WARM UP Take out your notebook Write the

WARM UP Take out your notebook. Write the Unit topic (Molarity, p. H & Solubility) on a new page. Copy down these topic objectives. UNIT 8 OBJECTIVES: At the end of this unit I will be able to: Classify substances using the hydronium and hydroxide concentrations. Summarize the properties of acids and bases. Infer the quantitative nature of a solution (molarity, dilution, and titration with a 1: 1 molar ratio). Summarize the properties of solutions. Interpret solubility diagrams. Explain the solution process.

AGENDA EQ: What happens to the concentration of a solution when you change the amount of solute or solvent? ØWarm Up ØAnnouncements ØMolarity

ANNOUNCEMENTS § Important dates: § Friday 5. 20: U 8 Test § Thursday 5. 26: Last Day to turn in missing work § Wednesday 6. 1. 16: Practice NC Final § Friday 6. 3 – Thursday 6. 9: State testing; Review packets due on test days § Tutoring series resumes today. Sign up on class website. § Today: Last day to finish U 7 Test

ANNOUNCEMENTS § Today: Last day to finish U 7 Test (after school) § This Week: § Monday: Molarity § Tuesday: Solubility; U 8 Test Analysis § Wednesday: p. H (Acids & Bases) § Thursday: Review (with possible lab) § Friday: U 8 Quest (Quiz/Test); Time Management Activity
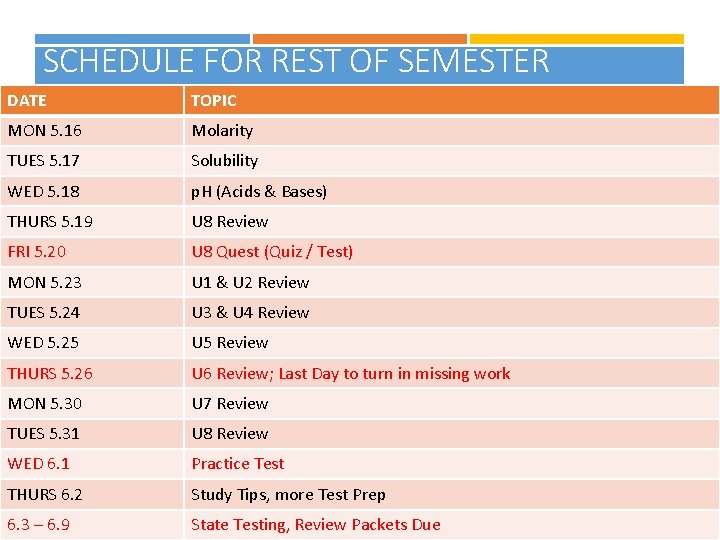
SCHEDULE FOR REST OF SEMESTER DATE TOPIC MON 5. 16 Molarity TUES 5. 17 Solubility WED 5. 18 p. H (Acids & Bases) THURS 5. 19 U 8 Review FRI 5. 20 U 8 Quest (Quiz / Test) MON 5. 23 U 1 & U 2 Review TUES 5. 24 U 3 & U 4 Review WED 5. 25 U 5 Review THURS 5. 26 U 6 Review; Last Day to turn in missing work MON 5. 30 U 7 Review TUES 5. 31 U 8 Review WED 6. 1 Practice Test THURS 6. 2 Study Tips, more Test Prep 6. 3 – 6. 9 State Testing, Review Packets Due

UNIT 8 Short unit featuring: 1 -2 labs Molarity p. H webquest and activity Solubility Quiz on Thursday

SOLUTIONS SOLUBILITY AND MOLARITY
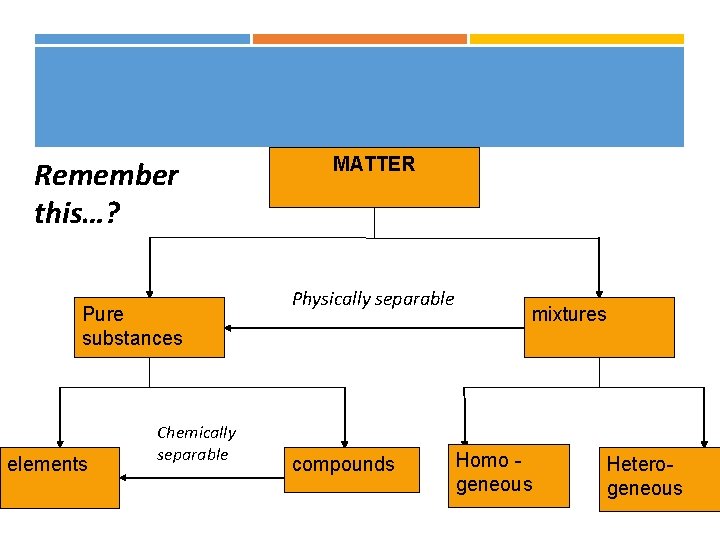
Remember this…? Pure substances elements Chemically separable MATTER Physically separable compounds mixtures Homo geneous Heterogeneous

