WARM UP Take out your notebook Write the

WARM UP Take out your notebook. Write the Unit topic (Chemistry Matters) on a new page. Copy down these topic objectives. UNIT 1 OBJECTIVES: At the end of this unit I will be able to: � Use appropriate significant figures and scientific notation. � Explain the use of the metric system and perform conversions. � Categorize objects based on the type of matter. � Describe the basic structure of atoms.

UNIT 1 - CHEMISTRY MATTERS

AGENDA ESSENTIAL QUESTION: In what ways are basic mathematic al concepts used in Chemistry? ? Warm Up Diagnostic Test Analysis Significant Figures Scientific notation The Metric System Exit Quiz

ANNOUNCEMENTS Remember my expectations of you… You will get the Unit 1 Homework Packet (Part 1) today. It is due next Wednesday 2. 3. 16

Diagnostic Test Analysis Diagnostic tests give teachers an idea of what you already know. We will be tracking your data throughout this class. Today I will show you how to use the test tracker. Follow closely, so that you know exactly what to do. CORRECT = INCORRECT =

Unit 1 – Subtopic 1: MATH FOR SCIENCE - Significant Figures - Scientific Notation *** Remember NOTES are in blue!***
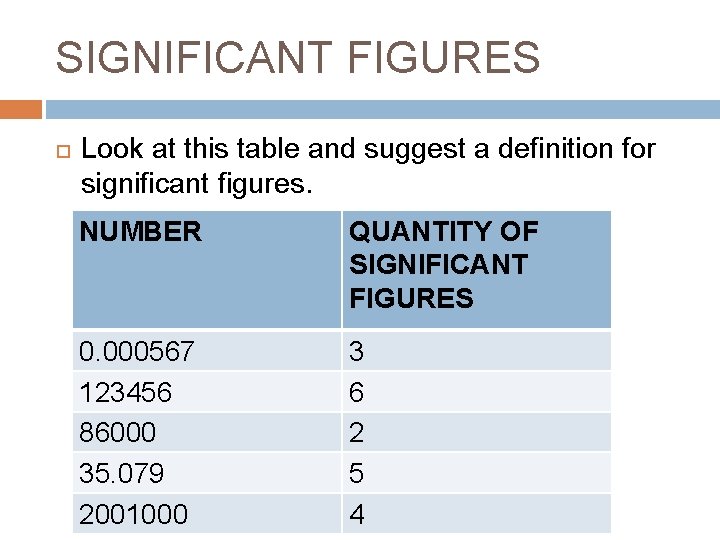
SIGNIFICANT FIGURES Look at this table and suggest a definition for significant figures. NUMBER QUANTITY OF SIGNIFICANT FIGURES 0. 000567 123456 86000 35. 079 2001000 3 6 2 5 4

Significant Figures Definition: Numerals that affect the value of a number when written in scientific notation. Rules: � All numerals from 1 -9 are significant. � Zeroes are significant if they are between two significant figures. This is important when doing calculations in science – � Always write your answer based on the component of your problem with the least

Let’s practice: How many significant figures are there in each of these numbers? 1. 2. 3. 4. 5. 3. 461728 4. 091 0. 97001 5. 2631 3. 461728 + 4. 091 + 0. 97001 + 5. 2631 (Solve the problem and then write the answer to the correct number of significant figures) Check your answers at the solution station when you are done then return to your seats.

Let’s practice (your turn!): How many significant figures are there in each of these numbers? 1. 2. 3. 43705 0. 0663 363587. 006 + 433. 12 + 68. 03 (Solve the problem and then write the answer to the correct number of significant figures) Check your answers at the solution station when you are done then return to your seats.

WARM UP 1. 2. What are the 2 rules for significant figures? How many significant figures are there in: 1. 2. 3. 4. 3. Answer these questions on the same page you used for Monday’s Warm up. 90210 9002. 010 0. 009021 90. 21000 Solve these equations: 1. 2. 36. 35 +3. 65 +3005. 6 4. 78 + 4. 0078 + 0. 00478

AGENDA ESSENTIAL QUESTION: In what ways are basic mathematic al concepts used in Chemistry? ? Warm Up Math for Chemistry Matter Physical & Chemical Change Exit Quiz Home Work

SCIENTIFIC NOTATION How much money would you like to have in your entire lifetime? (Write down that amount in numerals in your notebook. )

Scientific Notation There is an easier way to write very large numbers and very small numbers – they can be written in scientific notation. Scientific notation is expressed in this way: �A x 10 B � Where A is a number greater than or equal to 1 but less than 10. (Therefore it is a number that only has 1 significant figure before the decimal point) B is an exponent of 10. For example: � 0. 0000437 = 4. 37 x 10 -5 � 165, 000, 000 = 1. 65 x 1011

Scientific Notation To change numbers less than 1 to scientific notation: � Move the decimal point to the right until there is only one significant figure in front of the decimal point. This new number will be “A”. � Count the number of places you have moved your decimal point. Place a negative sign in front of this number and make it your exponent of 10 (B). � For example: 0. 0004835 = 4. 835 x 10 -4

Scientific Notation To change numbers greater than 1 to scientific notation: � Move the decimal point to the left until there is only one significant figure in front of the decimal point. This new number will be “A”. � Count the number of places you have moved your decimal point. Make this number your exponent of 10 (B). � For example: 4835000 = 4. 835 x 106
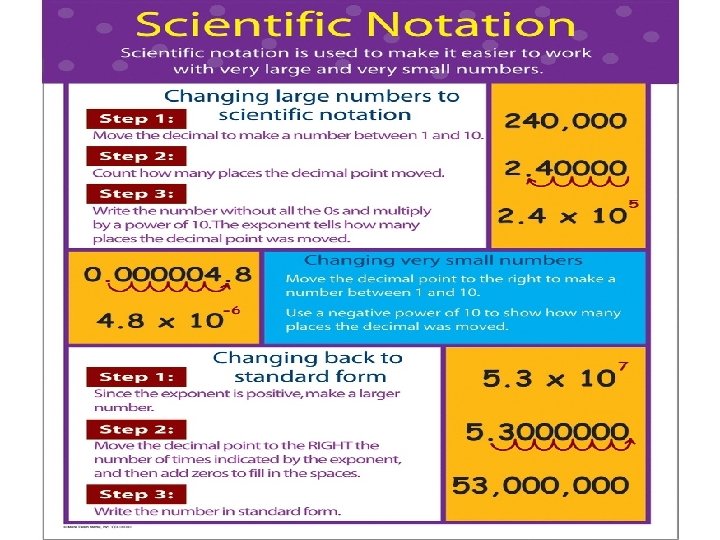

Let’s practice: Write these numbers in scientific notation. 1. 2. 3. 4. 5. 56000 40765 0. 0328 0. 000831 910. 0248

WARM UP – DAY 2 (Do all warm ups for this Unit on the same sheet of paper) What are three guidelines to follow in this class? Why is it sometimes necessary to write numbers in scientific notation? When adding numbers, you should always make sure that your answer has the same amount of significant figures that matches the number with the A. B. smallest / largest significant figures (choose one)

AGENDA ESSENTIAL QUESTION: In what ways are basic mathematic al concepts used in Chemistry? ? Warm Up Announcements The Metric System Classification of Matter Exit Quiz

ANNOUNCEMENTS Unit 1 Homework Packet (Part 1) is due next Wednesday 2. 3. 16 You will have a Quiz next Tuesday / Wednesday (depending on how far we get to). You will have a test next Friday / the Monday after that. Turn in the yellow packet (if you haven’t done so already).

