VTE Prevention Quality Improvement Strategies VTE venous thromboembolismDVTPE

VTE Prevention Quality Improvement Strategies VTE: venous thromboembolism-DVT/PE Jordan Messler, MD, SFHM Morton Plant Hospitalists Medical Director
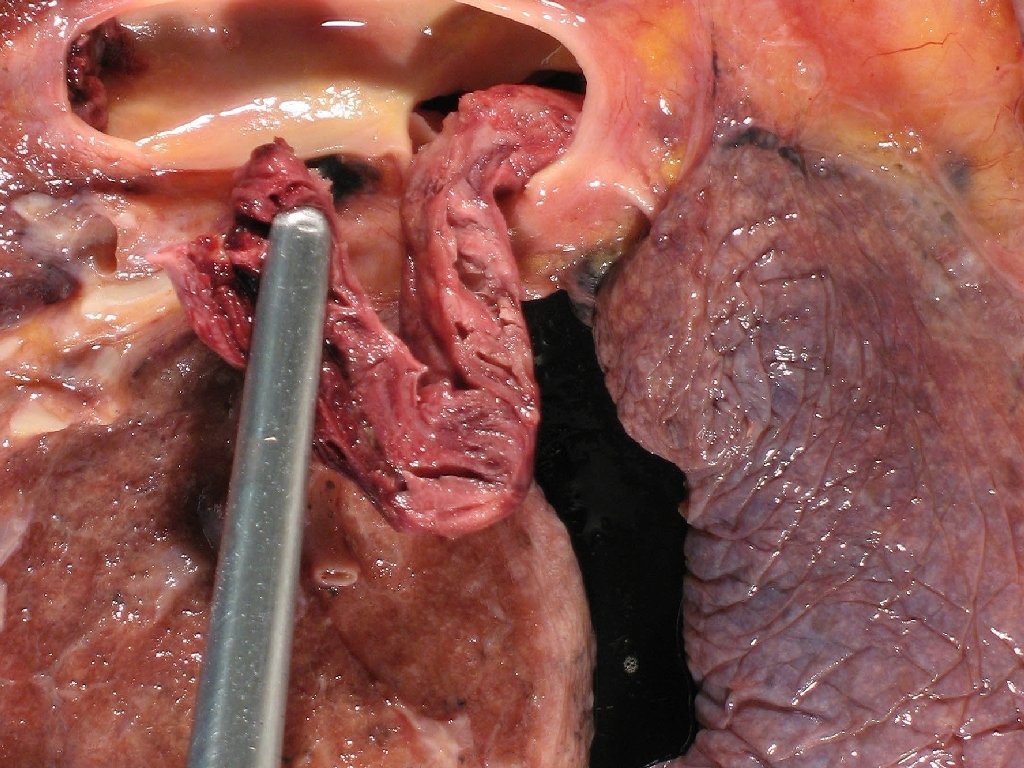
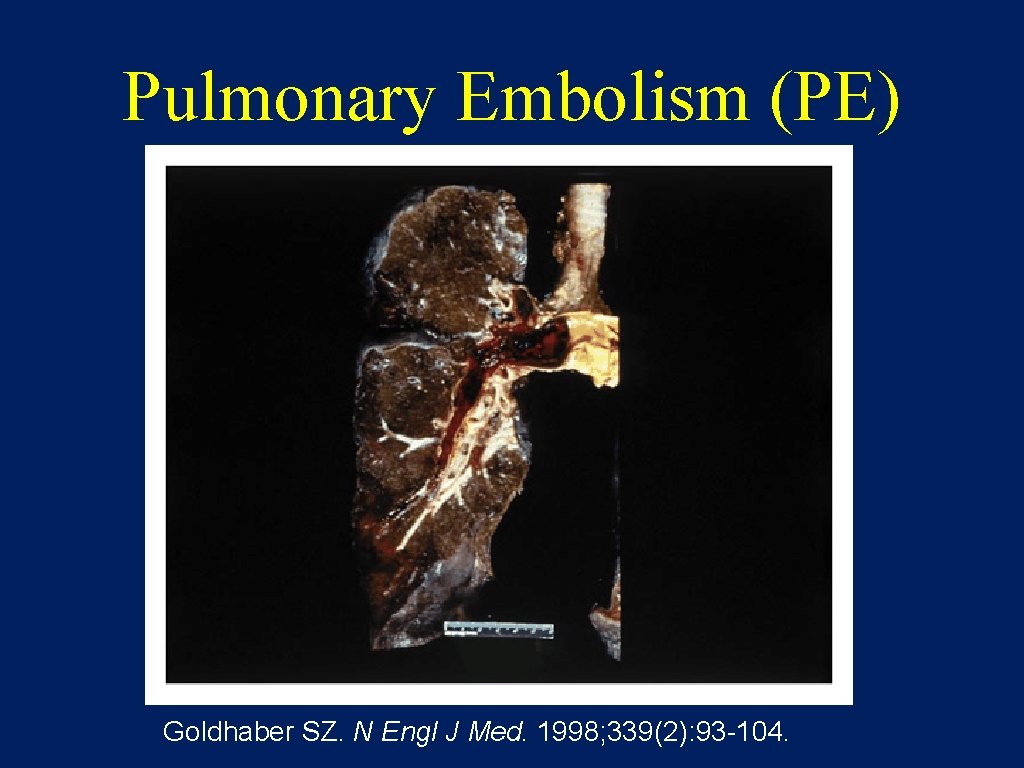
Pulmonary Embolism (PE) Goldhaber SZ. N Engl J Med. 1998; 339(2): 93 -104.

Objectives • Understand the epidemiology of VTE and the impact of VTE prevention • Identify quality improvement strategies to improve VTE prevention at the local level • Understand some of the latest updates in VTE prevention guidelines

• VTE prevention basics • Quality Improvement strategies • ACCP 2012 guidelines • Other VTE prophylaxis pearls
VTE: A Major Source of Mortality and Morbidity • • 350, 000 to 650, 000 with VTE per year 100, 000 to > 200, 000 deaths per year Most are hospital related VTE is primary cause of fatality in half– More than HIV, MVAs, Breast CA combined – Equals 1 jumbo jet crash / day • 10% of hospital deaths – May be the #1 preventable cause • Huge costs and morbidity (recurrence, postthrombotic syndrome, chronic PAH) Surgeon General’s Call to Action to Prevent DVT and PE 2008 DHHS

A 500 bed hospital can expect 250 hospital acquired DVT/PE per year. Half of these are preventable Going from 50 to 90% VTE prophylaxis over the course of a year will avoid 68 DVTs, 29 PEs, & 5 Deaths
8

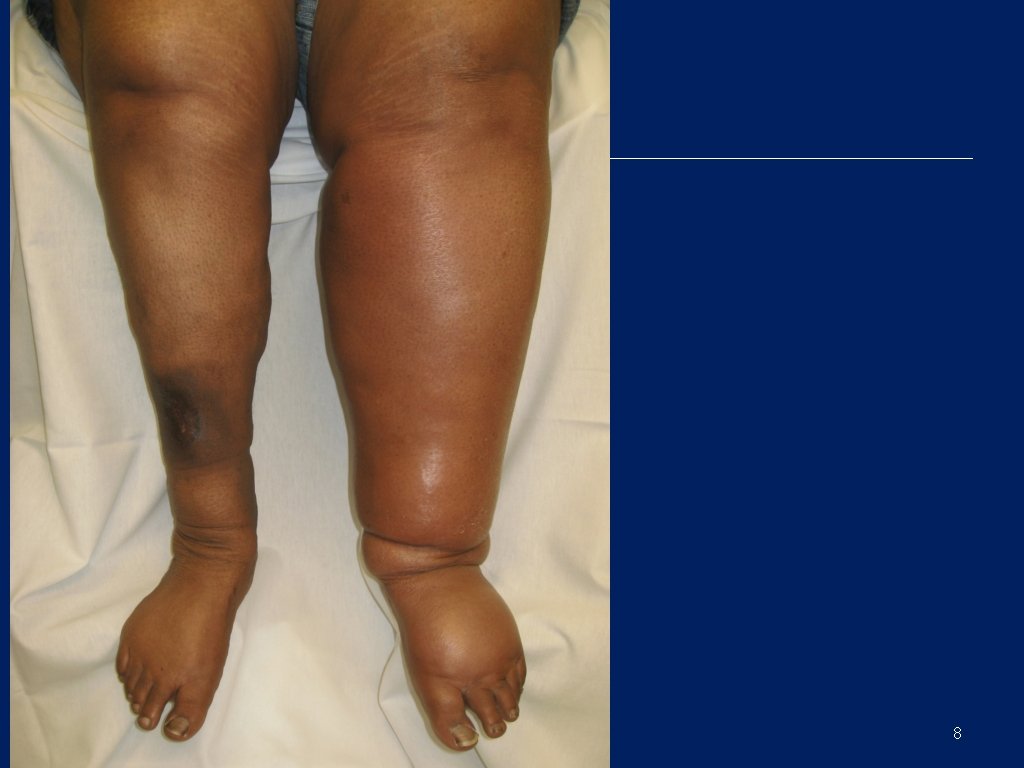
VTE Consequences • • Leg swelling, discomfort (DVT) Dyspnea, chest pain, hemoptysis, hypoxemia (PE) Extended hospital LOS Fatal PE (RV failure) ≥ 3 months of anticoagulant treatment Post thrombotic syndrome Chronic thromboembolic pulmonary HTN (~4%) Pengo et al. N Engl J Med. 2004; 350: 2257 -2264.

