
Acid and Bases
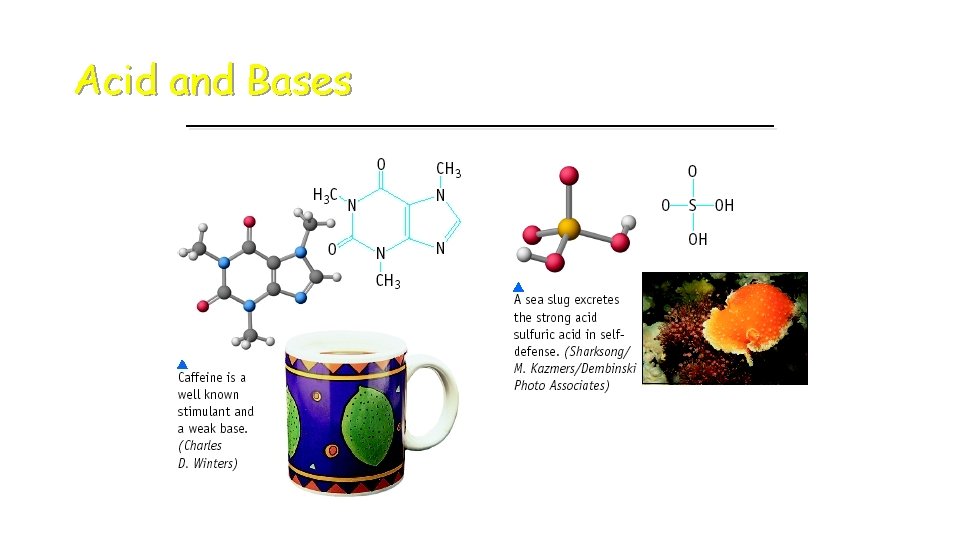
Acid and Bases

Some Properties of Acids þ Produce H+ (as H 3 O+) ions in water (the hydronium ion is a hydrogen ion attached to a water molecule) þ Taste sour þ Corrode metals þ Electrolytes þ React with bases to form a salt and water þ p. H is less than 7 þ Turns blue litmus paper to red “Blue to Red A-CID”

Some Properties of Bases þ Produce OH- ions in water þ Taste bitter, chalky þ Are electrolytes þ Feel soapy, slippery þ React with acids to form salts and water þ p. H greater than 7 þ Turns red litmus paper to blue “Basic Blue” Bases are also called “alkaline. ”

Some Common Bases Na. OH sodium hydroxide KOH potassium hydroxide Ba(OH)2 barium hydroxide Mg(OH)2 magnesium hydroxide Al(OH)3 aluminum hydroxide lye liquid soap stabilizer for plastics Milk of magnesia Maalox (antacid)

