VUR UTI and Antibiotic Prophylaxis How to Use

VUR, UTI, and Antibiotic Prophylaxis How to Use an Article About Therapy or Prevention Journal Club Amy K Evans PGY 2 August 15, 2006

The Case n Kali is a 14 mo female who presents to WRAMC ED with fever to 102. Your stellar Peds Intern suggests obtaining a UA/UCx, which results in the diagnosis of acute pyelonephritis.

The Case Kali is a 14 mo female who presents to WRAMC ED with fever to 102. Your stellar Peds Intern suggests obtaining a UA/UCx, which results in the diagnosis of acute pyelonephritis. n Kali is admitted to Wd 51 for 48 hrs of IV abx, then, afebrile, discharged to complete po course. n

The Case Kali is a 14 mo female who presents to WRAMC ED with fever to 102. Your stellar Peds Intern suggests obtaining a UA/UCx, which results in the diagnosis of acute pyelonephritis. n Kali is admitted to Wd 51 for 48 hrs of IV abx, then, afebrile, discharged to complete po course. n She undergoes renal US and VCUG 3 weeks later, which reveal grade II VUR on the left. n

The Question n Should we treat her prophylactically? n Short-term: n n Will this decrease recurrent infections? Long-term: Will this decrease renal scarring? n Why else would it matter? n
Background Vesicoureteral Reflux (VUR) n Primary – congenital incompetence of VU valve (shortened submucosal tunnel) n Secondary – multiple anatomic abnormalities n

Background Incidence 1 -10% n Siblings 30 -45% (3/4 asymptomatic) n Diagnosed via VCUG n UTI workup – 40% (girls); 70% (infants <1 yo) n Antenatal hydronephrosis – 9% (boys) n n Why worry? n VUR pyelonephritis renal scarring n HTN, renal insufficiency, ESRD, pre-eclampsia
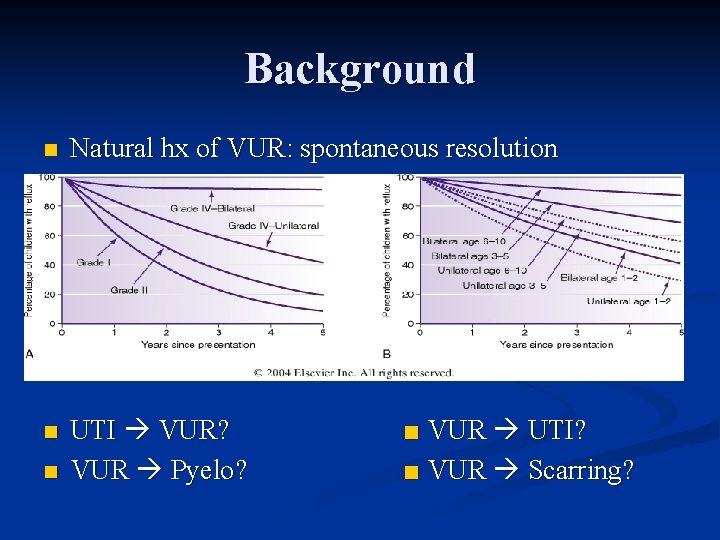
Background n Natural hx of VUR: spontaneous resolution n UTI VUR? VUR Pyelo? n ■ VUR UTI? ■ VUR Scarring?

Current Treatment Recs n Workup: Febrile UTI (any age) n UTI <5 yo n UTI x 2 in school-age girls n UTI in any boy n n To treat or not to treat? n Imaging: n Renal US n 40% sensitive (VUR) n VCUG n Diagnostic! n DMSA
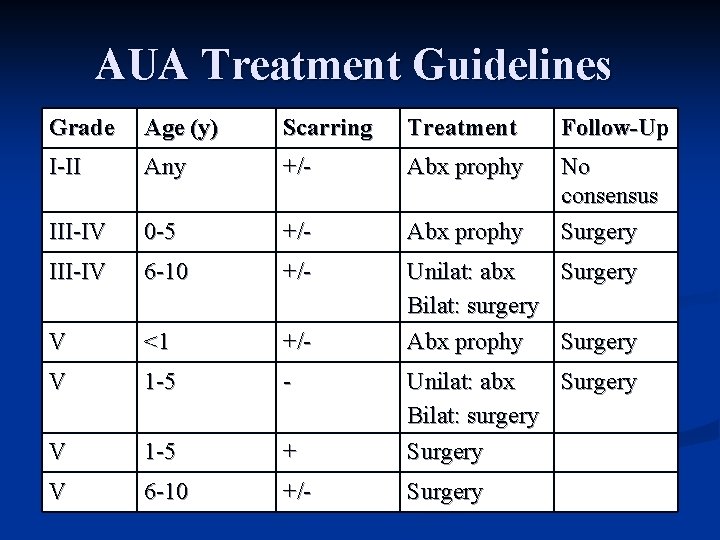
AUA Treatment Guidelines Grade Age (y) Scarring Treatment Follow-Up I-II Any +/- Abx prophy III-IV 0 -5 +/- Abx prophy No consensus Surgery III-IV 6 -10 +/- V <1 +/- Unilat: abx Surgery Bilat: surgery Abx prophy Surgery V 1 -5 - V 1 -5 + Unilat: abx Surgery Bilat: surgery Surgery V 6 -10 +/- Surgery