Risk Factors for VTE Stasis Hypercoagulability Age > 40 Anesthesia Immobility Heart Failure Stroke Paralysis Spinal cord injury Polycythemia Severe COPD Obesity Varicose Veins Cancer Inflammatory Bowel Medications Nephrotic Syndrome Sepsis Smoking Pregnancy Thrombophilia Endothelial Damage Surgery Prior VTE Central lines Trauma Most hospitalized patients have at least one risk factor for VTE Anderson FA Jr. & Wheeler HB. Clin Chest Med 1995; 16: 235. Bick RL & Kaplan H. Med Clin North Am 1998; 82: 409.
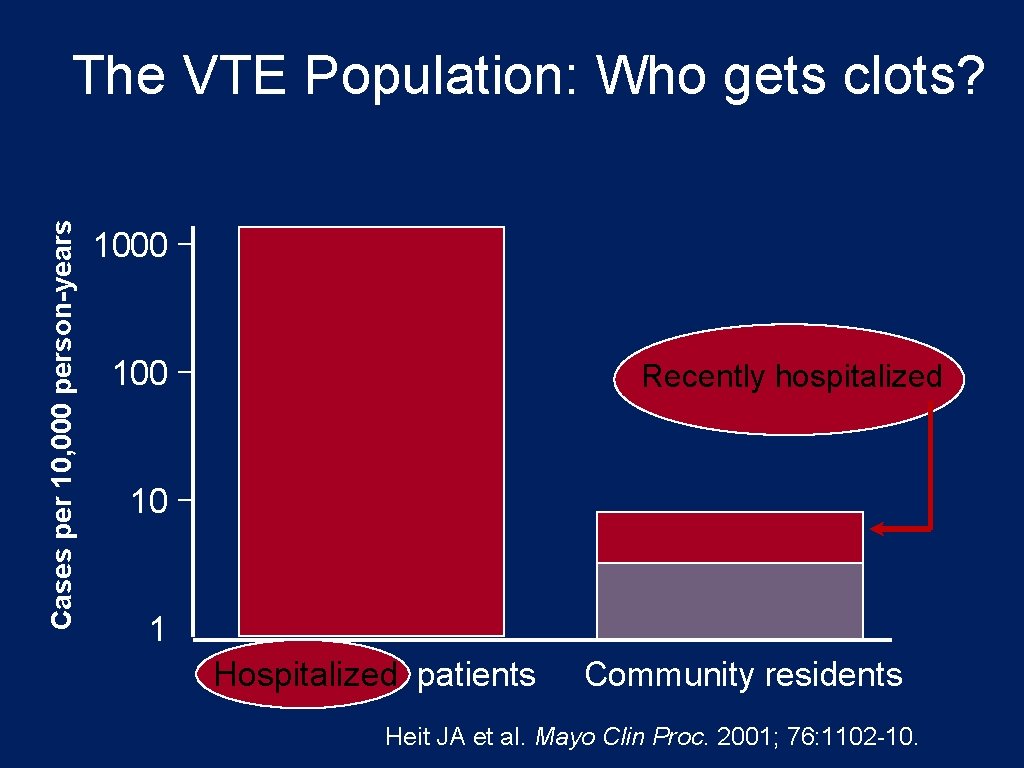
Cases per 10, 000 person-years The VTE Population: Who gets clots? 1000 100 Recently hospitalized 10 1 Hospitalized patients Community residents Heit JA et al. Mayo Clin Proc. 2001; 76: 1102 -10.

Many Surgical Patients At-Risk • 2003 Nationwide Inpatient Sample • Adult surgical patients, LOS ≥ 2 days • 7. 8 million surgical discharges • • 44% low risk 15% moderate risk 24% high risk 17% very high risk • 4. 4 million at risk for VTE Anderson et al. Am J Hematol. 2007; 82: 777 -782.

Effective, Safe, and Cost-Effective • Pharmacologic prophylaxis substantially reduces the risk for VTE • Bleeding complications are rare • HIT: a serious but relatively rare complication of heparin therapy – 2. 37% with prolonged UFH in ill perioperative patients – 0. 06% with LMWH – Monitoring for HIT is warranted • Cost-effectiveness of VTE prophylaxis well documented HIT = heparin-induced thrombocytopenia LMWH = low molecular weight heparin UFH = unfractionated heparin Geerts WH et al. Chest. 2008; 133(6 suppl): 381 S-453 S. Shojania KG et al. Making health care safer. Martel N et al. Blood. 2005; 106: 2710 -5.
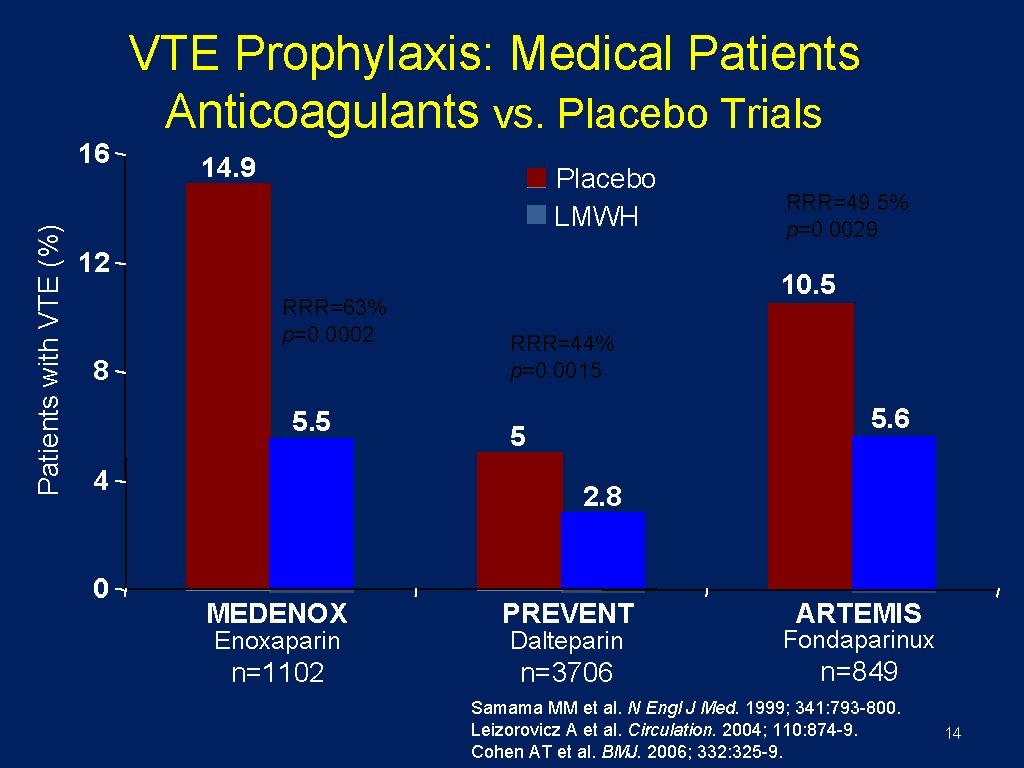
VTE Prophylaxis: Medical Patients Anticoagulants vs. Placebo Trials Patients with VTE (%) 16 14. 9 Placebo LMWH 12 RRR=63% p=0. 0002 8 5. 5 4 0 RRR=49. 5% p=0. 0029 10. 5 RRR=44% p=0. 0015 5. 6 5 2. 8 MEDENOX PREVENT Dalteparin Fondaparinux n=1102 n=3706 n=849 Enoxaparin ARTEMIS Samama MM et al. N Engl J Med. 1999; 341: 793 -800. Leizorovicz A et al. Circulation. 2004; 110: 874 -9. Cohen AT et al. BMJ. 2006; 332: 325 -9. 14

VTE Prophylaxis Meta-Analysis • • 9 studies 19, 958 medical patients Anticoagulant prophylaxis vs no treatment Results – 57% reduction in RR for symptomatic PE – 62% reduction in RR for fatal PE – 53% reduction in DVT – No significant increase in major bleeding Dentali F, et al. Ann Intern Med. 2007; 146: 278 -288.