Definitions Arrehenius • Acids – produce H+ • Bases - produce OH- only in water Bronsted-Lowry any solvent • Acids – donate H+ • Bases – accept H+ Lewis used in organic chemistry, wider range of substances • Acids – accept e- pair • Bases – donate e- pair
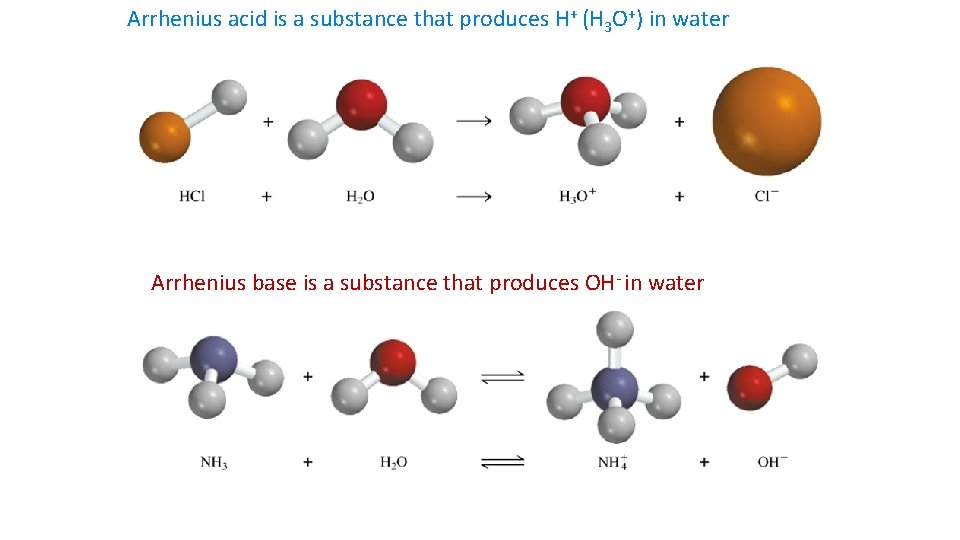
Arrhenius acid is a substance that produces H+ (H 3 O+) in water Arrhenius base is a substance that produces OH- in water
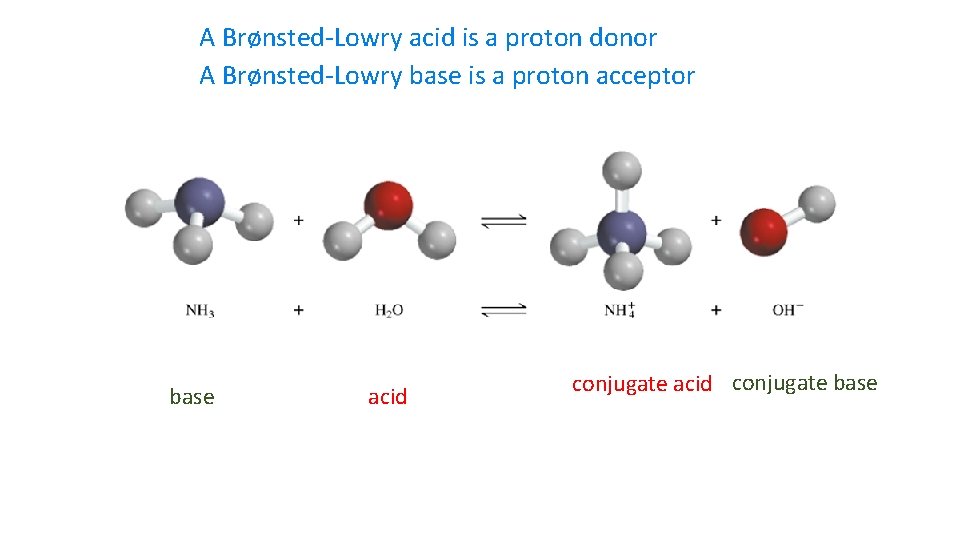
A Brønsted-Lowry acid is a proton donor A Brønsted-Lowry base is a proton acceptor base acid conjugate base
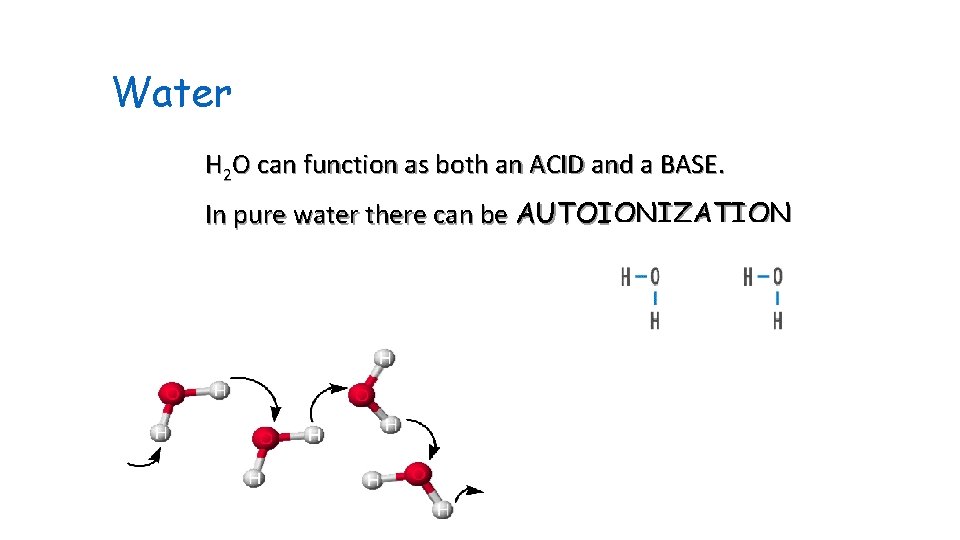
Water H 2 O can function as both an ACID and a BASE. In pure water there can be AUTOIONIZATION

The p. H scale is a way of expressing the strength of acids and bases. Instead of using very small numbers, we just use the NEGATIVE power of 10 on the Molarity(concentration) of the H+ (or OH-) ion. Under 7 = acid 7 = neutral Over 7 = base Notice acids come before bases on the p. H scale, you can remember that because they