Unit 1 – Subtopic 2: THE METRIC SYSTEM - What is the Metric System - Metric Conversions

THE METRIC SYSTEM
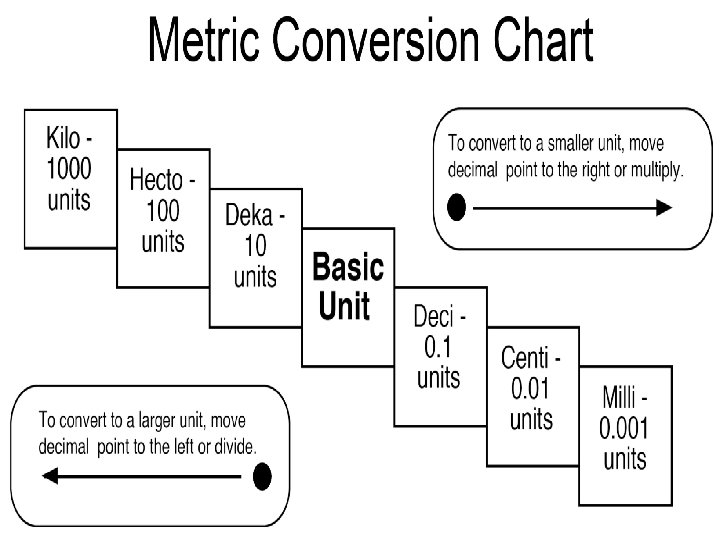
The Metric Staircase
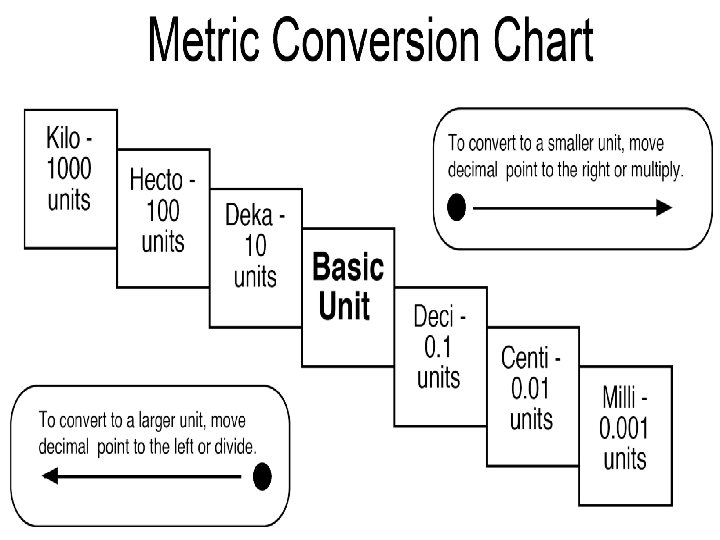
The Metric Staircase

The Metric System This is a system used internationally for measuring items. The Metric System relies on base units for every type of measurement that exists. Each measurement can be converted into smaller or larger units by multiplying or dividing by 10 and adding an appropriate prefix to the base unit.

The Metric System Conversions To go from smaller units to larger units, divide by 10 each step of the staircase. � For example: Convert 36000 mm to m. From mm to m, we take 3 steps. Therefore we must divide 36000 by 10, 3 times. 36000/10/10/10 = 36000 / 103 = 36000/1000 = 36 m. To go from larger units to smaller units, multiply by 10 for each step of the staircase. � For example: How many liters are there in 45 k. L? From k. L to L, we take 3 steps. Therefore we must multiply 45 by 10, 3 times. 45 x 10 x 10 = 45 x 103 = 45 x 1000 = 45000 L.
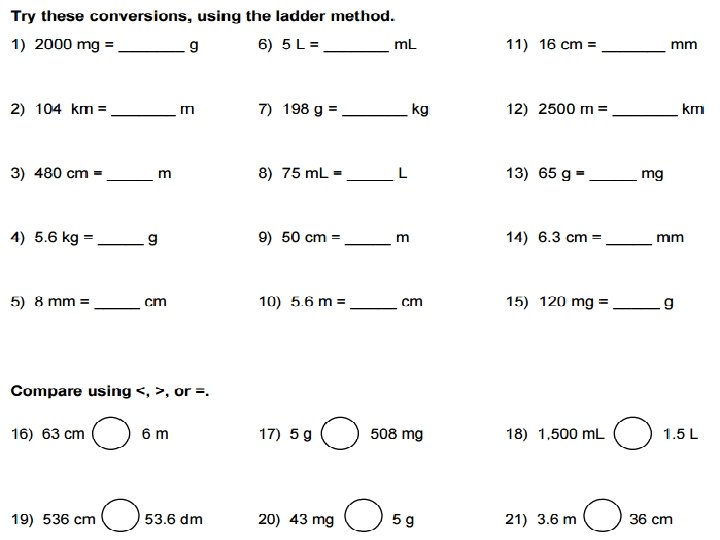

RESEARCH: Work individually or in pairs to prepare a product summarizing the Metric System. You should include: � What is the Metric System? � A mnemonic device to help you remember the Metric System. � What are the metric base units used for each of these types of measurements? Mass, Length, Volume, Time, Pressure, Temperature, Energy � Which metric unit (prefix included) would you use to measure: A Pencil

EXIT QUIZ Convert to scientific notation: � 999. 8865 � 0. 0003405 How many significant figures are there in: � 999. 8865 � 0. 0003405 How many centigrams are there in 237 decagrams?

WARM UP Convert to scientific notation: � 999. 8865 � 0. 0003405 How many significant figures are there in: � 999. 8865 � 0. 0003405 How many centigrams are there in 237 decagrams?