Registries Highlight the Underuse of VTE Prophylaxis in Hospitalized Patients 15, 000 patients 1, 800 patients 68, 000 patients IMPROVE 1 CURVE 2 ENDORSE 3 90% at risk 60% any VTE prophylaxis 23% any VTE prophylaxis (16% appropriate) 40% appropriate VTE prophylaxis Only a minority of hospitalized medical patients receive appropriate VTE prophylaxis 1. Tapson VF et al. Chest. 2007; 132: 936 -45. 2. Kahn SR et al. Thromb Res. 2007; 119: 145 -55. 3. Cohen AT et al. Lancet. 2008; 371: 387 -94.
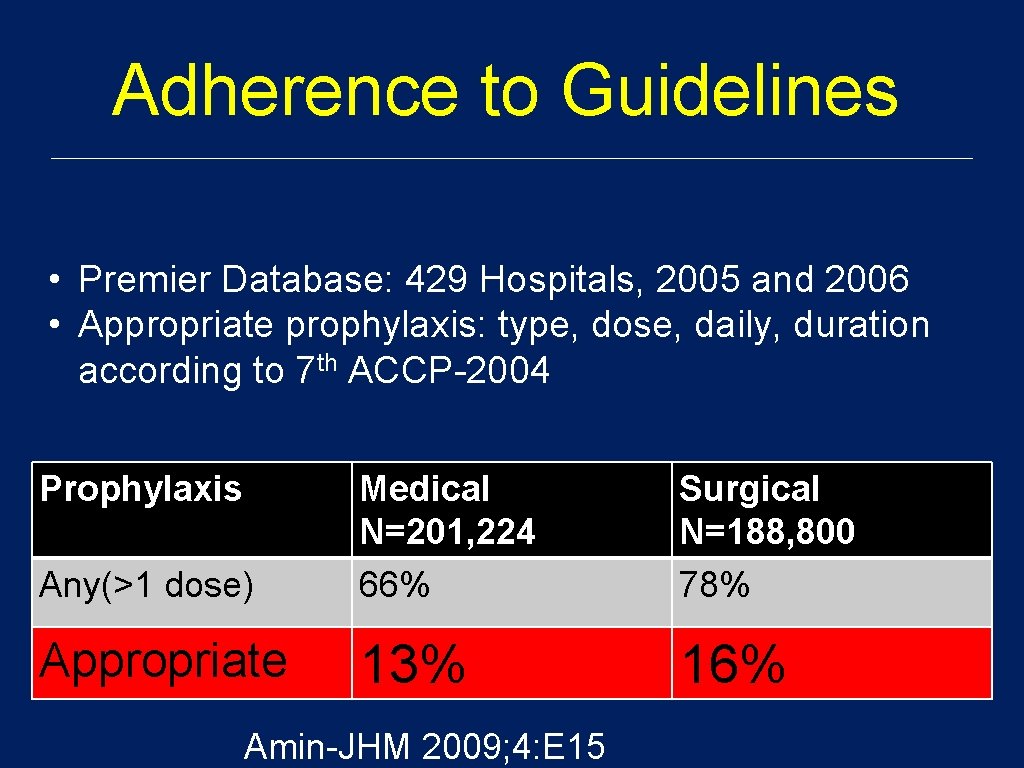
Adherence to Guidelines • Premier Database: 429 Hospitals, 2005 and 2006 • Appropriate prophylaxis: type, dose, daily, duration according to 7 th ACCP-2004 Prophylaxis Any(>1 dose) Medical N=201, 224 66% Surgical N=188, 800 78% Appropriate 13% 16% Amin-JHM 2009; 4: E 15
CT Pulmonary Angiography Goldhaber SZ. N Engl J Med. 1998; 339(2): 93 -104. Kearon SF. CMAJ. 2003; 168: 183 -194.


Why don’t we do better? • Competing Priorities • National Policies / Incentives / Initiatives / Accreditation not all in place • Lack of awareness of guidelines, battling guidelines • Underestimation of clot risk, overestimation of bleeding risk • Validated and practical risk assessment models needed • Measurement Issues • Translating complicated guidelines into everyday practice is difficult • Medical training failures (QI and systems re-design) • Failure to use a good QI framework
My First Algorithm for Process Improvement

VTE Prevention Guides www. ahrq. gov/qual/vtguide Maynard G, Stein J. Agency for Healthcare Research and Quality. August 2008. http: //www. ahrq. gov/qual/vtguide/

Quality Improvement is… • Focus on processes of care • Reduced variation by shifting entire practice • A change in the design of care Quality Improvement is NOT… • Forcing people to work harder / faster / safer • Traditional quality assurance or peer review • Creating order sets or protocols without monitoring use or effect

VTE Prophylaxis A Multidisciplinary Approach • TEAM • AIM STATEMENT: Goals • Measurement • Analyze Process • STANDARDIZE based on best evidence • Identification and Mitigation Maynard G, Stein J. Agency for Healthcare Research and Quality. August 2008. 24
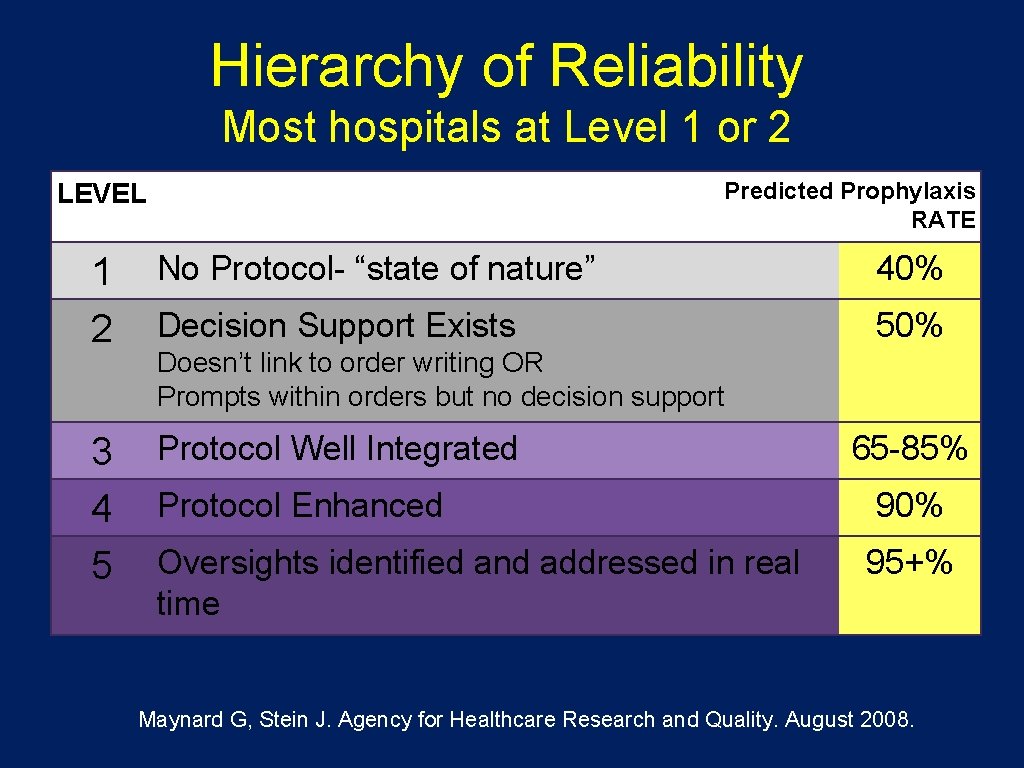
Hierarchy of Reliability Most hospitals at Level 1 or 2 Predicted Prophylaxis RATE LEVEL 1 2 No Protocol- “state of nature” 40% Decision Support Exists 50% 3 4 5 Protocol Well Integrated Doesn’t link to order writing OR Prompts within orders but no decision support 65 -85% Protocol Enhanced 90% Oversights identified and addressed in real time 95+% Maynard G, Stein J. Agency for Healthcare Research and Quality. August 2008.

The Multidisciplinary Team: The Engine of Quality Improvement • Backbone of quality improvement (QI) efforts • Impact the interventions developed AND their implementation • Synergistic • Productive capacity = more than the sum of all individual team members taken together

Metrics are Essential for VTE Prevention Efforts • Key component of institutional support • Use consistent metrics over time • Report results regularly to med staff/ admin

Different Types of Metrics in VTE Prevention Efforts Structure Dedicated quality improvement team? Standardized order sets, protocols? Electronic Health Record? CPOE? Process % with VTE risk assessment documented % with prophylaxis or contraindication within 24 hrs % using standardized order set % receiving pharmacologic prophylaxis, mechanical prophylaxis % adherence to mechanical prophylaxis

Different Types of Metrics in VTE Prevention Efforts • Outcomes • # of patients with Hospital-Acquired VTE (HA VTE) • Rate of HA VTE per 1000 days or 1000 discharges • % of HA VTE that were potentially preventable • Balancing Measures • Bleeding incidence • Heparin-induced Thrombocytopenia incidence • Cost (Savings) Maynard G, Stein J. Agency for Healthcare Research and Quality. August 2008.