SOME DEFINITIONS A solution is a homogeneous mixture of 2 or more substances in a single phase. One constituent is usually regarded as the SOLVENT and the others as SOLUTES.

PARTS OF A SOLUTION SOLUTE – the part of a solution that is being dissolved (usually the lesser amount) Solute Solvent Example solid Steel solid liquid Ocean Water liquid Vinegar gas liquid Carbonated Water SOLVENT – the part of a solution that dissolves the solute (usually the greater amount) Solute + Solvent = Solution SOLVATION- the process of making a solution (you know it better as “dissolving”)
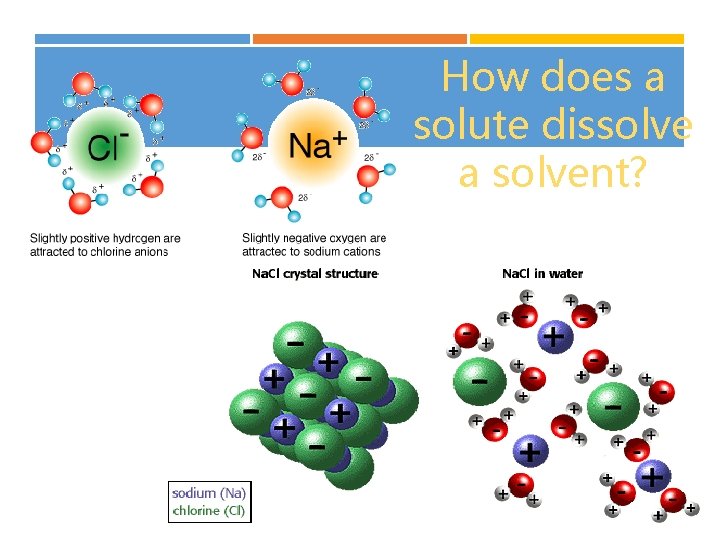
How does a solute dissolve a solvent?

LIKE DISSOLVES LIKE Polar (water, alcohol, etc…) will dissolve polar/charged molecules (salts, acids, bases, etc…) Non-polar (oils) dissolves non-polar For example… salt WILL NOT dissolve in oil!! Fats will NOT dissolve in water!

HOW DO WE “SOLVATE” THE PROBLEM? Water (as well as alcohol, another common solvent), is POLAR. It has a PARTIAL NEGATIVE (Oxygen) and PARTIAL POSITIVE (Hydrogen)

COLLIGATIVE PROPERTIES When adding a solute to a solvent, the properties of the solvent are modified. Melting point decreases Boiling point increases Vapor pressure decreases These changes are called COLLIGATIVE PROPERTIES. They depend only on the NUMBER of solute particles relative to solvent particles, not on the KIND of solute particles.

FACTORS THAT AFFECT THE RATE OF SOLVATION How would you make something dissolve faster? Agitation (shaking) Surface Area Temperature