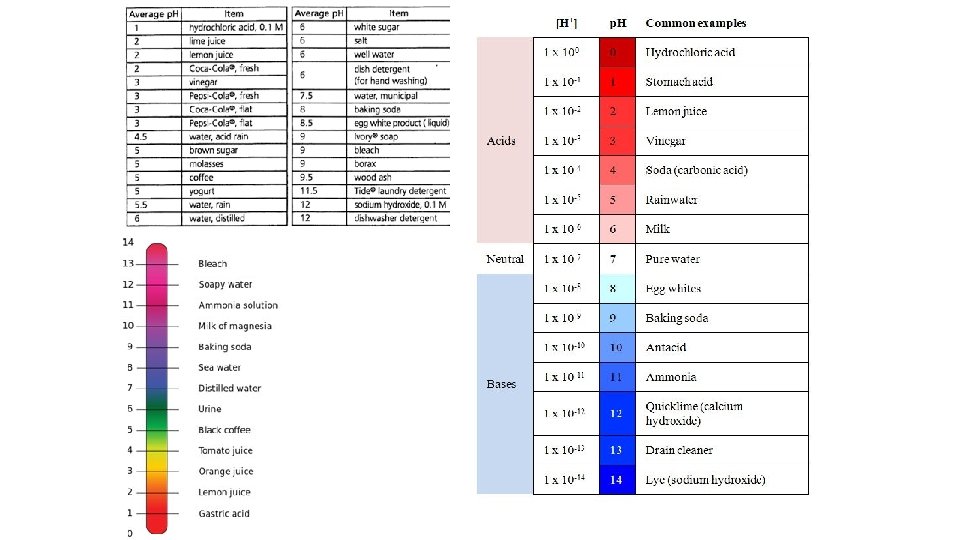
p. H of Common Substances
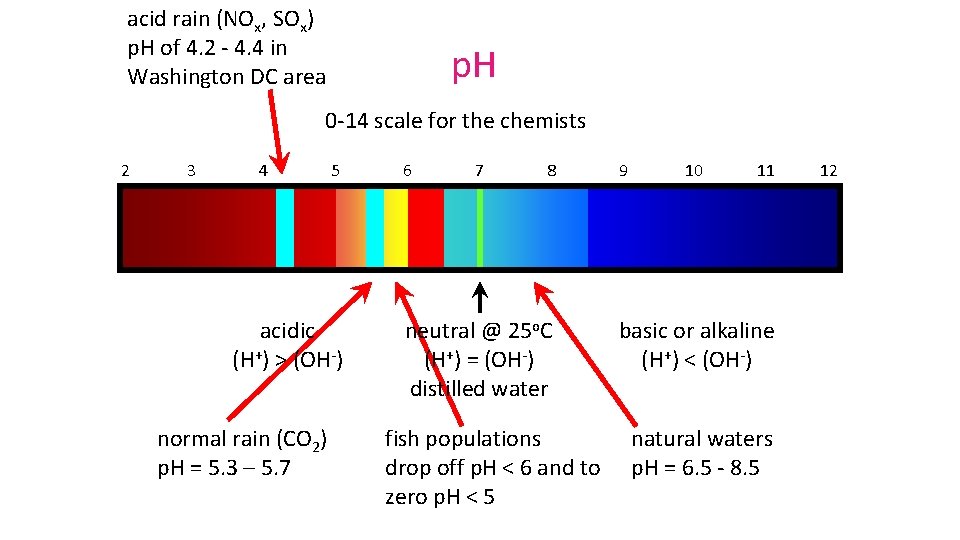
acid rain (NOx, SOx) p. H of 4. 2 - 4. 4 in Washington DC area p. H 0 -14 scale for the chemists 2 3 4 5 acidic (H+) > (OH-) normal rain (CO 2) p. H = 5. 3 – 5. 7 6 7 8 neutral @ 25 o. C (H+) = (OH-) distilled water fish populations drop off p. H < 6 and to zero p. H < 5 9 10 11 basic or alkaline (H+) < (OH-) natural waters p. H = 6. 5 - 8. 5 12
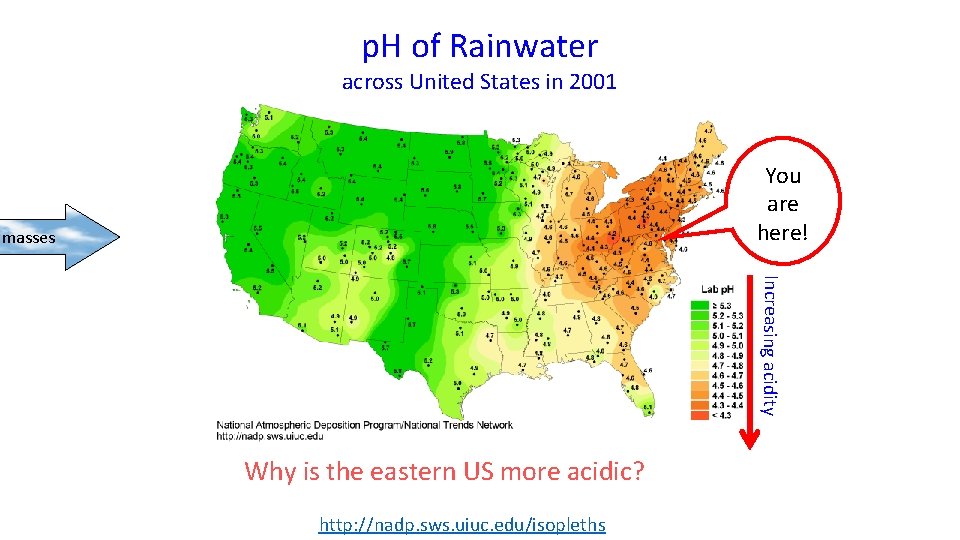
p. H of Rainwater across United States in 2001 You are here! masses Increasing acidity Why is the eastern US more acidic? http: //nadp. sws. uiuc. edu/isopleths