VTE Prophylaxis Audits Assessing Prevalence of Adequate VTE Prophylaxis • Order set use • Detailed audits based on your protocol • Less detailed audits • (Red / Yellow / Green strategy) 30
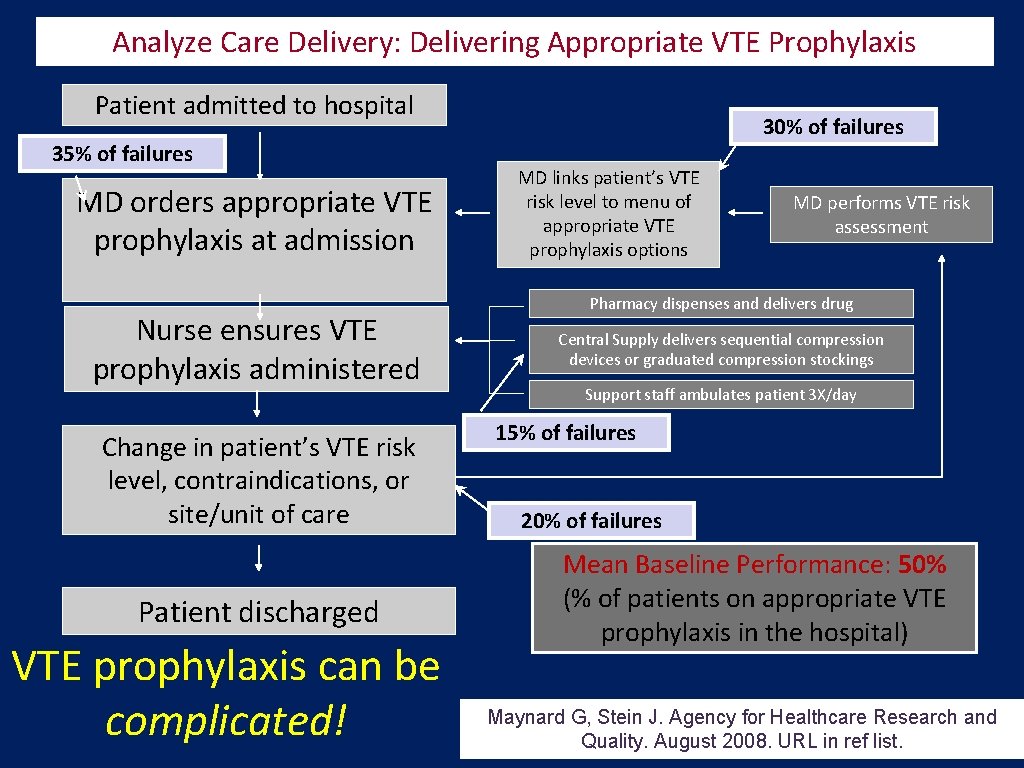
Analyze Care Delivery: Delivering Appropriate VTE Prophylaxis Patient admitted to hospital 35% of failures MD orders appropriate VTE prophylaxis at admission Nurse ensures VTE prophylaxis administered Change in patient’s VTE risk level, contraindications, or site/unit of care Patient discharged VTE prophylaxis can be complicated! 30% of failures MD links patient’s VTE risk level to menu of appropriate VTE prophylaxis options MD performs VTE risk assessment Pharmacy dispenses and delivers drug Central Supply delivers sequential compression devices or graduated compression stockings Support staff ambulates patient 3 X/day 15% of failures 20% of failures Mean Baseline Performance: 50% (% of patients on appropriate VTE prophylaxis in the hospital) Maynard G, Stein J. Agency for Healthcare Research and Quality. August 2008. URL in ref list.

Is your VTE order set in competition?

VTE Protocol Key Principles 1. 2. 3. 4. 5. Keep protocol simple to access and use Don’t interrupt the workflow Design reliability into the new process Monitor use of your protocol Allow for variation from the protocol based on patient characteristics (rather than providers) - improve protocol based on feedback and justifiable variation 6. Fail faster (pilot small scale w/ongoing feedback & refinement before wider implementation)

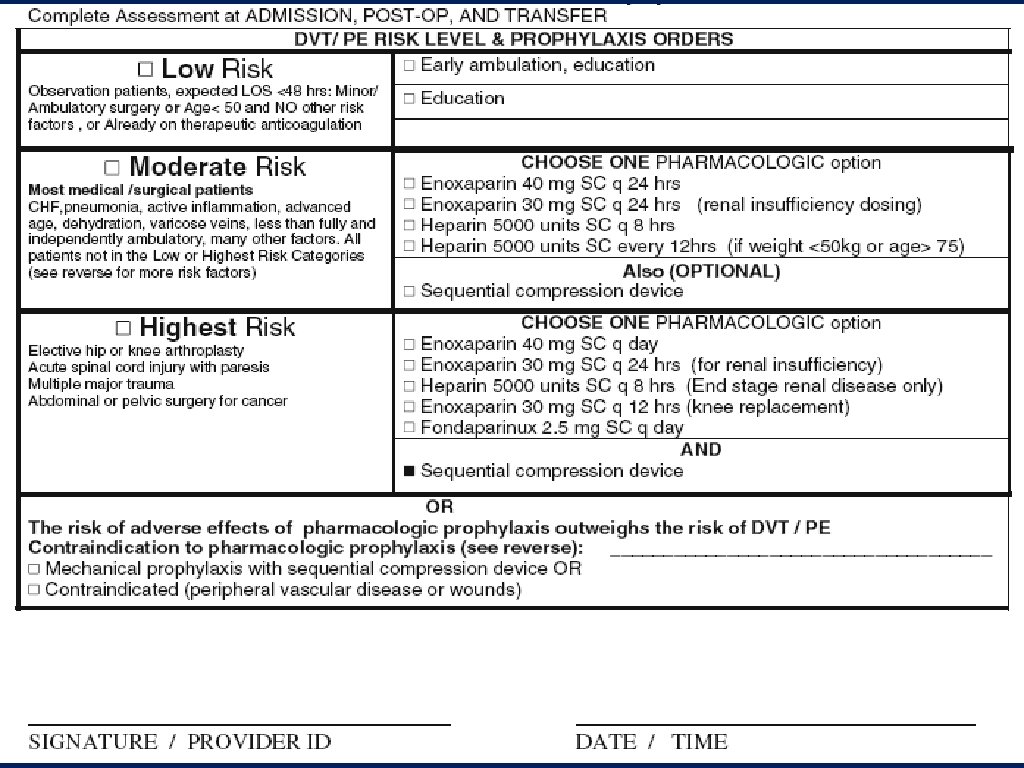
Too Little Guidance Prompt ≠ Protocol DVT PROPHYLAXIS ORDERS q q q q Anti thromboembolism Stockings Sequential Compression Devices UFH 5000 units Sub. Q q 12 hours UFH 5000 units Sub. Q q 8 hours LMWH (Enoxaparin) 40 mg Sub. Q q day LMWH (Enoxaparin) 30 mg Sub. Q q 12 hours No Prophylaxis, Ambulate

VTE Prophylaxis: 3 Patient Groups Low risk Moderate risk High risk Geerts WH et al. Chest. 2008; 133(6 suppl): 381 S-453 S.
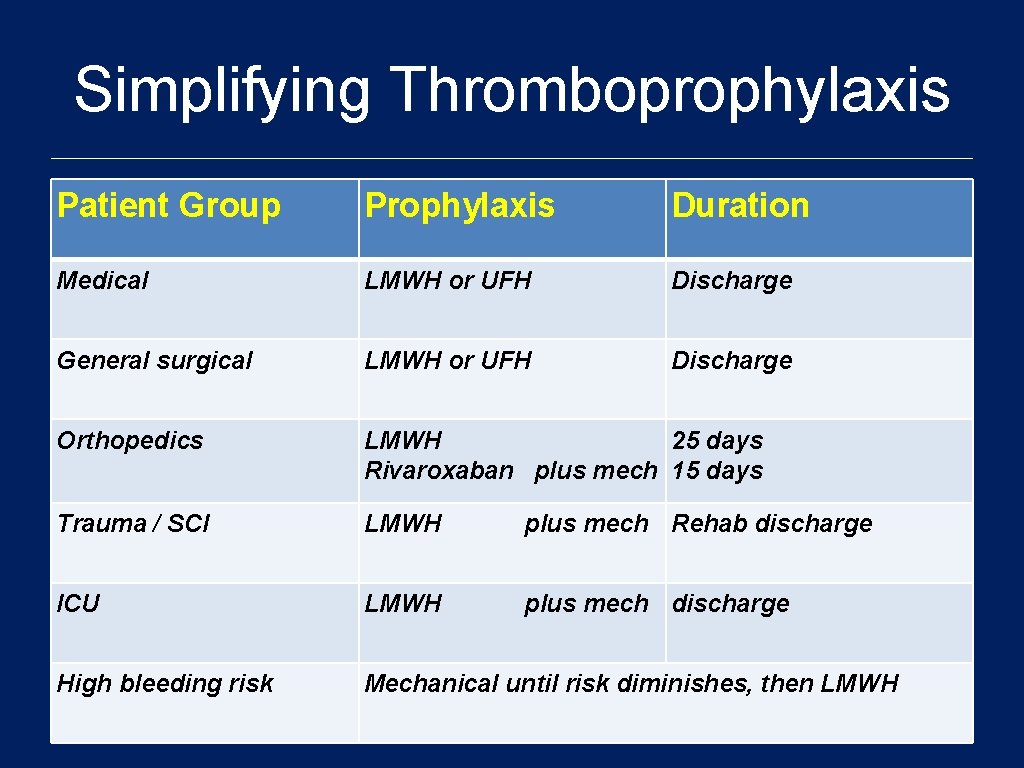
Simplifying Thromboprophylaxis Patient Group Prophylaxis Duration Medical LMWH or UFH Discharge General surgical LMWH or UFH Discharge Orthopedics LMWH 25 days Rivaroxaban plus mech 15 days Trauma / SCI LMWH plus mech Rehab discharge ICU LMWH plus mech discharge High bleeding risk Mechanical until risk diminishes, then LMWH

Validated Risk Scoring Systems University of California San Diego – Retrospective analysis – Three-tier risk assessment model (RAM) – Predicts Risk – Validated in actual use Maynard GA et al. J Hosp Med. 2010; 5: 10 -8.