Unit 1 – Subtopic 3: MATTER - What is Matter? - Types of Matter

MATTER Matter is anything that has mass and takes up space. There are three states of matter: � Solid Particles closely arranged, little kinetic energy Volume and shape are constant. � Liquid Particles loosely arranged, medium kinetic energy Volume is constant, shape varies based on container � Gas Particles very loosely arranged, high kinetic energy Volume and shape vary

CLASSIFICATION OF MATTER Sort these items into four groups. Make sure to give a reason for placing each item into the group you placed it in. � Chocolate chip cookie � Gold � Carbon dioxide � Kool aid � Water � Copper � Air � Soil � Pizza

Classification of Matter Scientists also place items into groups. In Biology, we classify living things into the major kingdoms and then smaller groups. In Chemistry, we classify (sort / group) items based on the type of matter. � Pure Substances Elements Compounds � Mixtures (Impure substances) Homogenous mixtures Heterogeneous mixtures

Classification of Matter Elements – these are made up of one type of atom. They are found on the Periodic Table.

Classification of Matter Compounds – These are made from more than one atom and arranged in a particular formula.

Classification of Matter – Homogeneous Mixtures – These do not have a specific chemical formula, but they look the same throughout.

Classification of Matter – Heterogeneous Mixtures – These do not have a specific formula and may look different from one section to the next.

Classification of Matter Use your notes and this concept map to write 5 sentences about matter. Give definitions of the four major types of matter, and examples of each one.

YOUR TURN Sort these items into the four types of matter. � Chocolate chip cookie � Gold � Carbon dioxide � Kool aid � Water � Copper � Air � Soil � Pizza

WARM UP 1. How many significant figures are there in: 1. 2. 3. 4. Answer these questions on the same page you used for Friday’s Warm up. 500. 0046 37. 12 900. 8 + 809 (What is the correct answer? ) Convert 12000 cg to kg What classification of matter is made up of more than one type of atom in a specific chemical formula? Describe solids in terms of their volume and particle movement.

AGENDA ESSENTIAL QUESTION: In what ways can matter change from one form to another? Warm Up Announcements Science Career Monday Physical & Chemical Change Family Style Lab Exit Quiz - Trashketball

ANNOUNCEMENTS Upcoming Events: � Unit 1 Quiz tomorrow � Unit 1 Homework due Wednesday 2. 3. 16 � Unit 1 Test Friday 2. 5. 16 Turn in the yellow packet (if you haven’t done so already).

SCIENCE CAREER MONDAY: PHARMACOLOGIST Pharmacologists study the effects of drugs and other chemical substances on cells, animals, humans and the environment. As a pharmacologist you would carry out research to discover and develop new drugs and medicines, and to make sure they are used safely. Work hours: 37 -40 hrs / week Salary ranges from 40000 – 114000 per year Skills needed: Problem solving; Analytical & Measurement Skills Basic Entry Requirements: B. Sc. In Chemistry / Biochemistry

Unit 1 – Subtopic 4: PHYSICAL & CHEMICAL CHANGES - What are physical & chemical changes? - How do you know what type of change has occurred?

Physical & Chemical Changes Substances can be changed into a different form. The change may be physical or chemical.

Physical Changes A substance that undergoes a physical change ends up being the same substance. It is indicated by: � Change in size � Change of state � Change of texture
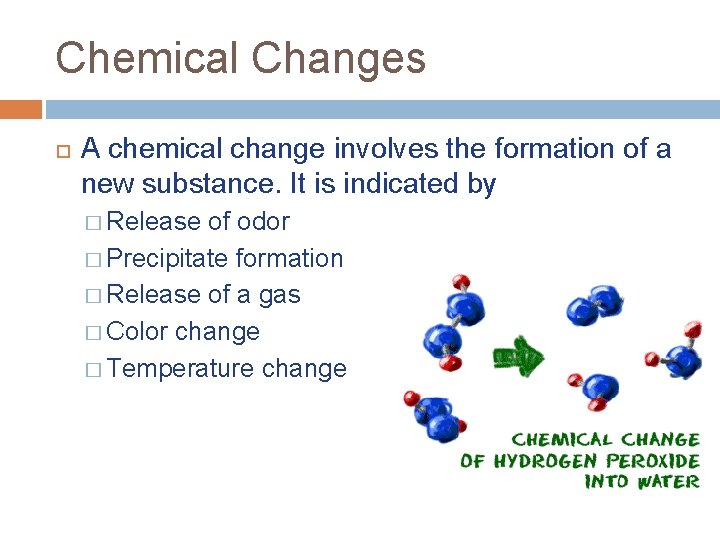
Chemical Changes A chemical change involves the formation of a new substance. It is indicated by � Release of odor � Precipitate formation � Release of a gas � Color change � Temperature change

Physical & Chemical Changes State whether each of these changes are physical or chemical. Give a reason for your answer.

Family Style Lab This lab will be a demo. You must turn in your observations sheet at the end of class today. Follow along and record the observations.

REVIEW & PRACTICE FOR QUIZ So far we learned: � Classroom � Lab guidelines Safety � The Scientific Method � Significant Figures � The Metric System � States of Matter � Classification of Matter � Physical & Chemical Changes

TRASHKETBALL!!!

Arrange three states of matter (solid, liquid, and gas) in order of DECREASING space between particles (greatest amount of space to leastamount of space).

Which state of matter has a fixed (the same) volume but not a fixed shape?

What classification of matter is this substance? �Copper (Cu)

What classification of matter is this substance? �Chicken noddle soup (with chicken and noodles)

What classification of matter is this substance? �H 2 NO 3

How many millimeters in 12 kilometers?

Why is boiling a physical change?

12. 53 + 0. 0003 + 56 = ?
Review & Practice For Quiz Station Review � Visit the stations for the topics you need some more clarity on. � Look at the examples and practice the questions.
Review & Practice For Quiz Kahoot Table Competition � Pull out your phones. � Log on to Kahoot. it

WARM UP 1. When you have completed your Warm Up, fill out the Learning Styles Survey on the back of your “facebook” sheet. Identify the physical and chemical changes, giving reasons for your answers: This summer, Alice’s dad put ground beef on the grill and made well-done burgers. Her brother cut slices of cheese to place on the burgers. Alice left her drink next to the grill and the ice cubes melted. 2. Place each of these into the four categories of matter: Pizza, silver, C 2 H 6, vanilla ice cream, Salt (Na. Cl), Special fried rice,

AGENDA ESSENTIAL QUESTION: What are atoms made of? ? Warm Up & Announcements Lab Completion Quiz Intro to the Atom Exit Quiz Home Work