What is acid rain? Dissolved carbon dioxide lowers the p. H CO 2 (g) + H 2 O H 2 CO 3 H+ + HCO 3 - Atmospheric pollutants from combustion NO, NO 2 + H 2 O … HNO 3 both strong acids SO 2, SO 3 + H 2 O … H 2 SO 4 p. H < 5. 3

p. H testing • There are several ways to test p. H • Blue litmus paper (red = acid) • Red litmus paper (blue = basic) • p. H paper (multi-colored) • p. H meter (7 is neutral, <7 acid, >7 base) • Universal indicator (multi-colored) • Indicators like phenolphthalein • Natural indicators like red cabbage, radishes

Paper testing • Paper tests like litmus paper and p. H paper • Put a stirring rod into the solution and stir. • Take the stirring rod out, and place a drop of the solution from the end of the stirring rod onto a piece of the paper • Read and record the color change. Note what the color indicates. • You should only use a small portion of the paper. You can use one piece of paper for several tests.

p. H paper

p. H meter • Tests the voltage of the electrolyte • Converts the voltage to p. H • Very cheap, accurate • Must be calibrated with a buffer solution

p. H indicators • Indicators are dyes that can be added that will change color in the presence of an acid or base. • Some indicators only work in a specific range of p. H • Once the drops are added, the sample is ruined • Some dyes are natural, like radish skin or red cabbage

Warm-Up 12/12 SWBAT distinguish between strong and weak acids or bases. 1. What are the 3 definitions of acids, and bases? 2. Why are there 3 definitions? 3. What are some properties of acids and bases? 4. Can you give a few examples of common acids and bases? • Take out your agenda! • Quiz – blank sheet of paper, no notes, it can be in your composition book

Schedule • Today: finish acid and base notes, physical and chemical properties quiz on Study Island • Tuesday: Multiple choice test • Wednesday: Practice summary • Thursday: Performance summary • Friday: Blogging or SI test • Monday: SI test or Blogging • Tuesday: Misconception test • Wednesday: Grades due/ Demo Labs
Homework: Physical and Chemical Properties Quiz