UCSD VTE Protocol Validated • Easy to use, on direct observation – a few seconds • Inter-observer agreement – – 150 patients, 5 observers- Kappa 0. 8 and 0. 9 • Predictive of VTE • Implementation = high levels of VTE prophylaxis –From 50% to sustained 98% adequate prophylaxis –Rates determined by over 2, 900 random sample audits • Safe – no discernible increase in HIT or bleeding • Effective – 40% reduction in HA VTE – 86% reduction in risk of preventable VTE J Hosp Med 2010 Jan: 5(1): 10 -18.
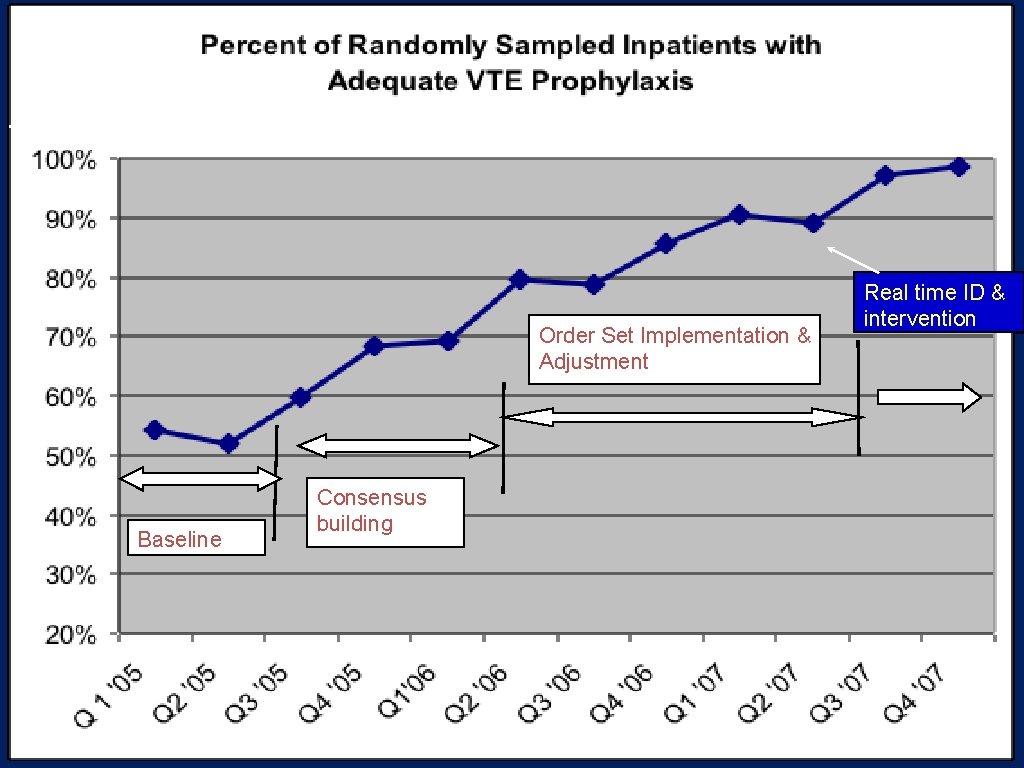
J Hosp Med 2010 Jan: 5(1): 10 -18. N = 2, 944 mean 82 audits / month Order Set Implementation & Adjustment Baseline Real time ID & intervention Consensus building 32
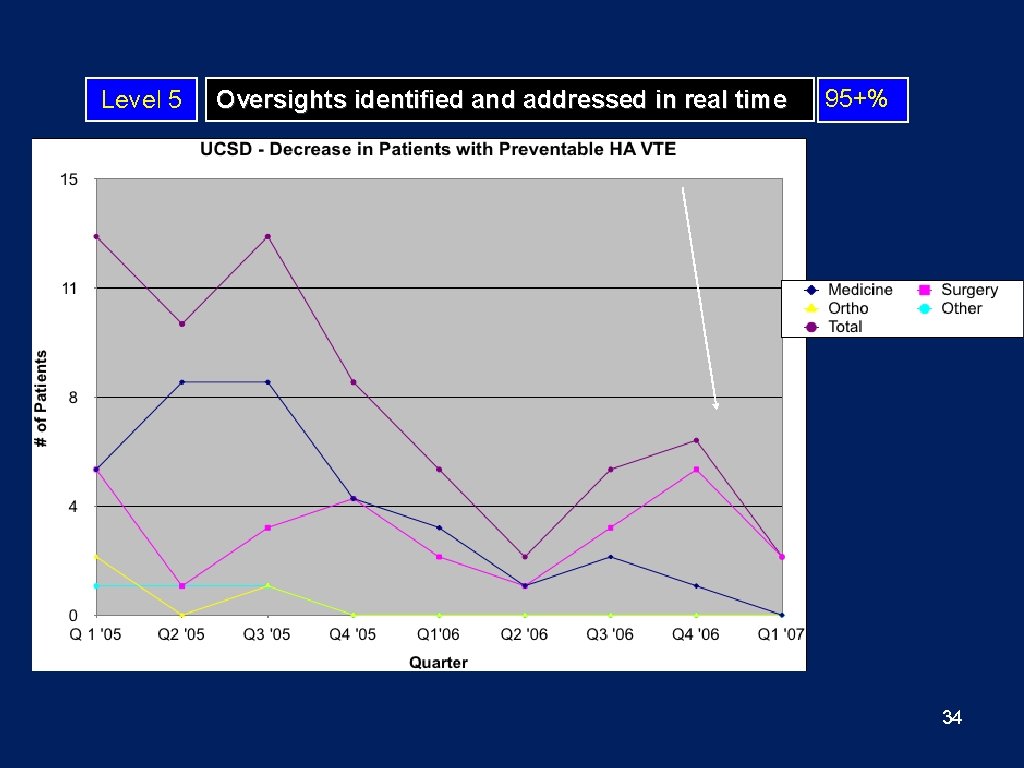
Level 5 Oversights identified and addressed in real time 95+% 34

High Reliability Principles • Standardize VTE and anticoagulation risk assessment into the process of admission and transfers • “Opt out” of default choices (not opt in) • Prompts for VTE risk assessment at pointof-care • Scheduled reassessments • Redundant responsibility and prompts

Strategies to Improve Prophylaxis Rates • BASIC INTERVENTIONS • In-services • Newsletters • Quality improvement presentations Dobesh PP et al. J Manag Care Pharm. 2005; 11: 755 -62.