ANNOUNCEMENTS Upcoming Events: � Unit 1 Quiz today � Unit 1 Homework due tomorrow 2. 3. 16 � Unit 1 Test Friday 2. 5. 16 Turn in the yellow packet (if you haven’t done so already).
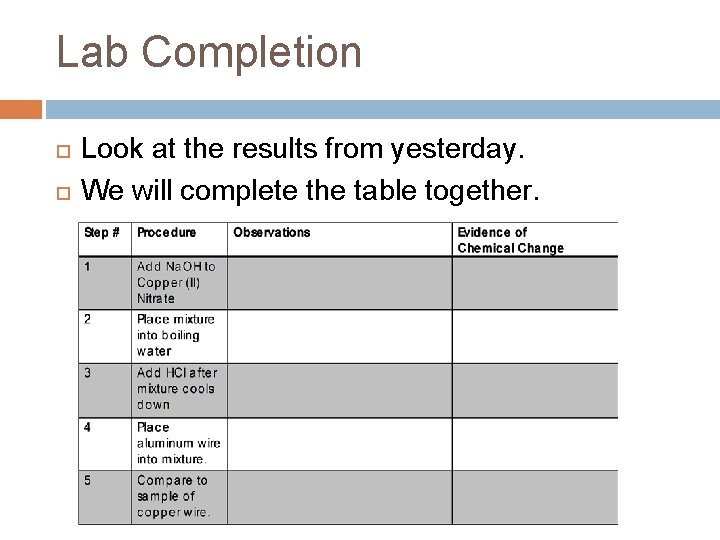
Lab Completion Look at the results from yesterday. We will complete the table together.

QUIZ 20 minutes No cellphones. Answer all questions on the paper. No communication between students. Do your best!

Unit 1 – Subtopic 5: BASIC ATOMIC STRUCTURE - What is an atom made of? - Arrangement of Subatomic Particles

History of the Atom How Small is the Atom?
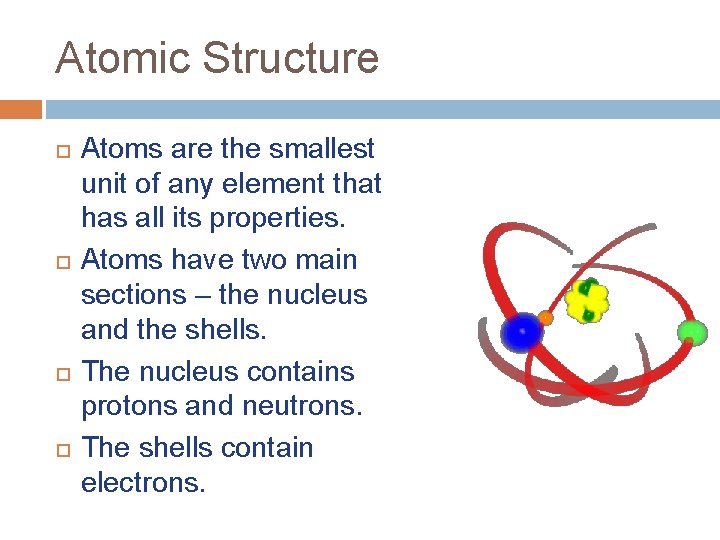
Atomic Structure Atoms are the smallest unit of any element that has all its properties. Atoms have two main sections – the nucleus and the shells. The nucleus contains protons and neutrons. The shells contain electrons.

Atomic Structure Electrons are negatively charged particles. Their mass is negligible (very close to 0). Protons are positively charged particles. They have a mass of 1 atomic mass unit (a. m. u. ). Neutrons are neutral – they have no charge. They have a mass of 1 a. m. u. Atoms have the same number of protons and electrons. This makes them neutral.
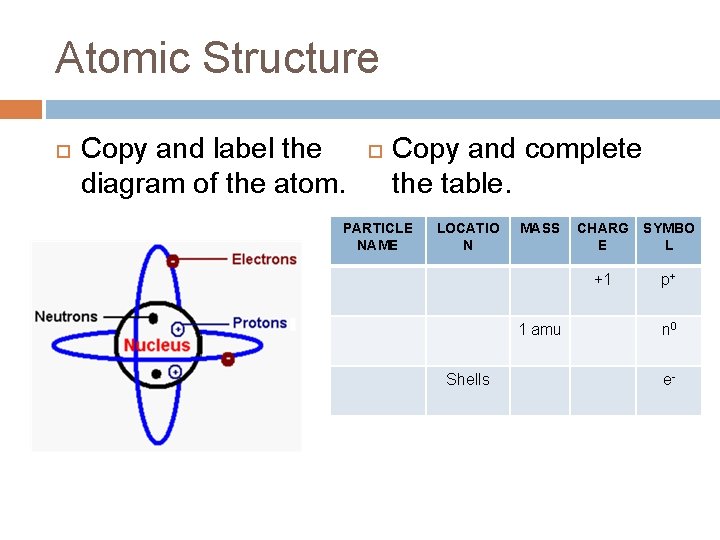
Atomic Structure Copy and label the diagram of the atom. Copy and complete the table. PARTICLE NAME LOCATIO N MASS 1 amu Shells CHARG E SYMBO L +1 p+ n 0 e-
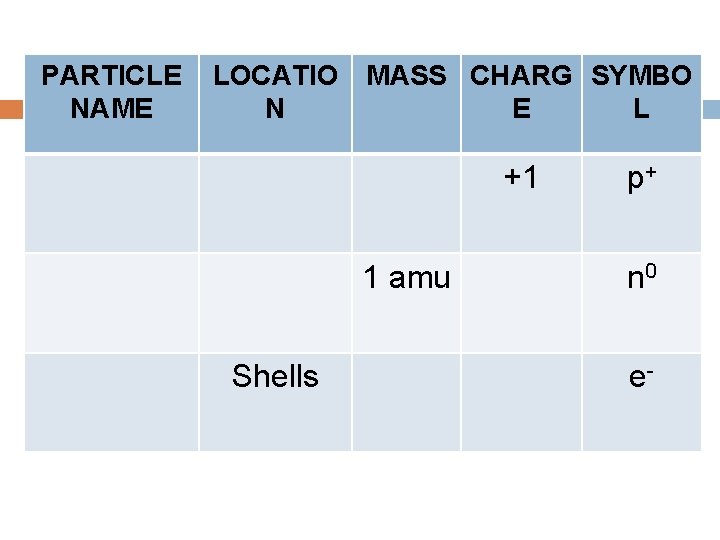
PARTICLE NAME LOCATIO MASS CHARG SYMBO N E L +1 1 amu Shells p+ n 0 e-

ATOMIC STRUCTURE History of the Atom Watch this video and write down the names of any 4 scientists that contributed to atomic structure. Create a product that: �States the contribution of your scientist to atomic structure. (at least 5 points) �Contains a picture of the scientist’s opinion of what the atom looks like.