Current Treatment Recs n AUA Pediatric VUR Guidelines Panel (1997) n “The panel recommendations to offer continuous abx prophylaxis…are based on limited scientific evidence. To our knowledge controlled studies comparing the efficacy of continuous prophylaxis and intermittent therapy on health outcomes…have not been performed. ” No controlled studies? n Then what are we basing treatment on? n

The State of the Art n Williams et. al. (2001) Systematic review of RCTs on UTI/abx prophy n Five trials, 1968 -1978 n Best 2: 71 patients total, normal anatomy, 92% girls n n Garin et. al. (1998) UTI VUR? no n VUR UTI? no Scars? n VUR Scarring? no n ■ VUR Pyelo? ■ Degree VUR

We Need A Study That… n Will help us decide whether or not to prophylax this patient Includes patients with symptomatic VUR n Compares antibiotic prophylaxis to a control n Looks at clinically important outcomes n

Clinical significance of primary vesicoureteral reflux and urinary antibiotic prophylaxis after acute pyelonephritis: a multicenter, randomized, controlled study. Garin EH, Olavarria F, Garcia Nieto V, Valenciano B, Campos A, Young L. Pediatrics 2006; 117: 626 -632.

Study Questions n Does VUR correlate with UTI/renal scarring? n Does antibiotic prophylaxis correlate with UTI/renal scarring?

Study Design Randomized, controlled, multicenter trial n Inclusion: n 3 mo-18 yo n Acute pyelonephritis n n Exclusion: Grade IV-V VUR n Anatomic abnormalities n Pregnancy n
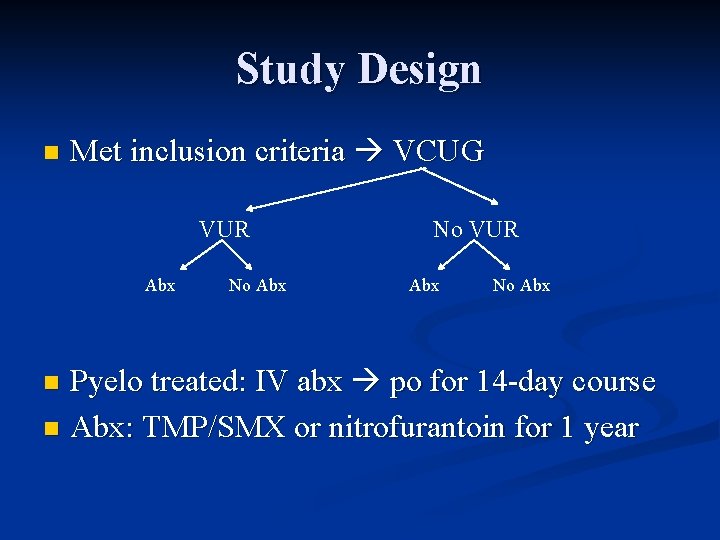
Study Design n Met inclusion criteria VCUG VUR Abx No Abx Pyelo treated: IV abx po for 14 -day course n Abx: TMP/SMX or nitrofurantoin for 1 year n
Follow Up At entry: UA/UCx, DMSA, VCUG, Renal US n At Q 3 mo clinic visit: UA/UCx n At 6 mo: DMSA n At 12 mo: VCUG, Renal US n n Endpoints: Recurrent UTI n Renal scarring n
Study Results

Analysis of Results n Fisher’s Exact Test 2 x 2 comparison tables n Control vs. variable n Smaller sample size n Gives p value n Does not give CI n n Goal: p<. 05! http: //www. childrensmercy. org/stats/ask/fishers. asp