Optimize Strategies for Effective VTE Prevention • Alert Systems • • • Electronic alerts (E-alerts) • Human alerts Computerized decision support Raising situational awareness Audit and feedback Measure-vention
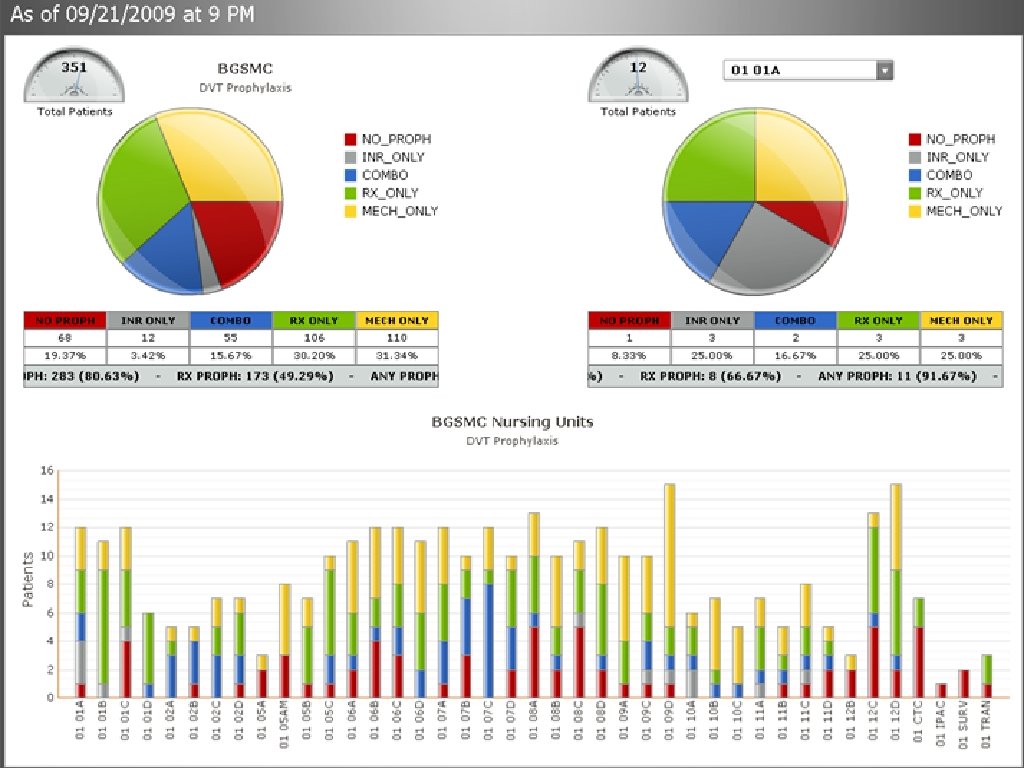

MEASURE-VENTION Daily measurement drives concurrent intervention Identify patients not receiving VTE prophylaxis in real time Ongoing assessment, creates data, reports data Use for real-time intervention

Map to Reach Level 5 Situational Awareness and Measure-vention • Identify patients not receiving anticoagulation • Empower nurses to place SCDs on these patients as standing order (if no contraindications) • Contact MD when patients are not receiving anticoagulant with no obvious contraindication • Template note, ‘text’ page, etc. • Administration to support these interventions with clear message that physicians should not “shoot the messenger” SCDs = sequential compression devices

Map to Reach Level 5 95+ % Prophylaxis Use Medication Administration Record (MAR) or automated reports to classify all patients in one of three zones: GREEN ZONE receiving anticoagulation YELLOW ZONE receiving mechanical prophylaxis only RED ZONE receiving no prophylaxis Act to move patients at risk for VTE out of the RED!

Digging Deeper on “Yellow” Patients Is patient low risk? – – – Ambulating Independently with 0 -1 VTE Risk Factors Expected LOS <48 hours Minor Surgery with NO VTE Risk Factors If yes, prophylaxis adequate, if no…. . Obvious contraindication to pharmacologic prophylaxis? – – – – Active hemorrhage now or within last 3 days Post operative bleeding concerns Platelet count < 50, 000 Units INR > 1. 8 Known bleeding disorder, post op bleeding high risk Hgb < 8. 0 g/d. L Concern over CNS bleeding (brain or spinal cord surgery in last week, recent intracranial hemorrhage, proximity in time to epidural insertion or removal, for example) – Hypertensive urgency / emergency – Comfort care only patient If yes, mechanical prophylaxis alone adequate, if no, prophylaxis inadequate
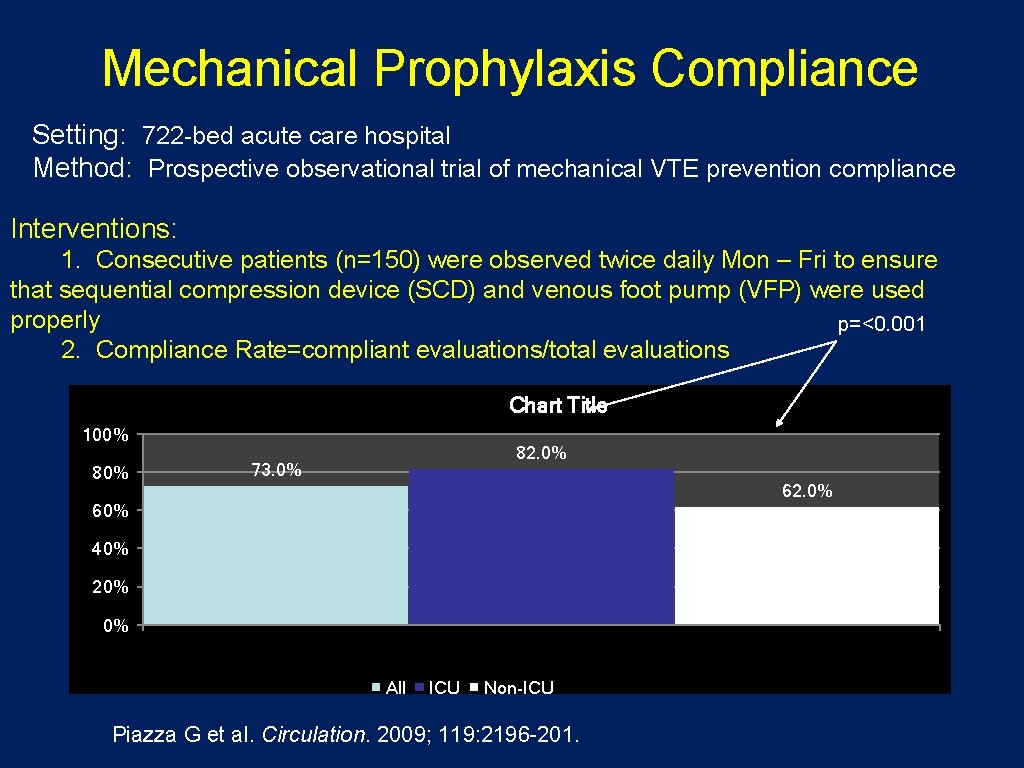
Mechanical Prophylaxis Compliance Setting: 722 -bed acute care hospital Method: Prospective observational trial of mechanical VTE prevention compliance Interventions: 1. Consecutive patients (n=150) were observed twice daily Mon – Fri to ensure that sequential compression device (SCD) and venous foot pump (VFP) were used properly p=<0. 001 2. Compliance Rate=compliant evaluations/total evaluations Chart Title 100% 82. 0% 73. 0% 62. 0% 60% 40% 20% 0% All ICU Non-ICU Piazza G et al. Circulation. 2009; 119: 2196 -201.
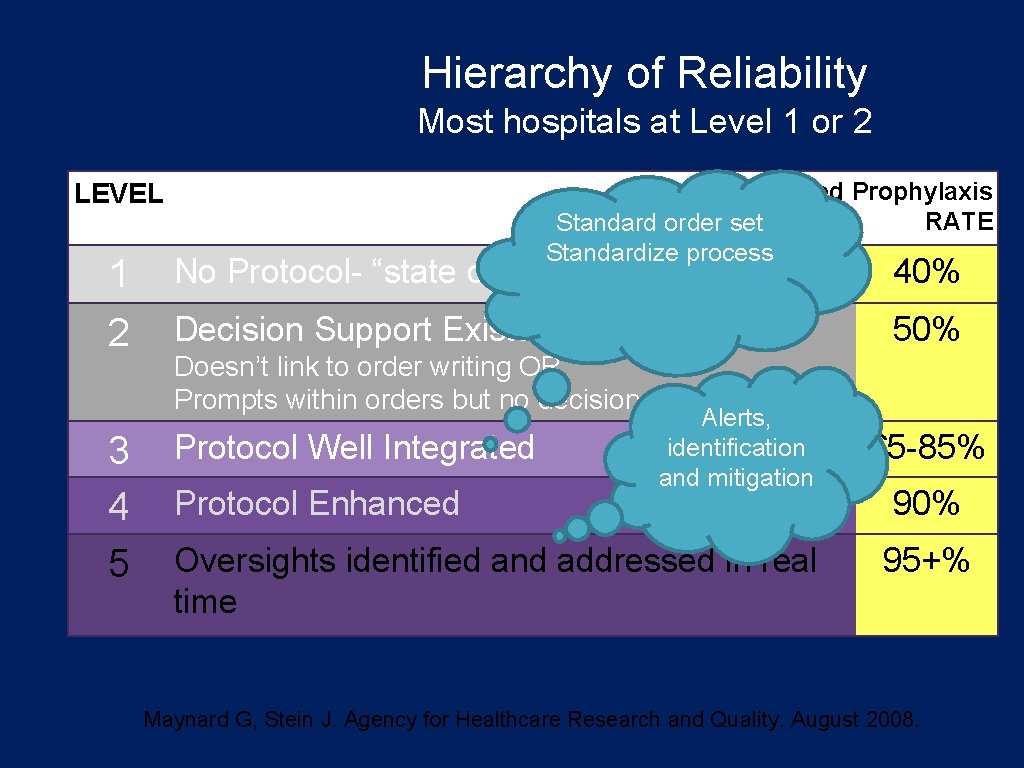
Hierarchy of Reliability Most hospitals at Level 1 or 2 Predicted Prophylaxis RATE Standard order set Standardize process LEVEL 1 2 3 4 5 No Protocol- “state of nature” 40% Decision Support Exists 50% Doesn’t link to order writing OR Prompts within orders but no decision support Protocol Well Integrated Protocol Enhanced Alerts, identification and mitigation Oversights identified and addressed in real time 65 -85% 90% 95+% Maynard G, Stein J. Agency for Healthcare Research and Quality. August 2008.