HISTORY OF THE ATOM

Exit Quiz 1. 2. 3. 4. Name three subatomic particles. In a neutral atom, there always equal amounts of _____ and _____. The mass of the atom comes from the nucleus where _____ and _____ are found. The electrons are located in ______ around the nucleus.

Warm Up 1. 2. 3. 4. 5. 6. Define the word “atom”. Neutrons, protons and electrons are described as the three ______ particles. What two particles must be in equal amounts to make atoms neutral? What are the symbols used to represent each of the three particles found in the atom? Where are electrons found? The most dense region of the atom is the _______.

Unit 1 – Subtopic 6: ATOMIC STRUCTURE - Subatomic particles (recap) - Ions - Isotopes *** Remember NOTES are in blue!***

AGENDA ESSENTIAL QUESTION: What are atoms made of? ? Warm Up & Reminderss The Atoms – go deeper Isotopes & Ions Allotropes Exit Quiz Home Work

ANNOUNCEMENTS Home Work �Due TODAY – Turn it in!!! Check Quiz Grade online – retakes are possible. Unit 1 Test Friday
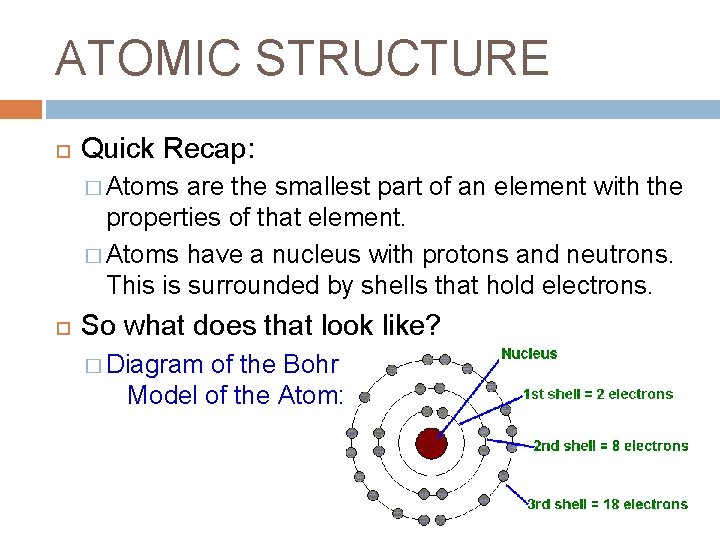
ATOMIC STRUCTURE Quick Recap: � Atoms are the smallest part of an element with the properties of that element. � Atoms have a nucleus with protons and neutrons. This is surrounded by shells that hold electrons. So what does that look like? � Diagram of the Bohr Model of the Atom:
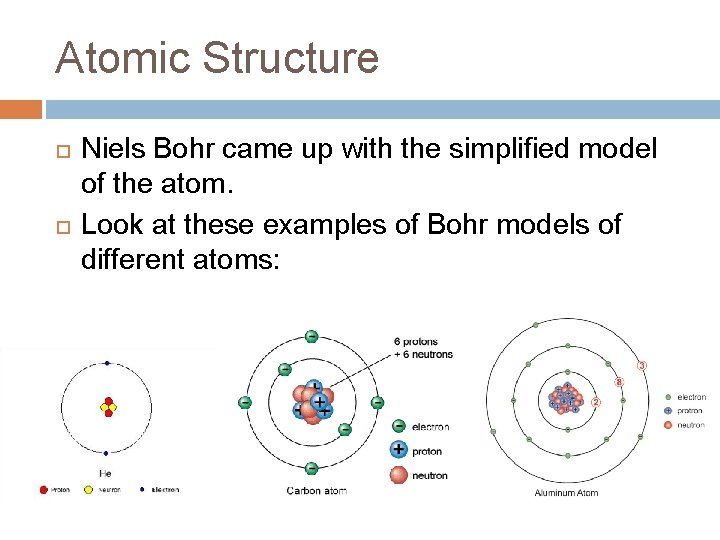
Atomic Structure Niels Bohr came up with the simplified model of the atom. Look at these examples of Bohr models of different atoms:
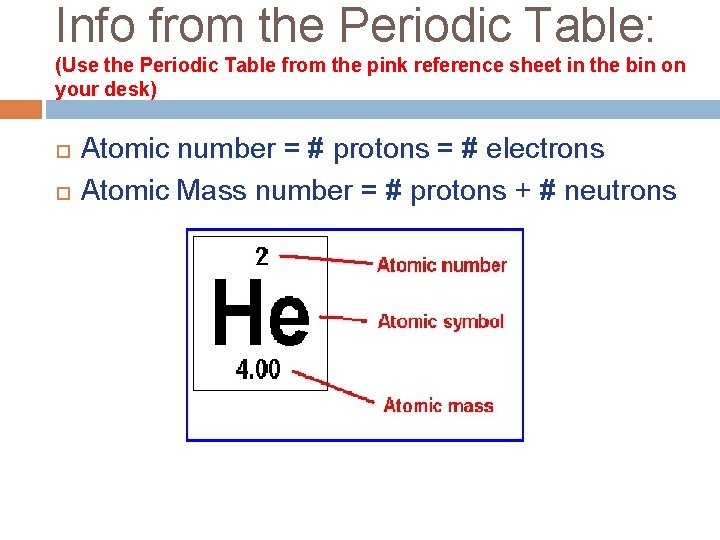
Info from the Periodic Table: (Use the Periodic Table from the pink reference sheet in the bin on your desk) Atomic number = # protons = # electrons Atomic Mass number = # protons + # neutrons
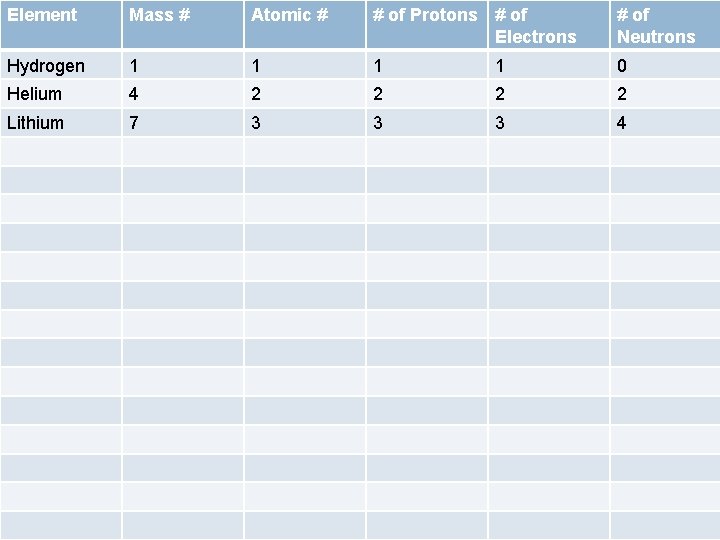
Element Mass # Atomic # # of Protons # of Electrons # of Neutrons Hydrogen 1 1 0 Helium 4 2 2 Lithium 7 3 3 3 4