Quiz Label the products and reactants and balance the equation. 1. Sodium metal and chlorine gas react to form solid sodium chloride. 2. Sodium metal and water react to form solid sodium hydroxide (Na. OH) and hydrogen gas. 3. Potassium and chlorine gas react to form solid potassium chloride. 4. Calcium and bromine react to form solid calcium bromide. 5. Calcium and oxygen react to form solid calcium oxide. 6. N 2 + H 2 -> NH 3 7. KCl. O 3 -> KCl + O 2 8. C 3 H 8 + O 2 -> CO 2 + H 2 O
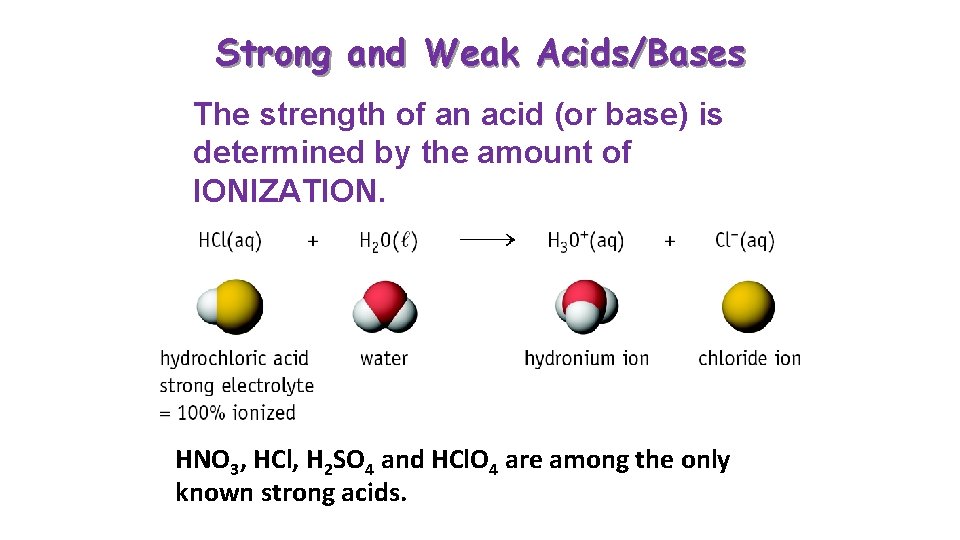
Strong and Weak Acids/Bases The strength of an acid (or base) is determined by the amount of IONIZATION. HNO 3, HCl, H 2 SO 4 and HCl. O 4 are among the only known strong acids.
