VSEPR THEORY Valence Shell Electron Pair Repulsion Theory







- Slides: 11

VSEPR THEORY (Valence Shell Electron Pair Repulsion Theory) Take notes on the slides Adapted from Mr. M. Mc. Isaac Carleton North High School, Bristol, NB t

n While going through this powerpoint, visit the following website to see the animated VSEPR Shapes n http: //www. chemmybear. com/shapes. ht ml n Click on each of the animated shapes 2

What Is The VSEPR Theory? n n VSEPR Theory is used to predict the shapes of molecules. Think of bonded pairs or BP (shared) or lone pairs or LP (nonbonded, unshared) n n To achieve the most stable condition: n n n e-’s are negatively charged clouds that repel each other. clouds must be as far apart as possible in 3 -D, thereby decreasing repulsion. The amount of repulsion can be ordered: LP-LP > LP-BP > BP-BP In order to determine the shape, the Lewis dot structures must be drawn first. 3

2 Bond Pairs/Electron Groups n n Molecules that only have 2 bonding pairs(BP) on the central atom will have a LINEAR SHAPE with a bond angle of 180° e. g. Be. F 2, CO 2, CS 2 General Formula: AX 2 Central atom A from group 2; 2 BP 0 LP 4

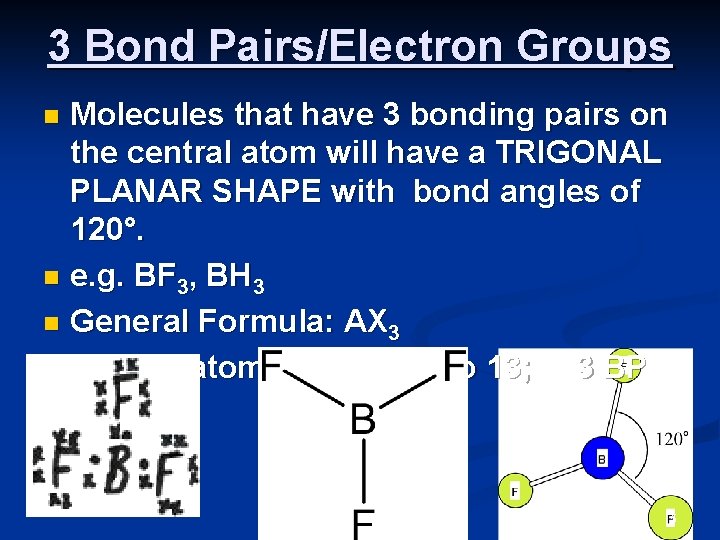
3 Bond Pairs/Electron Groups Molecules that have 3 bonding pairs on the central atom will have a TRIGONAL PLANAR SHAPE with bond angles of 120°. n e. g. BF 3, BH 3 n General Formula: AX 3 n Central atom A from group 13; 3 BP 0 LP n 5

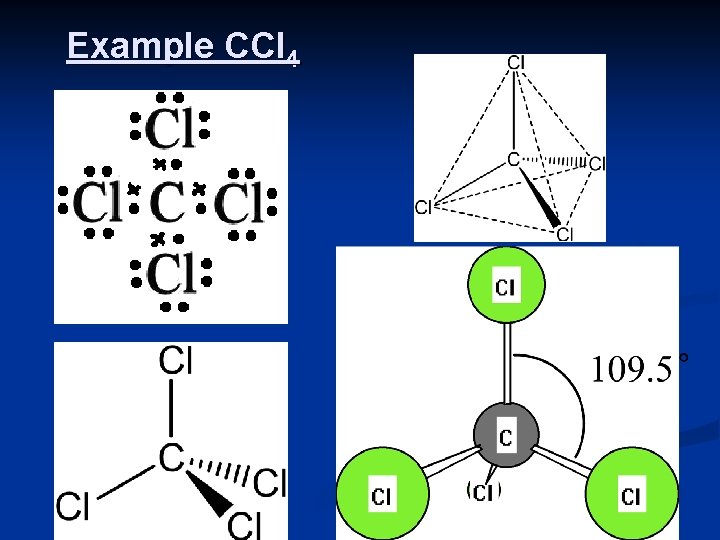
4 Bonding Pairs/Electron Groups n If the central atom is placed at the center of a sphere, than each of the four pairs of electrons will occupy a position to be as far apart as possible. n n n results in the electron pairs being at the corners of a regular tetrahedron these molecules are said to have a TETRAHEDRAL SHAPE. The angle between each bond will be 109. 5° e. g. CCl 4, CH 4, Si. H 4 General Formula: AX 4 Central atom A from group 14; 4 BP 0 LP 6