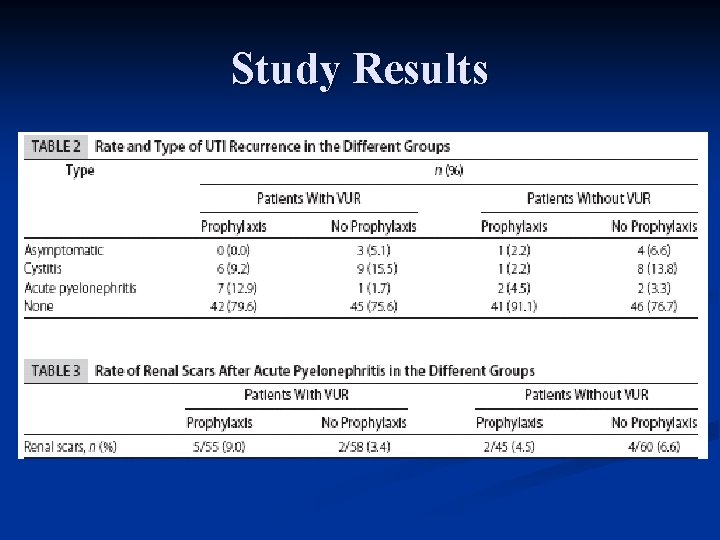
Study Results n Recurrence of UTIs Timing n Type n Recurrent Pyelonephritis & Antibiotics n Recurrent Pyelonephritis & VUR Degree n Renal Scarring n VUR n Antibiotics n

Study Results n Recurrence of UTI n Type of Recurrence n Overall – 20. 1% VUR not significant No abx (p=. 9999) n Cystitis (no p value) VUR – 22. 4% No VUR – 23. 3% n n n Abx (p=0. 633) n n VUR – 23. 6% No VUR – 8. 8% n n VUR – 8. 6% No VUR – 13. 3% Pyelonephritis (p=. 3781) n n VUR – 7. 1% No VUR – 3. 8%

Study Results n Recurrent Pyelo and Antibiotics No benefit of abx (p=. 0291) n 7: 1 abx: none n n Recurrent Pyelo and VUR Degree 6/8 Grade III (cystitis: 46%) n 2/8 Grade II (cystitis: 40%) n 4/4 pts without VUR n

Study Results n Renal Scarring n No evidence VUR increased scarring (p=. 9999) VUR (6. 2%) = No VUR (5. 7%) n Abx (7. 0%) = No Abx (5. 1%) n Grade I VUR – 5. 3% with scars n Grade II VUR – 5. 2% n Grade III VUR – 13. 5% n

Study Conclusions n Mild/moderate VUR not associated with UTI, pyelonephritis, or scarring n Antibiotic prophylaxis not associated with UTI, pyeloneprhitis, or scarring

Critically Evaluating… (JAMA Users’ Guide) n Are the results valid? n What were the results? n Will the results help me to take care of my patient?

Are the results valid? Primary Guides Was the assignment of patients to treatment randomized? YES. n Were all who entered the study accounted for? n n Was follow-up complete? n NO. Enrolled 236, lost 18 n Lost from what groups? n Would this change results? n Were patients analyzed in the groups assigned to? n NO. Exclusion of noncompliants

Are the results valid? Secondary Guides n Were pts, clinicians, & study personnel blinded? n n NO (no blinding to +/- VUR, abx; no placebos) Were groups similar at start, & treated equally? n YES (age, gender, degree of reflux)

What were the results? n How large was the treatment effect? n n n ARR – risk difference of variable vs. control RRR – variable reduced risk by Z% relative to that occurring in control patients; bigger = better! For example, in presence of VUR: n n 23. 6% of those on abx developed UTI (X%) 22. 4% without abx developed UTI (Y%) ARR = X-Y =. 236 -. 224 =. 012 RRR = (1 -Y/X)x 100% = (1 -. 224/. 236)x 100% = 5. 1%

What were the results? n How large was the treatment effect? n n ARR/RRR not reported! How precise was the estimated treatment effect? Confidence Intervals (CIs) not reported! n 95% CI: n n Range that includes the true RRR 95% of time n Positive? Negative? Zero? n Statistically vs. clinically significant results

What were the results? n POWER! n n n Ability of a study to detect a true difference Directly related to sample size 1 -β (β = type II error) Study powered to detect a clinically significant difference of 20% (power 80%), 95% CI Need 60/group = 240 subjects n Enrolled 236, Completed 218 “POWER : research design : : SENSITIVITY : diagnostic test”

Will the results help me take care of my patient? n Can the results be applied? YES. n n Could Kali have been enrolled? All clinically important outcomes considered? YES. Substitute endpoints vs. “POEMS” n Adverse effects on other outcomes n n Are likely benefits worth potential harms/risks? NNT = 1/ARR n Consider baseline risk without intervention n

Criticisms: Study Population n What about <3 mo? Present earlier = Higher-grade reflux? Already abx? n Included in study? n Exclusion of noncompliants? n Exclusion of pyelonephritis x 2? n Initial presentation with cystitis? n Febrile UTI without DMSA changes? n n How many therefore excluded?