WHAT IS MEANT BY THESE TERMS: 1. Solute 2. Solvent 3. Solution 4. Saturated 5. Unsaturated 6. Supersaturated
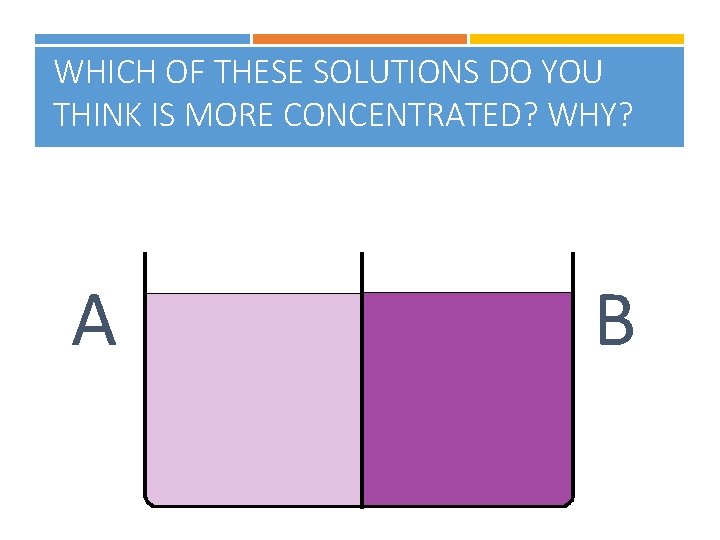
WHICH OF THESE SOLUTIONS DO YOU THINK IS MORE CONCENTRATED? WHY? A B

TYPES OF SOLUTIONS Three types of solutions Unsaturated- LESS THAN the maximum amount of solute that can be dissolved Saturated- THE MAXIMUM amount of solute that can be dissolved in a given amount of solvent at a given temperature Supersaturated- MORE THAN the maximum amount of solute that can be dissolved
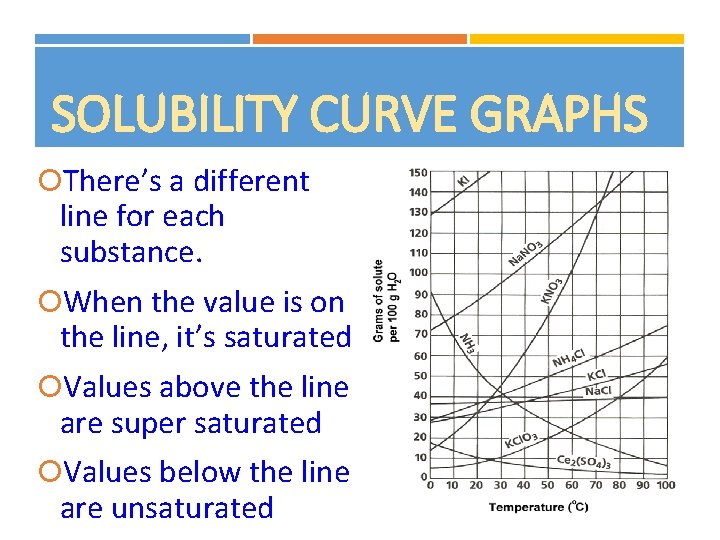
SOLUBILITY CURVE GRAPHS There’s a different line for each substance. When the value is on the line, it’s saturated Values above the line are super saturated Values below the line are unsaturated 19
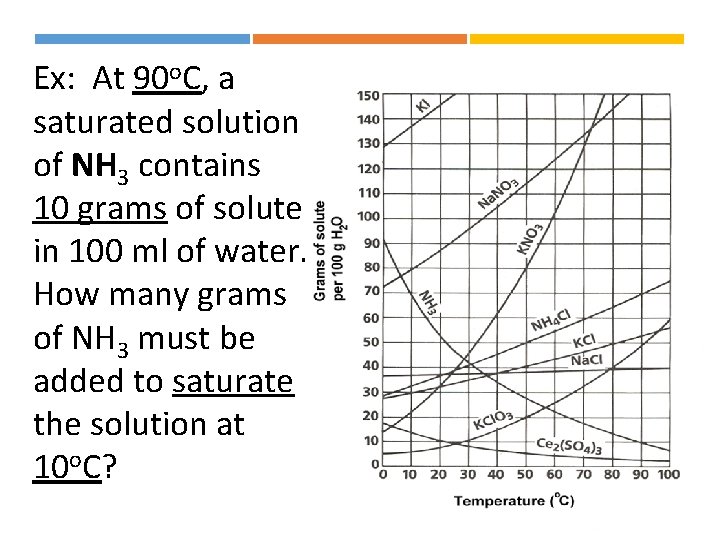
Ex: At 90 o. C, a saturated solution of NH 3 contains 10 grams of solute in 100 ml of water. How many grams of NH 3 must be added to saturate the solution at 10 o. C?