VTE Prevention Collaboratives Using UCSD Model Over 250 Hospitals • Society of Hospital Medicine (SHM) • AHRQ and Quality Improvement Organizations • Institute for Healthcare Improvement (IHI) Expedition • British Columbia Hospital Medicine • American Society of Healthsystems Pharmacists (ASHP) • Awards to UCSD, Emory, UNM, Washington DC VA, Blessing (Quincy IL) and British Columbia based on these strategies (all members of mentored implementation) • Effective across variety of settings – Paper and Computerized / Electronic – Small and large institutions – Academic and community

2 Common Questions and Biased Answers Q. What is the best VTE risk assessment model? A. Simple, text based model with only 2 -3 layers of VTE Risk Q. Who should do the VTE risk assessment? A. Doctors (via admit transfer order sets), with back up risk assessment by front line nurses or pharmacists, focusing on those without prophylaxis.

Key Points - Recommendations • VTE Risk Assessment embedded in order sets • Simple risk stratification schema, based on VTE-risk groups (2 -3 levels of risk should do it) • Customization for some services is desirable. • Simple measures for adequate VTE prophylaxis – More detail on selected patients • Follow Outcomes • Work on adherence to ordered prophylaxis • Use measure-vention to accelerate improvement • Share information / comparing notes helps Maynard G, Stein J. Designing and Implementing Effective VTE Prevention Protocols: Lessons from Collaboratives. J Thrombolysis 2010 Feb: 29(2): 159 74 -166.

The Joint Commission • VTE-1 Venous Thromboembolism Prophylaxis • VTE-2 Intensive Care Unit Venous Thromboembolism Prophylaxis • VTE-3 Venous Thromboembolism Patients with Anticoagulation Overlap Therapy • VTE-4 Venous Thromboembolism Patients Receiving Unfractionated Heparin with Dosages/Platelet Count Monitoring by Protocol • VTE-5 Venous Thromboembolism Discharge Instructions • VTE-6 Incidence of Potentially-Preventable Venous Thromboembolism http: //www. jointcommission. org/assets/1/6/Venous%20 Thromboembolism. pdf

The Joint Commission on VTE Risk Assessment/ Prophylaxis 1. Documentation of VTE risk assessment/prophylaxis within 24 hr of hospital admission 2. Documentation of VTE risk assessment/prophylaxis within 24 hr after admission to or transfer to ICU Treatment Outcomes 6. Incidence of potentially-preventable venous thromboembolism The Joint Commission. Venous thromboembolism (VTE) core measure set. 53

Latest guidelines

What’s New in the ACCP Guidelines • • • Decrease in 1 A recommendations Orthopedic prophylaxis Mechanical Prophylaxis VTE prophylaxis in hospitalized medical patients Risk Assessment Models, endorsement and extrapolation

Decrease in 1 A recommendations Hirsh J, Guyatt G, Lewis SZ. Chest. 2008 Jun; 133(6): 1293 -5. PMID: 18574282 Guyatt GH. Chest. 2012 Feb; 141(2 Suppl): 48 S-52 S. PMID: 22315255

Decrease in 1 A recommendations Weak recommendations replacing strong recommendations WHY? • More critical look at the evidence, “symptomatic DVT vs asymptomatic” • recognition of variability in values and preferences. • the endorsement of > 80% of panelists to make a strong recommendation required. • the exclusion of conflicted experts Guyatt GH. Chest. 2012 Feb; 141(2 Suppl): 48 S-52 S. PMID: 22315255

• “Relying on the perspective of unconflicted methodologists, rigorously applying the GRADE approach, and excluding those with financial and intellectual conflict of interests from bottom-line decisions regarding the quality of evidence and strength of recommendations led to re-evaluations of previously existing evidence” Page 50 S

What’s New in the ACCP Guidelines Decrease in 1 A recommendations Orthopedic prophylaxis Mechanical Prophylaxis VTE prophylaxis in hospitalized medical patients Risk Assessment Models, endorsement and extrapolation

2012 ACCP Guideline 2. 1. 1 pts undergoing THA or TKA We recommend use of one of the following for 10 -14 days rather than no anti-thrombotic prophylaxis: LMWH, fondaprinux, apixaban, dabigatran, rivaroxaban, LDUH, VKA, ASA(all grade 1 b), or an intermittent IPCD(Grade 1 c) THA/TKA=Total Hip or Knee Arthroplasty IPCD=intermittent pneumatic compression device LDUH=low dose unfractionated heparin VKA=Vitamin K Antagonist-warfarin LMWH=low molecular weight heparin

2012 ACCP Guideline 2. 3. 1. In patients undergoing THA or TKA, irrespective of the concomitant use of an IPCD or length of treatment, we suggest the use of LMWH in preference to the other agents we have recommended as alternatives: fondaparinux, apixaban, dabigatran, rivaroxaban, LDUH (all Grade 2 B) , adjusted-dose VKA, or aspirin (all Grade 2 C) Allow ASA as a choice (split decision) Allows IPC as stand alone option (with caveats)

ASA? • 2008 ACCP: recommend against LDUH, ASA as sole method, with others getting 1 a recommendation, for THR/TKR • 2007 AAOS: PE prevention – Standard risk for PE and major bleeding pts should receive ASA, LMWH, fondaparinux, warfarin. With concurrent mechanical • 2011 AAOS: more general – “we suggest the use of pharmacological agents and/or compressive device for the prevention of VTE in pts with elective THR/TKR”

ASA • There is evidence ASA works, recommend better than nothing. Suggest LMWH in preference to others • Don’t use ASA for high risk pts

What’s New in the ACCP Guidelines • Decrease in 1 A recommendations • Orthopedic prophylaxis • Mechanical Prophylaxis • VTE prophylaxis in hospitalized medical patients • Risk Assessment Models, endorsement and extrapolation

ACCP endorses a specific SCD type as stand alone Joe Cummings, Ph. D, manager UHC Technology Assessment Group

What’s New in the ACCP Guidelines • Decrease in 1 A recommendations • Orthopedic prophylaxis • Mechanical Prophylaxis • VTE prophylaxis in hospitalized medical patients • Risk Assessment Models, endorsement and extrapolation

Medical prophylaxis 2012 ACCP 2008 ACCP 2. 3. For acutely ill hospitalized medical patients at increased risk of thrombosis, we recommend anticoagulant thromboprophylaxis with LMWH, UFH or fondaparinux (Grade 1 B) 6. 0. 1. For acutely ill medical patients admitted to hospital with congestive heart failure or severe respiratory disease, or who are confined to bed and have one or more additional risk factors, including active cancer, previous VTE, sepsis, acute neurologic disease, or inflammatory bowel disease, we recommend thromboprophylaxis with LMWH (Grade 1 A), LDUH (Grade 1 A), or fondaparinux (Grade 1 A) 2. 4. For acutely ill hospitalized medical patients at low risk of thrombosis, we recommend against the use of pharmacologic prophylaxis or mechanical prophylaxis (Grade 1 B).

What’s New in the ACCP Guidelines • • • Decrease in 1 A recommendations Orthopedic prophylaxis Mechanical Prophylaxis VTE prophylaxis in hospitalized medical patients Risk Assessment Models, endorsement and extrapolation

Patients Undergoing General, GI, Urologic, Gynecologic, Bariatric, Vascular, Plastic, or Reconstructive Surgery For general and abdominal-pelvic surgery patients at moderate risk for VTE (~ 3. 0%; Rogers score, > 10; Caprini score, 3 -4) who are not at high risk for major bleeding complications, we suggest LMWH (Grade 2 B), LDUH (Grade 2 B), or mechanical prophylaxis, preferably with IPC (Grade 2 C), over no prophylaxis. Remarks: Three of the seven authors favored a strong (Grade 1 B) recommendation in favor of LMWH or LDUH over no prophylaxis in this group.