Criticisms: Study Design DMSA as inclusion criteria (multicenter)? n Account for 18 lost before study end? n Unknown prognostic factors n Recalculate results assuming they did well/poorly n n Blinding of patients/personnel? Placebo n Diagnosis n n Larger sample size?

Criticisms: Data Analysis n Reporting of CIs, ARR/RRR? Magnitude/precision of treatment effect n Rule in/out effect different from Ho n Data crunching using Chi-Square? n Different data combinations? n Did not achieve POWER n

Further Questions… n UTI prophylaxis vs. intermittent therapy? And risk of renal scarring n Over time, given resolution VUR n Larger sample size n VUR in context of abnormal anatomy? n Mechanism of scarring in pyelonephritis? n What else? n

Back to our patient… n What would you do? n Call Dr. Cartwright and Dr. Lechner and get those patients enrolled!

References 1. 2. 3. 4. 5. 6. 7. 8. 9. Atala A, Keating MA. Vesicoureteral reflux and megaureter. In Campbell’s Urology Vol 2, 7 th ed. Philadelphia: WB Saunders 1988. Behrman Re, Kliegman RB, Jenson HB. Nelson Textbook of Pediatrics, 17 th ed. Philadelphia: Saunders, 2004. Biggi A et. al. Prognostic value of the acute DMSA scan in children with first urinary tract infection. Pediatr Nephrol 2001; 16: 800 -804. Bjorgvinsson E, Majd M, Eggli KD. Diagnosis of acute pyelonephritis in children: comparison of sonography and 99 m. Tc-DMSA scintigraphy. Am J Roentgenol 1991; 157(3): 539 -543. Dawson B, Trapp RG. Basic and clinical biostatistics, 3 rd ed. New York: Lange Medical Books 2001. De. Sadeeler C et. al. A multicenter trial on interobserver reproducibility in reporting on 99 m. Tc-DMSA planer scintigraphy: a Belgian survey. J Nucl Med 2000; 41(1): 23 -26. Elder JS et. al. Pediatric vesicoureteral reflux guidelines panel summary report on the management of primary vesicoureteral reflux in children. J Urol 1997; 157(5): 1846 -1851. Garin EH et. al. Clinical significance of primary vesicoureteral reflux and urinary antibiotic prophylaxis after acute pyelonephritis: a multicenter, randomized, controlled study. Pediatrics 2006; 117: 626 -632. Garin EH, Campos A, Homsy Y. Primary vesicoureteral reflux: review of current concepts. Pediatr Nephrol 1998; 12: 249 -256.

References 10. 11. 12. 13. 14. 15. 16. 17. Gordon I et. al. Primary vesicoureteral reflux as a predictor of renal damage in children hospitalized with urinary tract infection: a systematic review and metaanalysis. J Am Soc Nephrol 2003; 14: 739 -744. Guyatt GH et. al. How to use an article about therapy or prevention. A. Are the results of the study valid? JAMA 1993; 270: 2598 -2601. Guyatt GH et. al. How to use an article about therapy or prevention. B. What were the results and will they help me in caring for my patients? JAMA 1994; 271: 59 -63. Lee RS et. al. Antenatal hydronephrosis as a predictor of postnatal outcome: a meta-analysis. Pediatrics 2006; 118(2): 586 -593. Penido Silva JM et. al. Clinical course of prenatally detected primary vesicoureteral reflux. Pediatr Nephrol 2006; 21: 86 -91. Schwab CW et. al. Spontaneous resolution of vesicoureteral reflux: a 15 -year perspective. J Urol 2002; 168: 2594 -2599. Williams G et. al. Antibiotics for the prevention of urinary tract infection in children: a systematic review of randomized controlled trials. J Pediatr 2001; 138(6): 868 -874. Yu RN, Roth DR. Treatment of vesicoureteral reflux using endoscopic injection of nonanimal stabilized hyaluronic acid/dextranomer gel: initial experience in pediatric patients by a single surgeon. Pediatrics 2006; 118(2): 698703.
- Slides: 39