Atomic Structure: Let’s practice drawing the Bohr model of these elements: R E M B E R Hydrogen Sodium Phosphorus Sulfur Boron Chlorine
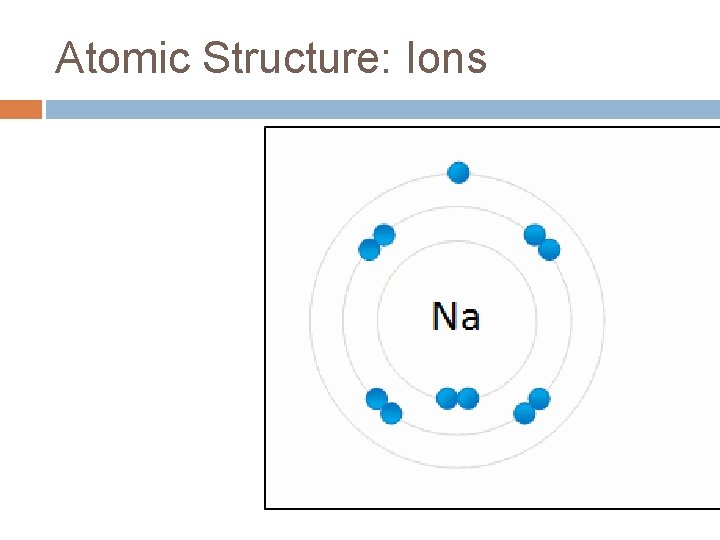
Atomic Structure: Ions
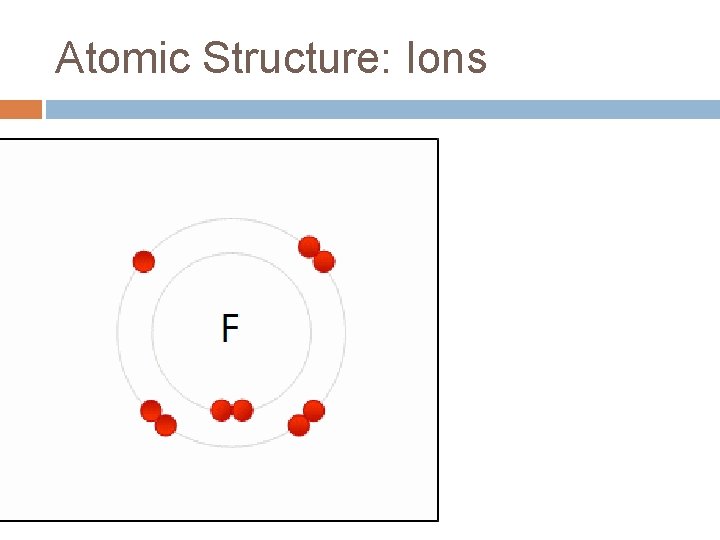
Atomic Structure: Ions

Atomic Structure: Ions are atoms that have lost or gained electrons. They do this to achieve a noble gas configuration to be more stable. They have unequal numbers of positively and negatively charged particles.

Atomic Structure: Ions This causes them to have a charge attached to their symbol. This charge is referred to as the oxidation number. �If an atom loses electrons, it will have a positive charge (cation). Eg. Mg+2 is a Magnesium ion with 12 protons but 10 electrons. �If an atom gains electrons, it will have a negative charge (anion). Eg P-3 is a Phosphorus ion with 15 protons but 18 electrons.

Warm Up Define the terms: 1. 2. 343. 05 x 63. 9 Sara weighs 75 kg. How many grams does she weigh? Give an example of each of the following: 2. 3. 4. 1. 2. 3. 4. 5. Cation Anion Heterogeneous mixture Homogeneous mixture Compound Element What are the 2 regions of the atom? What is found in each region?

AGENDA ESSENTIAL QUESTION: What are atoms made of? ? Warm Up & Announcements Isotopes & Ions Allotropes Density Exit Quiz Home Work – Study for Unit 1 Test