Ex: 30 g of KCl are dissolved in 100 m. L of water o at 45 C. Is this solution unsaturated, or supersaturated?
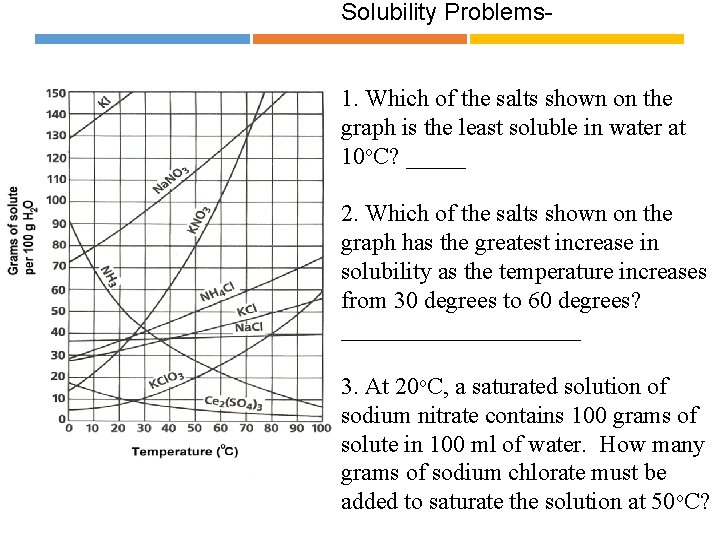
Solubility Problems 1. Which of the salts shown on the graph is the least soluble in water at 10 o. C? _____ 2. Which of the salts shown on the graph has the greatest increase in solubility as the temperature increases from 30 degrees to 60 degrees? __________ 3. At 20 o. C, a saturated solution of sodium nitrate contains 100 grams of solute in 100 ml of water. How many grams of sodium chlorate must be added to saturate the solution at 50 o. C? __________

NOW: Complete the Solutions / Solubility Worksheet. If you do not finish it in class, complete it for home work.

WARM UP 1. If I have 50 moles of Na. Cl in 65 L of water, what is my molarity? 2. If I have a 10 M solution of Na. OH in 20 L of water, how many moles do I have? 3. If I have 56 g of Mg(OH)2 in 10 L of water, what’s my molarity? (HINT: convert the grams to moles AND THEN solve!) 4. What are the constituents (parts) of a solution? Define them.

UNIT 7 TEST ANALYSIS Complete a data analysis sheet for this unit, and correct the sections you did not master. Staple your tracker to your test and turn it in to me for a grade.

WARM UP Do all Warm Ups for this week on the same paper!!! 1. How many moles are in 450 g of Mg. Br? 2. Will oil dissolve in water? Why or why not? 3. Will salt dissolve in water? Will it dissolve in oil? 4. When a substance melts, what happens to temperature? 5. Put the following elements in order of DECREASING electronegativity. Li, N, C, Be, F

ANNOUNCEMENTS § Important dates: § Friday 5. 20: U 8 Test § Thursday 5. 26: Last Day to turn in missing work § Wednesday 6. 1. 16: Practice NC Final § Friday 6. 3 – Thursday 6. 9: State testing; Review packets due on test days § Tutoring series is on today. Sign up on class website or don’t show up!!!

ANNOUNCEMENTS § This Week: § Monday: Solubility § Tuesday: Molarity; § Wednesday: p. H (Acids & Bases), U 8 Test Analysis, Updated progress reports § Thursday: Review § Friday: U 8 Quest (Quiz/Test); Time Management Activity
SCHEDULE FOR REST OF SEMESTER DATE TOPIC MON 5. 16 Solubility TUES 5. 17 Molarity WED 5. 18 p. H (Acids & Bases); Progress Reports THURS 5. 19 U 8 Review FRI 5. 20 U 8 Quest (Quiz / Test) MON 5. 23 U 1 & U 2 Review TUES 5. 24 U 3 & U 4 Review WED 5. 25 U 5 Review THURS 5. 26 U 6 Review; Last Day to turn in missing work MON 5. 30 U 7 Review TUES 5. 31 U 8 Review WED 6. 1 Practice Test THURS 6. 2 Study Tips, more Test Prep 6. 3 – 6. 9 State Testing, Review Packets Due