Example CCl 4 7

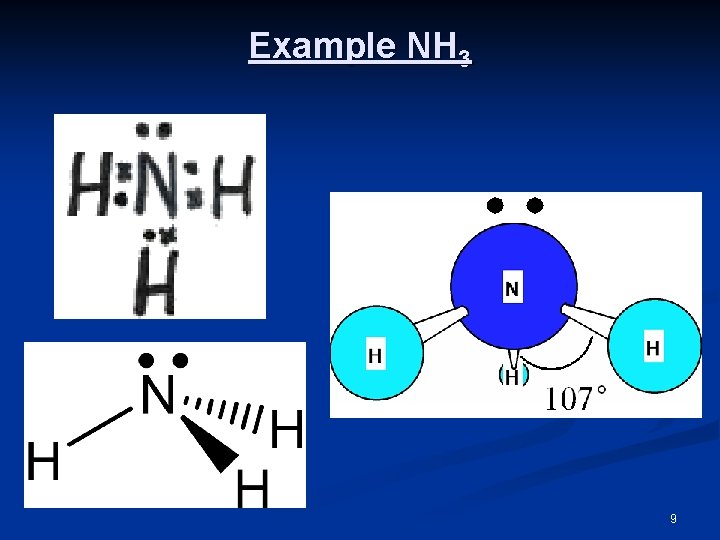
3 Bonding Pairs(BP) & 1 Nonbonding (LP) Pair n n n n Four pairs of electrons will always arrange themselves tetrahedrally around the central atom. The shape of the molecule is determined by the arrangement of the atoms not the electrons. As a result such molecules will have a TRIGONAL PYRAMIDAL shape. Due to the repulsion, a non-bonding electron pair (LP) requires more space than a bonding pair (BP), the angles in these molecules are 107° not 109. 5° as in the tetrahedral molecules. e. g. NH 3, PCl 3 General Formula: AX 3 E Central atom A from group 15; 3 BP 1 LP 8

Example NH 3 9

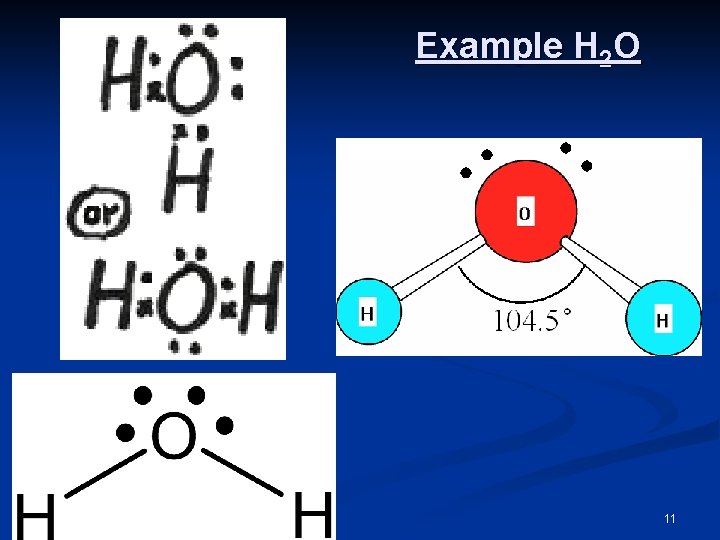
2 Bonding Pairs (BP)& 2 Nonbonding Pairs (LP) n n n n The four pairs of electrons will be arranged tetrahedrally but since only 2 pairs are bonding electrons, the surrounding atoms are at 2 corners of the tetrahedron. As a result these molecules will have a VSHAPE or BENT. The repulsion between the non-bonding pairs (LP) will result in a bond angle of 104. 5°. For each pair of non-bonding electrons, the bond angle decreases by 2. 5° e. g. H 2 O, H 2 S, OCl 2 General Formula: AX 2 E 2 Central atom A from group 16; 2 BP 2 LP 10

Example H 2 O 11