ANNOUNCEMENTS Home Work �OVERDUE – Turn it in!!! Check Quiz Grade online – retakes are possible. �Retake options During A-Lunch (Thursday / Friday) During tutoring (After school Monday / Tuesday) Unit 1 Test Friday
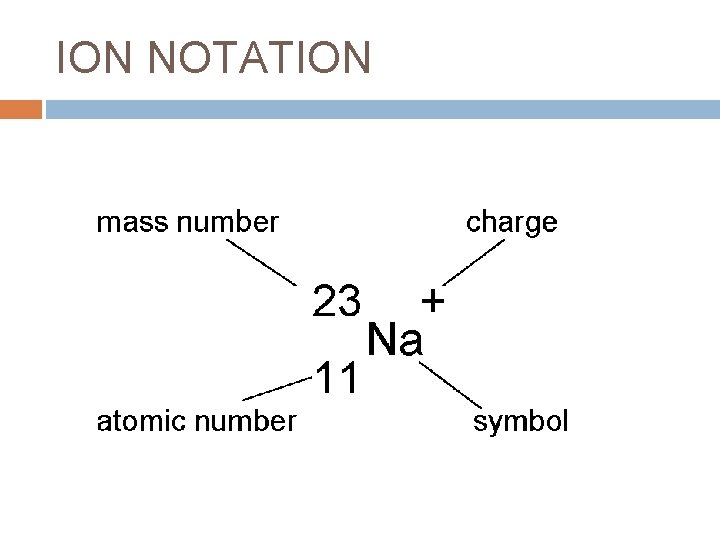
ION NOTATION
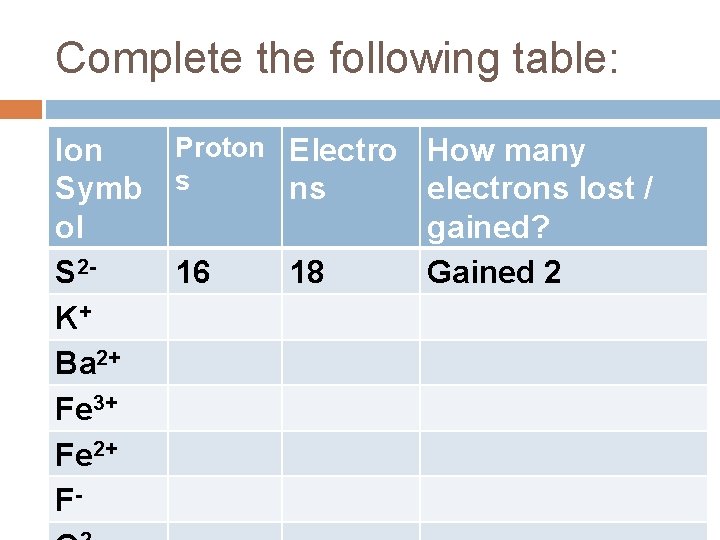
Complete the following table: Ion Symb ol S 2 K+ Ba 2+ Fe 3+ Fe 2+ F- Proton Electro s ns 16 18 How many electrons lost / gained? Gained 2
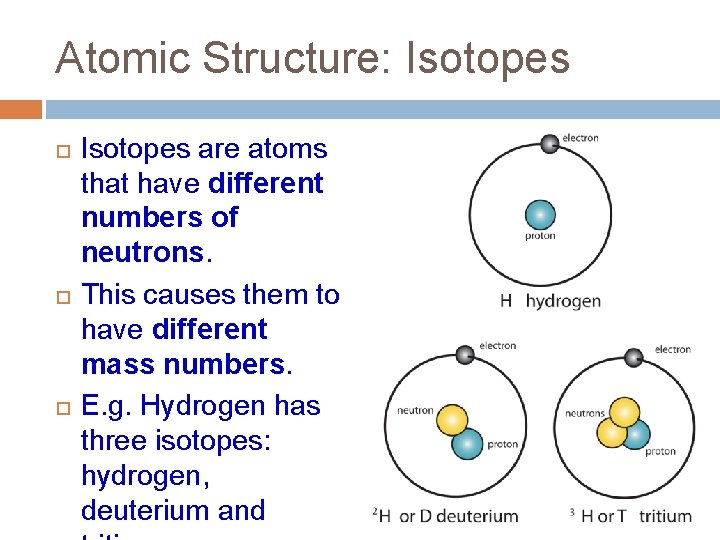
Atomic Structure: Isotopes are atoms that have different numbers of neutrons. This causes them to have different mass numbers. E. g. Hydrogen has three isotopes: hydrogen, deuterium and
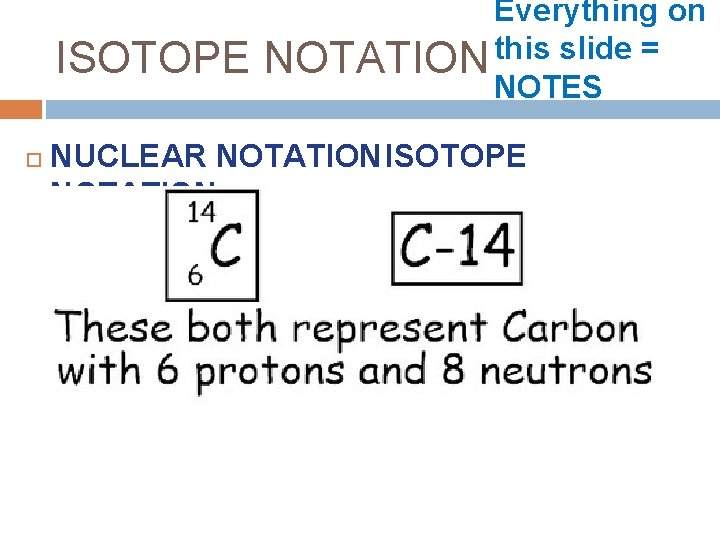
ISOTOPE Everything on this slide = NOTATION NOTES NUCLEAR NOTATIONISOTOPE NOTATION
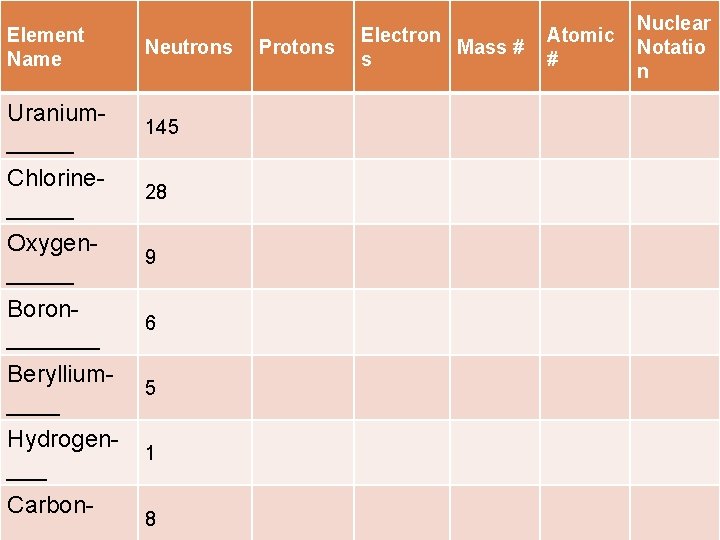
Element Name Neutrons Uranium_____ 145 Chlorine_____ 28 Oxygen_____ 9 Boron_______ 6 Beryllium____ 5 Hydrogen___ 1 Carbon- 8 Protons Electron Mass # s Atomic # Nuclear Notatio n
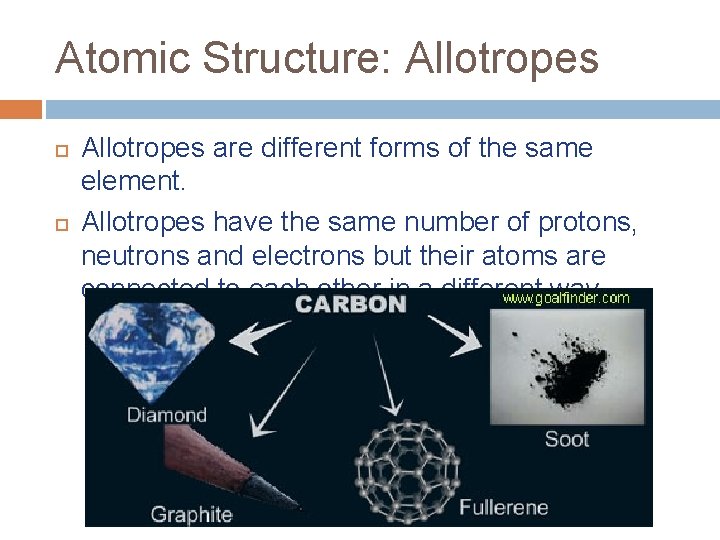
Atomic Structure: Allotropes are different forms of the same element. Allotropes have the same number of protons, neutrons and electrons but their atoms are connected to each other in a different way.