AGENDA EQ: What happens to the concentration of a solution when you change the amount of solute or solvent? ØWarm Up ØAnnouncements ØMolarity ØSolubility Curves
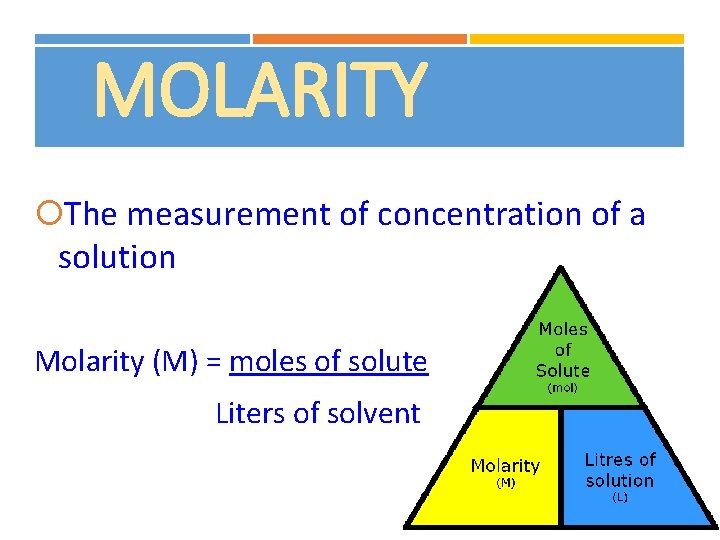
MOLARITY The measurement of concentration of a solution Molarity (M) = moles of solute Liters of solvent

MOLARITY If 2. 0 moles of Na. Cl are dissolved in 1 L of H 2 O what is the molarity of the solution? M = mol/L M = 2. 0 mol Na. Cl/1 L = 2. 0 M or 2. 0 mol/L

MOLARITY If 50 mol of HCl are dissolved in 75 L of H 2 O what is the molarity? How many moles of Ca. Cl 2 would be dissolved in 3. 0 L of a 0. 10 M solution of Ca. Cl 2?

BUT WAIT… What is the molarity of a solution that contains 50 g of Na. Cl dissolved in. 600 L of water?

YOU TRY… What is the molarity of a solution of Ca. Cl 2 if you add 650 g of it to a 4 L bottle of water? How many grams of KBr are in 3 L of a 5 M solution?

MOLARITY How do you change the molarity of a solution? ? Change the volume of the solution M 1 V 1 = M 2 V 2

MOLARITY If a 2. 0 L of a 2. 5 M solution is increased to 5. 0 L what is the new molarity? M 1 V 1 = M 2 V 2 M 1 = 2. 5 M V 1 = 2. 0 L M 2 = ? V 2 = 5. 0 L

MOLARITY M 1 V 1 = M 2 V 2 (2. 5 M x 2. 0 L) = (5. 0 L)M 2 5. 0 M x L/ 5. 0 L = M 2 1. 0 M = M 2

MOLARITY What was the original volume of a 2. 0 M solution if it has been changed to 5. 0 L and 1. 0 M? What is the new molarity of a solution that has been diluted from 100 m. L and 1. 0 M to 250 m. L?

EXIT QUIZ If I have 50 moles of Na. Cl in 65 L of water, what is my molarity?

NOW: Complete the Molarity Worksheet. Show it to me for a signature before the end of class.

EXIT TICKET 1. What is the molarity of a solution that contains 100 g of Na. Cl dissolved in 5 L of water? 2. What was the original volume of a 2. 0 M solution if it has been changed to 5. 0 L and 1. 0 M? 3. What is something that changes the freezing point of a solution?

WARM UP Do all Warm Ups for this week on the same paper!!! 1. How many grams of Na. OH do I need to produce a 5 M solution if I have 10 L of water? 2. Think about it: Does the solubility of solids increase or decrease with increased temperature? 3. For the reaction C + 2 H 2 CH 4 how many grams of hydrogen are required to produce 10 grams of methane (CH 4)? 4. Define deposition. 5. Write down the name of V 2 S 5.

ANNOUNCEMENTS § Important dates: § Friday 5. 20: U 8 Test § Thursday 5. 26: Last Day to turn in missing work § Wednesday 6. 1. 16: Practice NC Final § Friday 6. 3 – Thursday 6. 9: State testing; Review packets due on test days § Tutoring series is on today. Sign up on class website or don’t show up!!!