Rogers and Caprini Models in Surgical Patients • Endorsed by ACCP • Acknowledged that Rogers method is not practical • Caprini model said to be fairly easy to use – Collaborative improvement experience indicates otherwise!!!! • No mention of “ 3 bucket model” • Caprini model validation study ----only 10% at level very low, low risk that do not require AC
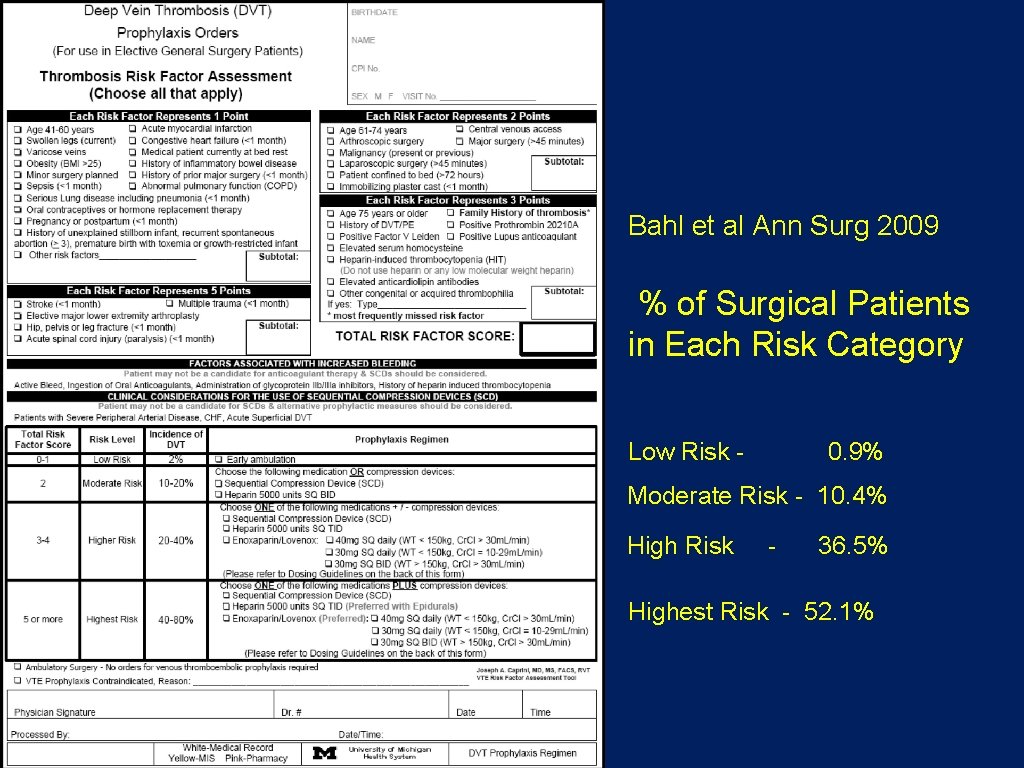
Bahl et al Ann Surg 2009 % of Surgical Patients in Each Risk Category Low Risk - 0. 9% Moderate Risk - 10. 4% High Risk - 36. 5% Highest Risk - 52. 1%

New Guidelines: Comments / Insights / Implications • Controversial guidelines notable for lack of practical guidance • Seemingly, one set of biased assumptions has been replaced by another, skewed in opposite direction • Recommended risk models cumbersome • Recommended risk models relatively untested in terms of inter-observer agreement and efficacy • Dozens in collaboratives have replicated UCSD results…. fewer VTE, no increase in bleeding • Valid points: Some inpatients not at significant risk, attention to possible over anticoagulation is warranted. • Carve outs? CABG patients reasonable • Ortho-----depends on your local culture

Other pearls Recent ACP guidelines Extended prophylaxis New agents

ACP guideline • Risk assessment needed for medical patients(thromboembolism and bleeding risk) • Pharmacologic prophylaxis with heparin or related drug for VTE in medical (stroke) patients • Avoid universal prophylaxis policies

Extended Prophylaxis • Cancer surgery: 4 weeks • 2008 ACCP • THR: 10 days to 35 days • 2007 AAOS • ASA up to 6 weeks • LMWH: 7 -12 days • Warfarin 2 -6 weeks • 2011 AAOS • Unable to make recommendations • 2012 ACCP • 10 -14 days orthopedic, hip fracture, THR/TKR • Up to 35 days

Patients Undergoing General, GI, Urologic, Gynecologic, Bariatric, Vascular, Plastic, or Reconstructive Surgery For high-VTE-risk patients undergoing abdominal or pelvic surgery for cancer who are not otherwise at high risk for major bleeding complications, we recommend extended-duration pharmacologic prophylaxis (4 weeks) with LMWH over limited-duration prophylaxis (Grade 1 B). Remarks: Patients who place a high value on minimizing out-ofpocket health-care costs might prefer limited-duration over extended-duration prophylaxis in settings where the cost of extended-duration prophylaxis is borne by the patient.

Patients Undergoing Major Orthopedic Surgery: Total Hip Arthroplasty (THA), Total Knee Arthroplasty (TKA), Hip Fracture Surgery (HFS) In patients undergoing hip fracture surgery (HFS), we recommend use of one of the following rather than no antithrombotic prophylaxis for a minimum of 10 to 14 days: LMWH, fondaparinux, LDUH, adjusted-dose VKA, aspirin (all Grade 1 B), or an IPCD (Grade 1 C). Remarks: We recommend the use of only portable, battery-powered devices capable of recording and reporting proper wear time on a daily basis for inpatients and outpatients. Efforts should be made to achieve 18 h of daily compliance. One panel member believed strongly that aspirin alone should not be included as an option.

Patients Undergoing Major Orthopedic Surgery: Total Hip Arthroplasty (THA), Total Knee Arthroplasty (TKA), Hip Fracture Surgery (HFS) For patients undergoing major orthopedic surgery, we suggest extending thromboprophylaxis in the outpatient period for up to 35 days from the day of surgery rather than for only 10 to 14 days (Grade 2 B).

NEWER AGENTS
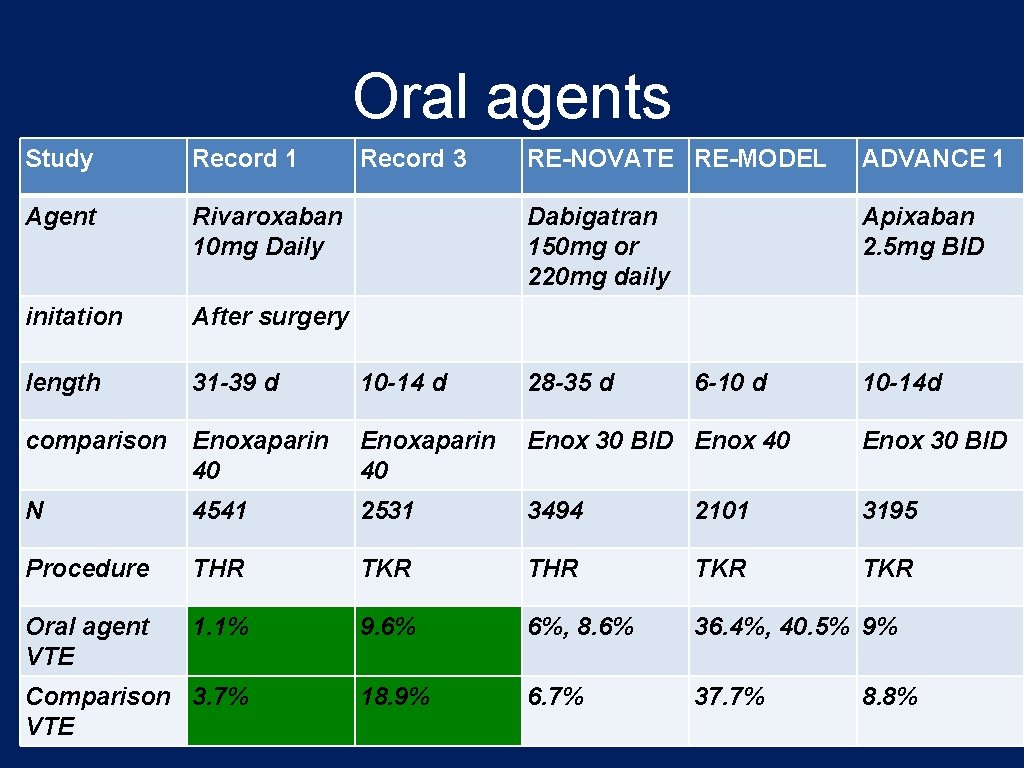
Oral agents Study Record 1 Agent Rivaroxaban 10 mg Daily initation After surgery length 31 -39 d 10 -14 d 28 -35 d comparison Enoxaparin 40 Enox 30 BID Enox 40 Enox 30 BID N 4541 2531 3494 2101 3195 Procedure THR TKR TKR Oral agent VTE 1. 1% 9. 6% 6%, 8. 6% 36. 4%, 40. 5% 9% 18. 9% 6. 7% 37. 7% Comparison 3. 7% VTE Record 3 RE-NOVATE RE-MODEL ADVANCE 1 Dabigatran 150 mg or 220 mg daily Apixaban 2. 5 mg BID 6 -10 d 10 -14 d 8. 8%

Bottom line • VTE prevention: safe/effective/cost effective • Do Risk assessment/Bleeding assessment • Quality Improvement team, using deliberate QI strategy • High reliability strategies: “measure-vention” • Avoid “mechanical only” in pts at risk and have no CI to pharmacological

Thanks! jordan. messler@baycare. org
- Slides: 85