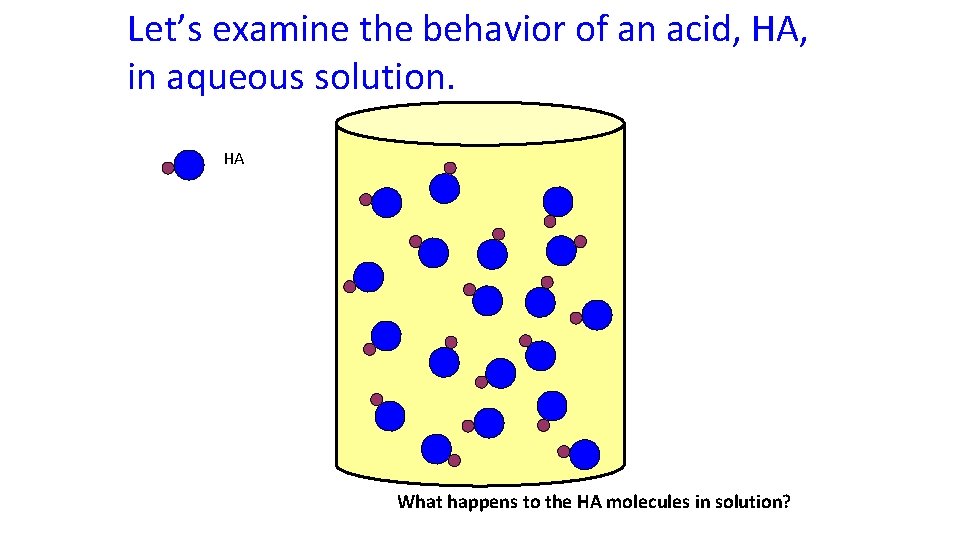
Let’s examine the behavior of an acid, HA, in aqueous solution. HA What happens to the HA molecules in solution?
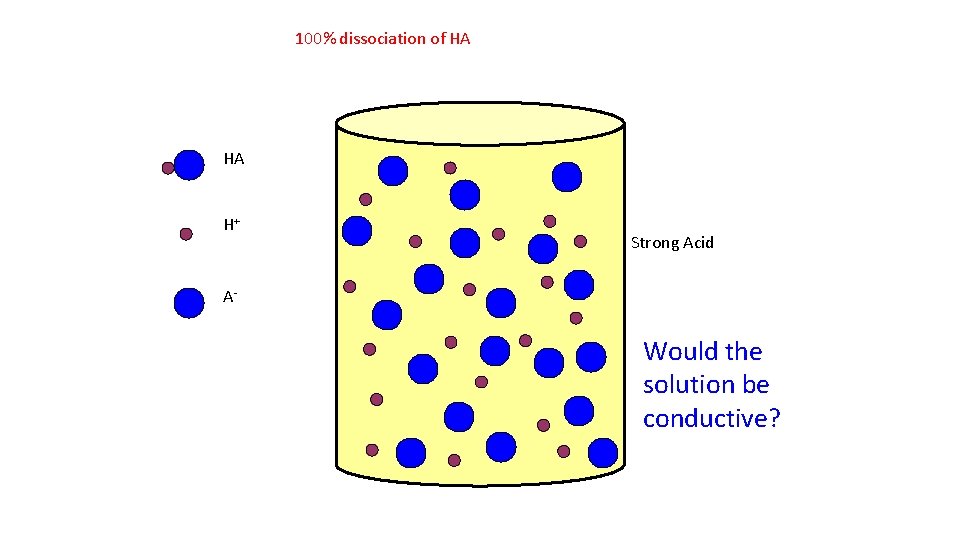
100% dissociation of HA HA H+ Strong Acid A- Would the solution be conductive?
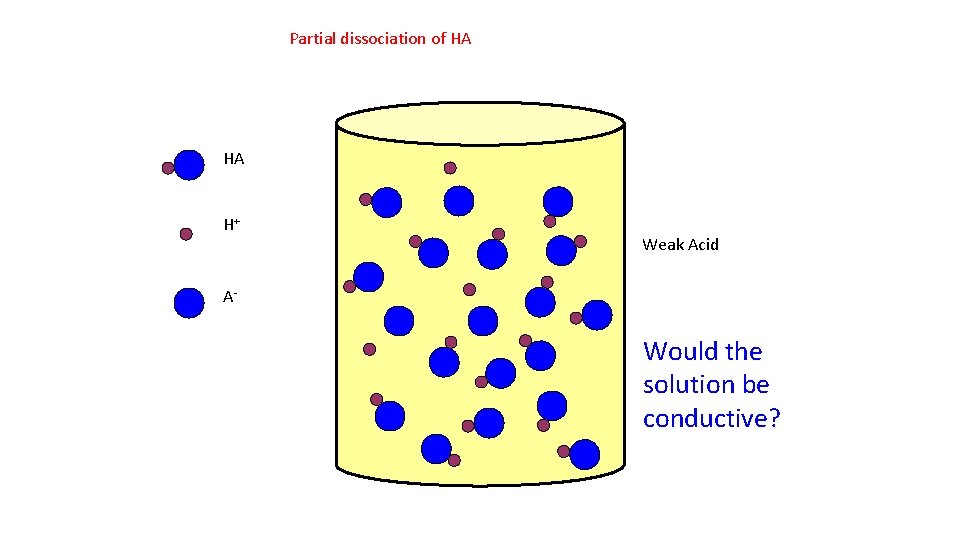
Partial dissociation of HA HA H+ Weak Acid A- Would the solution be conductive?
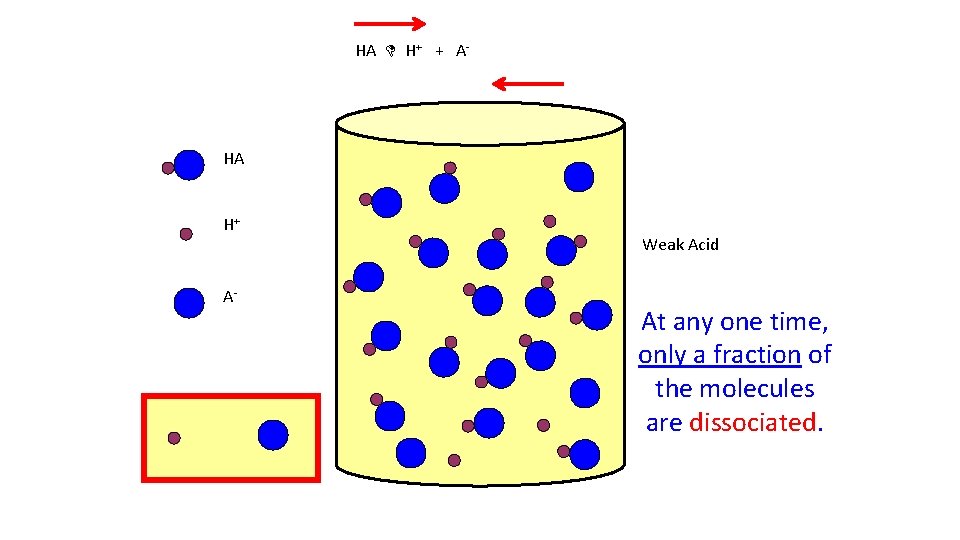
HA H+ + A- HA H+ A- Weak Acid At any one time, only a fraction of the molecules are dissociated.
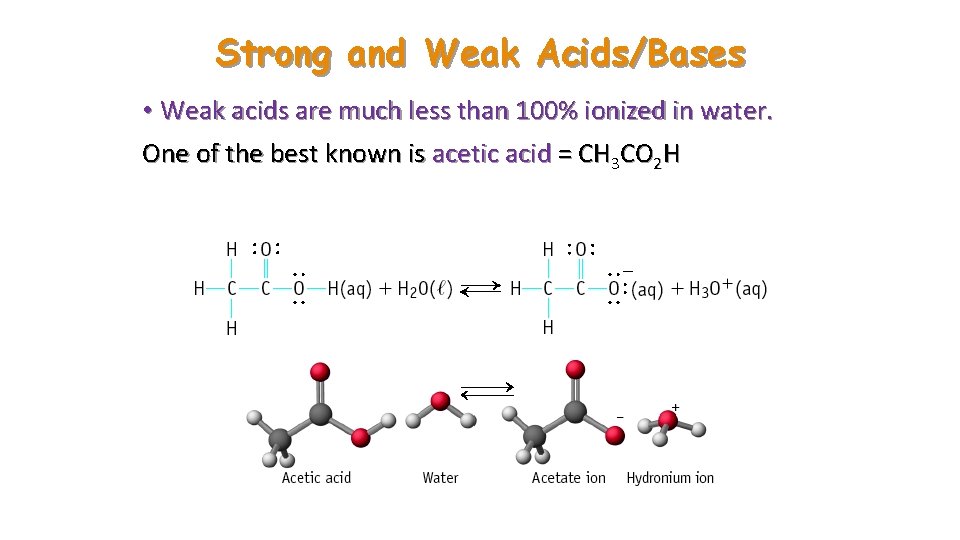