ANNOUNCEMENTS § This Week: § Monday: Solubility § Tuesday: Molarity; § Wednesday: p. H (Acids & Bases), U 8 Test Analysis, Updated progress reports § Thursday: Review § Friday: U 8 Quest (Quiz/Test); Time Management Activity
SCHEDULE FOR REST OF SEMESTER DATE TOPIC MON 5. 16 Solubility TUES 5. 17 Molarity WED 5. 18 p. H (Acids & Bases); Progress Reports THURS 5. 19 U 8 Review FRI 5. 20 U 8 Quest (Quiz / Test) MON 5. 23 U 1 & U 2 Review TUES 5. 24 U 3 & U 4 Review WED 5. 25 U 5 Review THURS 5. 26 U 6 Review; Last Day to turn in missing work MON 5. 30 U 7 Review TUES 5. 31 U 8 Review WED 6. 1 Practice Test THURS 6. 2 Study Tips, more Test Prep 6. 3 – 6. 9 State Testing, Review Packets Due

AGENDA EQ: How do we differentiate between acids and bases? ØWarm Up ØAnnouncements Øp. H

ACIDS & BASES Have you ever been stung by an insect? Have you ever used drain cleaner? Have you ever taken an aspirin?

Consensus: Everything in the world has a p. H – either it is acidic, basic, or neutral

ACID/BASE EXPLORATION LAB Information sheet is at EVERY lab bench. It is an entire sheet of background information regarding what acids and bases are. The analysis questions on the lab sheet can be answered using the info sheet AND/OR your previous knowledge from this class. Follow the procedure outlined on your LAB HANOUT. Answer all the questions in COMPLETE SENTENCES

ACID/BASE EXPLORATION LAB SAFETY You must wear goggles AT ALL TIMES You must stay at your lab bench, even if you are not immediately doing something Be cautious—be sure not to splash the chemicals DO NOT EAT THE CHEMICALS Procedure If you need to leave your bench, raise your hand I will probably say yes. You may only ask me questions about the procedure and not about the content! You can do it!

Acids • Taste sour • Strong acids conduct electricity • Have a p. H between 0 -7 • Give off hydrogen (H+ ) ions in water • Low p. H; indicators turn red Examples Aspirin Soda HCl (hydrochloric acid) H 2 SO 4 (sulphuric acid) Vinegar

Bases • Taste bitter • Strong bases conduct electricity Examples (write down 3) baking soda Windex Na. OH (sodium hydroxide) KOH (potassium hydroxide) bleach • Have a p. H between 7 -14 • Give of hydroxide (OH-) ions • High p. H; indicators turn blue

PRACTICE 1. HF – acid or base? 2. Li. OH – acid or base? 3. Mg(OH)2 – acid or base? 4. HCl. O 3 – acid or base?
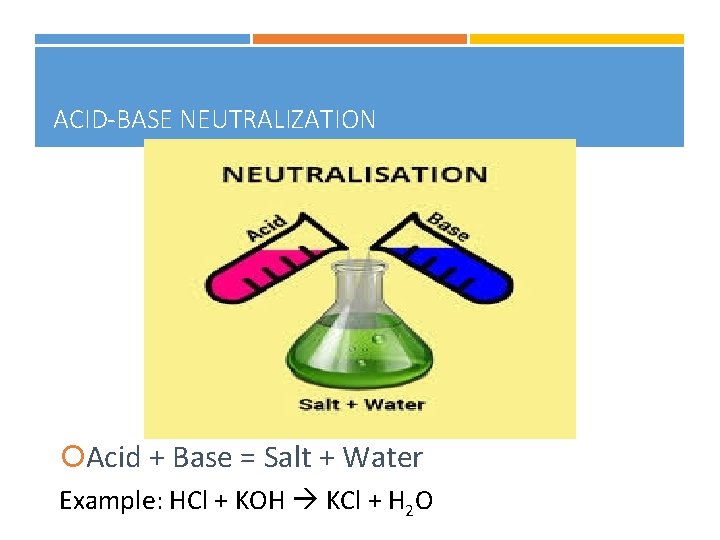
ACID-BASE NEUTRALIZATION Acid + Base = Salt + Water Example: HCl + KOH KCl + H 2 O

SO WHAT IS A PH OF 7? NEUTRAL!! Equal amounts of hydrogen (H+ ) and hydroxide (OH-) ions. Example: Distilled Water

INSTRUCTIONS: Work on the acid / base / p. H section of your worksheet. You should complete as many sections today as possible using the textbook and laptop you have been given. I will be calling you to meet with me throughout the block today. Please bring in any missing work.