DIFFERENTIATING BETWEEN ATOMS, IONS & ISOTOPES Work with your group to: �Create a paper plate model of the ATOM on your paper. �Draw a diagram of the isotope on your paper. �Describe how many electrons the ion given has lost or gained. �Attach each item to a large sheet of construction paper labelled “Atom, Ion &

Atomic Structure: Let’s Practice Complete the ions and isotopes worksheet.

DENSITY
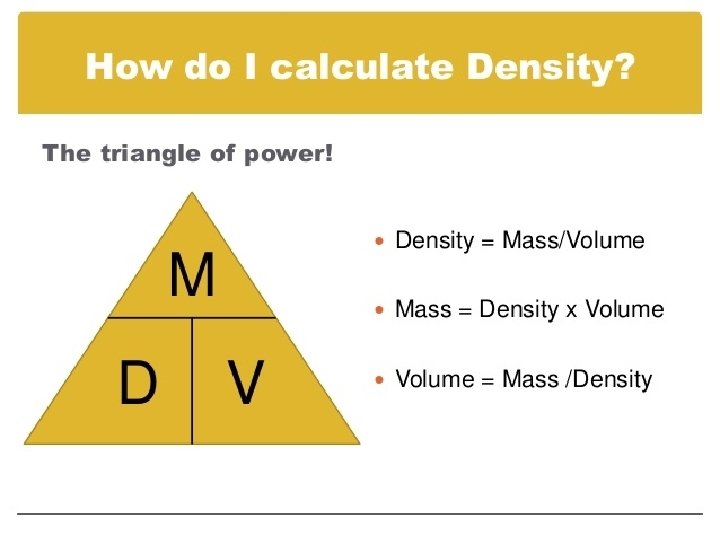

DENSITY Density is measured in g/m. L or g/cm 3 Mass is measured in g Volume is measured in m. L or cm 3 The more dense something is, the more likely it is to sink in water.

Density examples What is the density of a piece of wood that has a mass of 25. 0 grams and a volume of 29. 4 cm 3 A cup of gold colored metal beads was measured to have a mass 425 grams. The volume of the beads was calculated to be 48. 0 cm 3. Given the following densities, identify the metal. � Gold = 19. 3 g/m. L; Copper = 8. 86 g/m. L; Bronze = 9. 87 g/m. L I threw a plastic ball in the pool for my dog to fetch. The mass of the ball was 125 grams. What

Warm Up Define the terms: 1. 2. 343. 05 + 63. 9 Sara weighs 35 kg. How many centigrams does she weigh? Define each of the following: 2. 3. 4. 1. 2. 3. 4. 5. Isotope Allotrope Heterogeneous mixture Homogeneous mixture Compound Element What are the charges of each subatomic particle?

WARM UP What is the difference between: 1. 2. 3. 4. An atom and an ion? Anions and cations? An ion and an isotope? An isotope and an allotrope

AGENDA ESSENTIAL QUESTION: Have I mastered Unit 1? ? Warm Up & Reminders Station Review Kahoot! Home Work � Study for tomorrow’s test.

ANNOUNCEMENTS: Unit 1 Test Monday Tutoring: �Monday 2: 30 -3: 30 �Tuesday 2: 30 -3: 30 by appointment

DENSITY
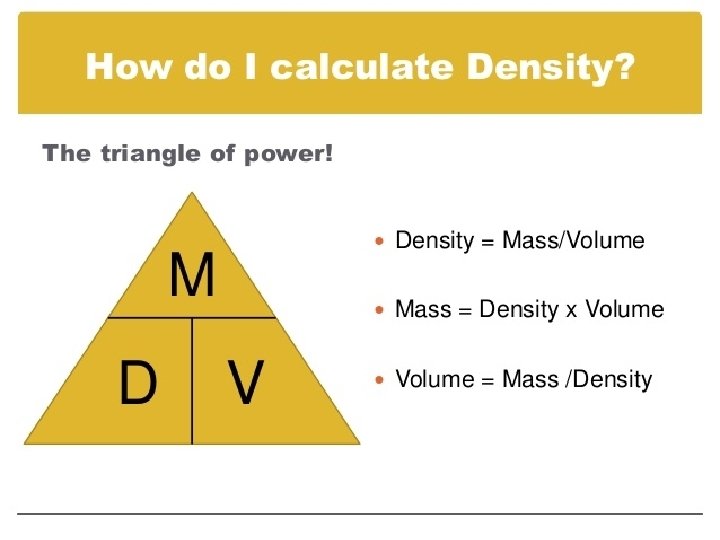

DENSITY Density is measured in g/m. L or g/cm 3 Mass is measured in g Volume is measured in m. L or cm 3 The more dense something is, the more likely it is to sink in water.

Density examples What is the density of a piece of wood that has a mass of 25. 0 grams and a volume of 29. 4 cm 3 A cup of gold colored metal beads was measured to have a mass 425 grams. The volume of the beads was calculated to be 48. 0 cm 3. Given the following densities, identify the metal. � Gold = 19. 3 g/m. L; Copper = 8. 86 g/m. L; Bronze = 9. 87 g/m. L I threw a plastic ball in the pool for my dog to fetch. The mass of the ball was 125 grams. What

UNIT 1 REVIEW Can you: 1. 2. 3. 4. Use appropriate significant figures and scientific notation? Explain the use of the metric system and perform conversions? Categorize objects based on the type of matter? Describe the basic structure of

Unit 1 Review – Stations (60 mins) Which area do you need the most work? � Visit every station starting with that topic. � Answer the questions at each station (5 questions from each station – do at least 30 questions). Then visit the other stations and complete the questions from each station. When the timer goes off, turn in the questions from each area for a grade.

SUPER WARM UP 1. 2. How are particles arranged in solids, liquids and gases? Jordan has a 25 c. L cup of soda. A. B. 3. 4. 5. 6. 7. How many significant figures is that? How many liters of soda is in his cup? What are the five indicators of chemical change? What are three indicators of physical change? What classification of matter is water (H 2 O)? Draw an atom of Fluorine. How many protons, neutrons & electrons? How many protons, neutrons and electrons are +2

AGENDA Warm up Multiple Intelligences Review Future Friday TEST After-test essay Home Work � Intro to the Periodic Table of the Elements Packet

MULTIPLE INTELLIGENCES REVIEW Collect your multiple intelligences survey. Identify your strength(s). Write down two things you should do to help you study based on your strength. Place your multiple intelligences survey and your two things in your binder / folder.

FUTURE FRIDAY PATCH PROGRAM Patch Website

TEST Write on the test paper. Show calculations. You may use a calculator. You may put one earphone in, but I should not hear your music. No communication between students. You may use your metric staircase. When you are done, complete your after-test essay.

HAPPY 3 -DAY WEEKEND! Do your home work! Relax and have fun New topic next week – The Electron and The Periodic Table of the Elements.
- Slides: 121