Strong and Weak Acids/Bases • Weak acids are much less than 100% ionized in water. One of the best known is acetic acid = CH 3 CO 2 H
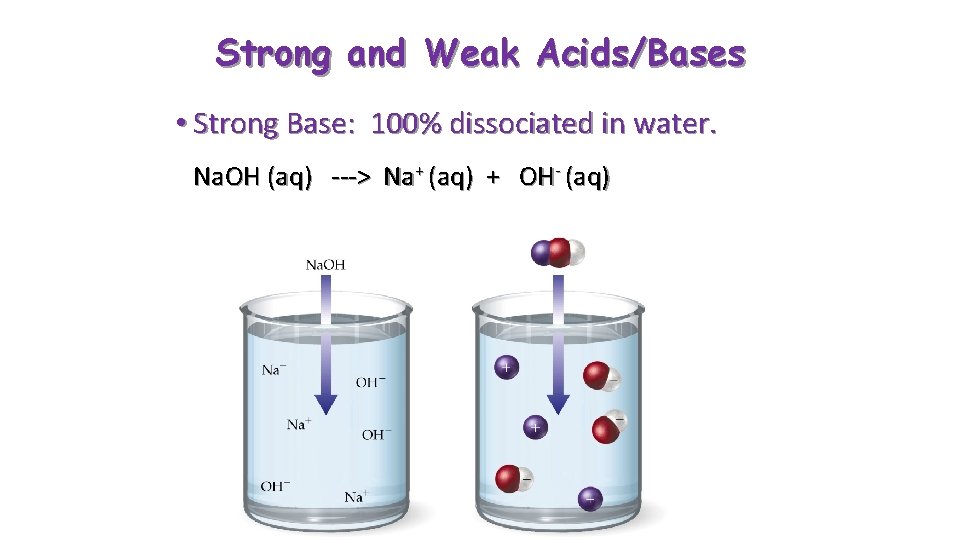
Strong and Weak Acids/Bases • Strong Base: 100% dissociated in water. Na. OH (aq) ---> Na+ (aq) + OH- (aq)
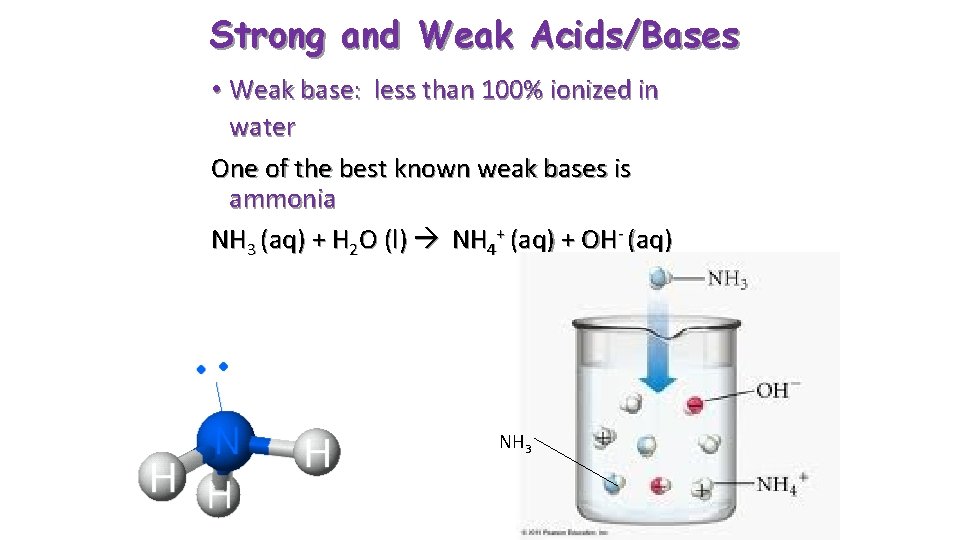
Strong and Weak Acids/Bases • Weak base: less than 100% ionized in water One of the best known weak bases is ammonia NH 3 (aq) + H 2 O (l) NH 4+ (aq) + OH- (aq) NH 3
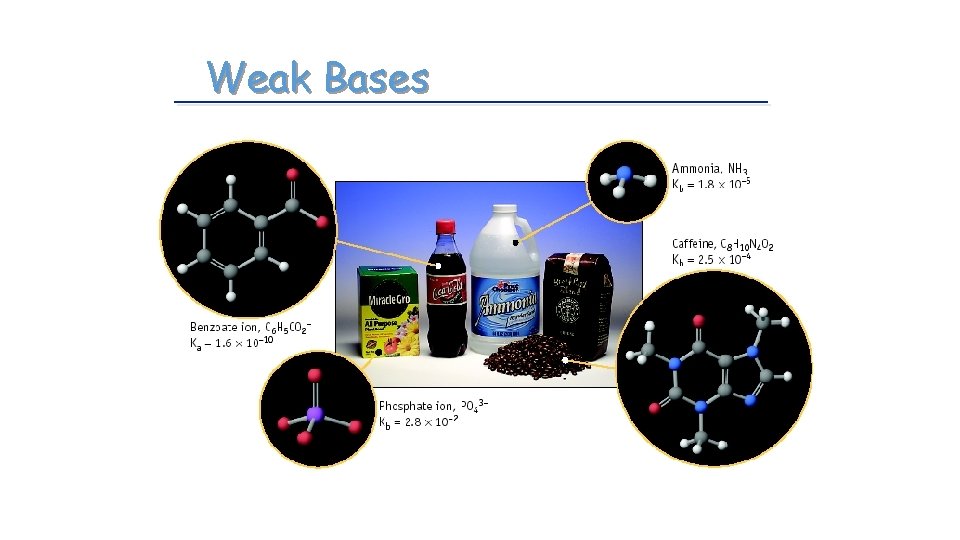
Weak Bases

Warm-Up 12/13/16 SWBAT write practice summary. We will take the multiple choice test tomorrow. We need more time for the practice summary and tomorrow is a short day.

Words you should be using in your summary: • Depends on • Determines • Known as • Caused by • According to • Because of • The effect of • Advice: • Keep it formal! It should look like it came from a text book not a text. • Write it out, don’t abbreviate! • Don’t be afraid to use a word more than once! • Whenever and wherever you can, EXPLAIN WHY AND/OR HOW • Constantly remind yourself of the topic you are writing about, this is not a list of definitions it is a summary about chemical reactions.

12/14/16 Multiple Choice Test

12/15/16 Summary Test
- Slides: 116