EXIT QUIZ WARM UP Do all Warm Ups for this week on the same paper!!! 1. What is the new molarity of a solution that has been diluted from 1. 2 L and 4 M to 3. 0 L? 2. How many moles are in 30 L of a 0. 25 M Ni. Cl 2 solution? 3. What is an electrolyte? 4. _____ + _____ = solution 5. What are the 3 factors that affect the rate of solvation? 6. Saturated/Unsaturated? 20 g of Na. NO 3 at 20 o. C. 7. Colligative properties _____ melting point and ____ boiling point

AGENDA EQ: How do we differentiate between acids and bases? ØWarm Up ØAnnouncements ØComplete p. H activity ØReview

ANNOUNCEMENTS § Important dates: § Friday 5. 20: U 8 Test § Thursday 5. 26: Last Day to turn in missing work § Wednesday 6. 1. 16: Practice NC Final § Friday 6. 3 – Thursday 6. 9: State testing; Review packets due on test days § Tutoring series is on today. Sign up on class website or don’t show up!!!

ANNOUNCEMENTS § This Week: § Monday: Solubility § Tuesday: Molarity; § Wednesday: p. H (Acids & Bases), U 8 Test Analysis, Updated progress reports § Thursday: Completion of Worksheet; Review § Friday: U 8 Quest (Quiz/Test); Time Management Activity
SCHEDULE FOR REST OF SEMESTER DATE TOPIC MON 5. 16 Solubility TUES 5. 17 Molarity WED 5. 18 p. H (Acids & Bases); Progress Reports THURS 5. 19 U 8 Review FRI 5. 20 U 8 Quest (Quiz / Test) MON 5. 23 U 1 & U 2 Review TUES 5. 24 U 3 & U 4 Review WED 5. 25 U 5 Review THURS 5. 26 U 6 Review; Last Day to turn in missing work MON 5. 30 U 7 Review TUES 5. 31 U 8 Review WED 6. 1 Practice Test THURS 6. 2 Study Tips, more Test Prep 6. 3 – 6. 9 State Testing, Review Packets Due
![CALCULATING PH p. H = -log[H+] 0. 1 0. 002 0. 035 0. 98 CALCULATING PH p. H = -log[H+] 0. 1 0. 002 0. 035 0. 98](http://slidetodoc.com/presentation_image_h2/71235012e0f7189093fdad4273669aff/image-63.jpg)
CALCULATING PH p. H = -log[H+] 0. 1 0. 002 0. 035 0. 98 Formula p. H
![CALCULATING PH p. OH = -log [OH-] 0. 0001 0. 000001 0. 58 0. CALCULATING PH p. OH = -log [OH-] 0. 0001 0. 000001 0. 58 0.](http://slidetodoc.com/presentation_image_h2/71235012e0f7189093fdad4273669aff/image-64.jpg)
CALCULATING PH p. OH = -log [OH-] 0. 0001 0. 000001 0. 58 0. 179 0. 634 Formula p. OH
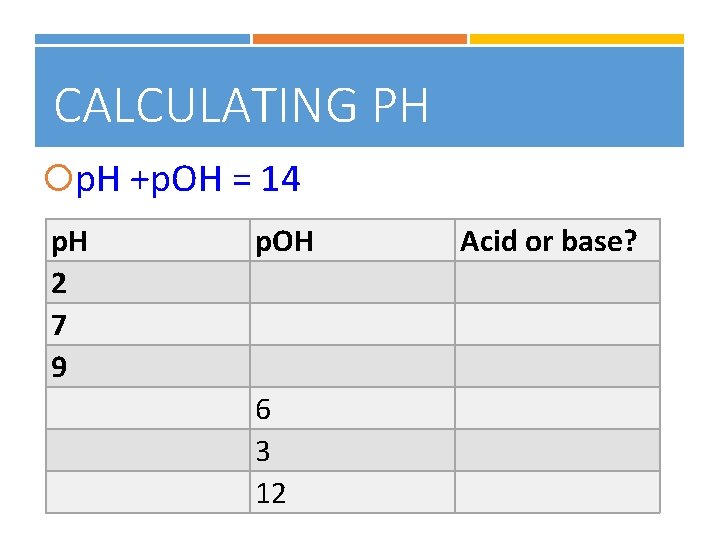
CALCULATING PH p. H +p. OH = 14 p. H 2 7 9 p. OH 6 3 12 Acid or base?

TITRATION CALCULATIONS A 0. 015 L sample of HCl is neutralized by 0. 03 L of 0. 994 M Na. OH. What is the molarity of HCl? 1. Calculate the missing info of substance 1 using the formula M= mol/L 2. Use the number of moles of Substance 1 with the info for substance 2 in the equation M = mol/L

TITRATION CALCULATIONS A 0. 05 L sample of a potassium hydroxide (KOH) solution is titrated with a 0. 8 M HCl solution. The titration requires 0. 028 L of HCl solution to be neutralized. What is the molarity of the KOH solution? 1. Calculate the missing info of substance 1 using the formula M= mol/L 2. Use the number of moles of Substance 1 with the info for substance 2 in the equation M = mol/L

TITRATION CALCULATIONS A 0. 012 L sample of NH 4 is neutralized by 1. 5 M HNO 3. A total of 0. 02 L of the acid is required to be neutralized. What is the molarity of the ammonia solution? 1. Calculate the missing info of substance 1 using the formula M= mol/L 2. Use the number of moles of Substance 1 with the info for substance 2 in the equation M = mol/L

WORK TIME You will have the rest of class to SILENTLY and INDEPENDENTLY work on the Unit 8 packet & Acid Base Worksheet All completed worksheets are due today. When you are done, complete the Review Activity with the people at your station (or on your own).

REVIEW ACTIVITY Prepare a product (Power. Point, poster, Song, etc) summarizing the Unit. You should include definitions, descriptions and examples of each subtopic in the unit including: Solubility: Polar / Nonpolar; Colligative properties; Solubility Curves, etc Molarity: (M = mol/L); (M 1 V 1 = M 2 V 2) p. H: Acids & Bases p. H: Calculating p. H: Titration Calculations (Honors only)

WARM UP Do all Warm Ups for this week on the same paper!!! 1. If I have 56 grams of Na. OH in 15 L of water, what is my molarity? 2. If I have 78 L of 10 M hydrochloric acid, how many grams of HCl do I have? 3. I have 18 moles of Na. Cl in 3 L of water. What is my molarity? 4. What are the 3 factors that effect solvation? 5. How is there both liquid water and frozen water at the poles?

AGENDA EQ: How do we differentiate between acids and bases? ØWarm Up ØAnnouncements ØComplete p. H activity ØReview

ANNOUNCEMENTS § Important dates: § Friday 5. 20: U 8 Test § Thursday 5. 26: Last Day to turn in missing work § Wednesday 6. 1. 16: Practice NC Final § Friday 6. 3 – Thursday 6. 9: State testing; Review packets due on test days § Tutoring series is on today. Sign up on class website or don’t show up!!!

ANNOUNCEMENTS § This Week: § Monday: Solubility § Tuesday: Molarity; § Wednesday: p. H (Acids & Bases), U 8 Test Analysis, Updated progress reports § Thursday: Completion of Worksheet; Review § Friday: U 8 Quest (Quiz/Test); Time Management Activity
SCHEDULE FOR REST OF SEMESTER DATE FRI 5. 20 MON 5. 23 TUES 5. 24 WED 5. 25 THURS 5. 26 TUES 5. 31 WED 6. 1 THURS 6. 2 6. 3 – 6. 9 TOPIC U 8 Quest (Quiz / Test) U 1 & U 2 Review U 3 & U 4 Review U 5 Review U 6 Review; Last Day for missing work U 7 & 8 Review Practice Test Study Tips, more Test Prep State Testing, Review Packets Due

TEST INSTRUCTIONS v. You may use calculators and periodic tables for this test. v. Refer to the whiteboard formulas. v. Do all work on the test paper. Take your time to answer each question carefully. v. Do not communicate with anyone other than me. v. You may not use your phone during the test but you may listen to music if it doesn’t disturb anyone (including me). v. When you are done, stick your hand up.

UNIT 8 QUEST TIME No talking, no cell phones, no wandering eyes If you have a question, raise your hand to get out of your seat and then you may come to the front of the room Do and be your best
- Slides: 77