Vorapaxar a Platelet Thrombin Receptor Antagonist in Acute

Vorapaxar, a Platelet Thrombin Receptor Antagonist, in Acute Coronary Syndromes Kenneth W. Mahaffey, MD, on behalf of the TRACER Investigators and Committees Trial funded by Merck Complete financial disclosures: www. DCRI. org
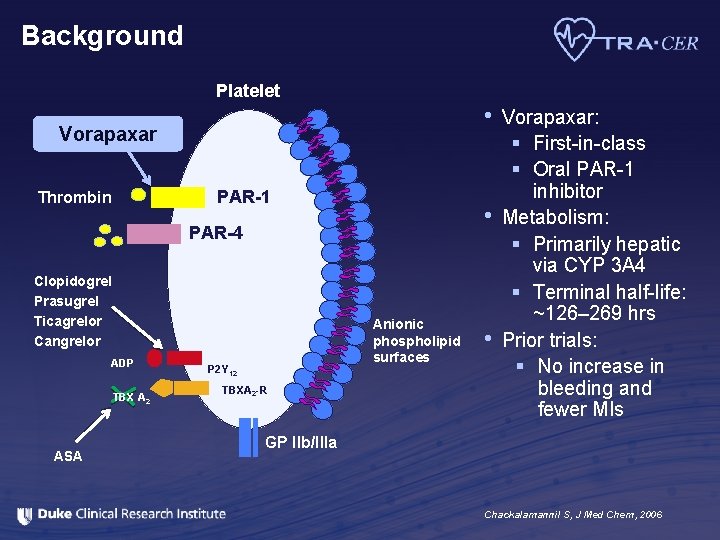
Background Platelet • Vorapaxar PAR-1 Thrombin • PAR-4 Clopidogrel Prasugrel Ticagrelor Cangrelor ADP TBX A 2 ASA Anionic phospholipid surfaces P 2 Y 12 TBXA 2 -R • Vorapaxar: § First-in-class § Oral PAR-1 inhibitor Metabolism: § Primarily hepatic via CYP 3 A 4 § Terminal half-life: ~126– 269 hrs Prior trials: § No increase in bleeding and fewer MIs GP IIb/IIIa Chackalamannil S, J Med Chem, 2006
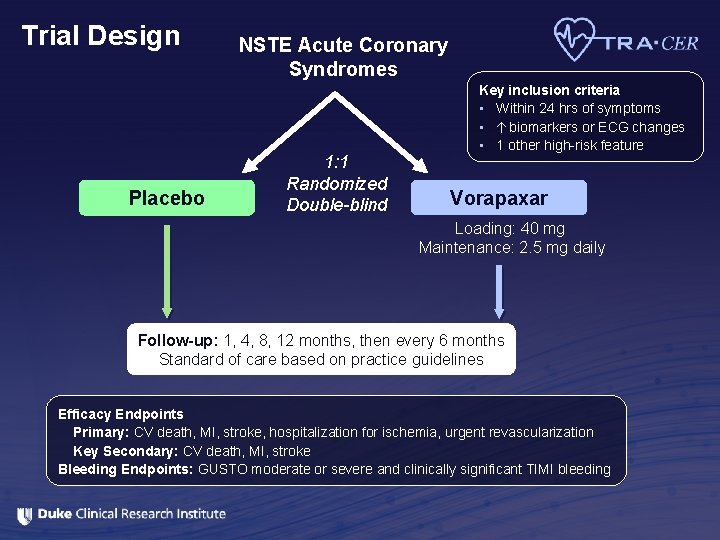
Trial Design Placebo NSTE Acute Coronary Syndromes 1: 1 Randomized Double-blind Key inclusion criteria • Within 24 hrs of symptoms • biomarkers or ECG changes • 1 other high-risk feature Vorapaxar Loading: 40 mg Maintenance: 2. 5 mg daily Follow-up: 1, 4, 8, 12 months, then every 6 months Standard of care based on practice guidelines Efficacy Endpoints Primary: CV death, MI, stroke, hospitalization for ischemia, urgent revascularization Key Secondary: CV death, MI, stroke Bleeding Endpoints: GUSTO moderate or severe and clinically significant TIMI bleeding

Statistical Considerations • • • Sample size § Event-driven with estimated 15% reduction § Power: • 1900 primary endpoint events >95% • 1457 key secondary endpoint events ≥ 90% § 12, 500 patients Analysis § Efficacy analyses: intention-to-treat § Bleeding analyses: all subjects with ≥ 1 dose and on drug § Hierarchical testing procedure to control overall type 1 error January 8, 2011: DSMB recommended to stop follow-up in the trial
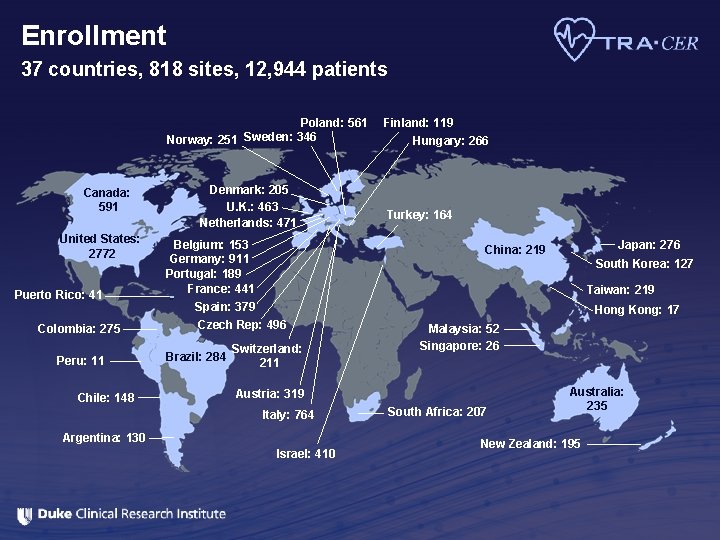
Enrollment 37 countries, 818 sites, 12, 944 patients Poland: 561 Norway: 251 Sweden: 346 Canada: 591 United States: 2772 Puerto Rico: 41 Colombia: 275 Peru: 11 Chile: 148 Denmark: 205 U. K. : 463 Netherlands: 471 Belgium: 153 Germany: 911 Portugal: 189 France: 441 Hungary: 266 Turkey: 164 Japan: 276 China: 219 South Korea: 127 Taiwan: 219 Spain: 379 Czech Rep: 496 Brazil: 284 Finland: 119 Switzerland: 211 Hong Kong: 17 Malaysia: 52 Singapore: 26 Austria: 319 Italy: 764 Argentina: 130 Israel: 410 South Africa: 207 Australia: 235 New Zealand: 195
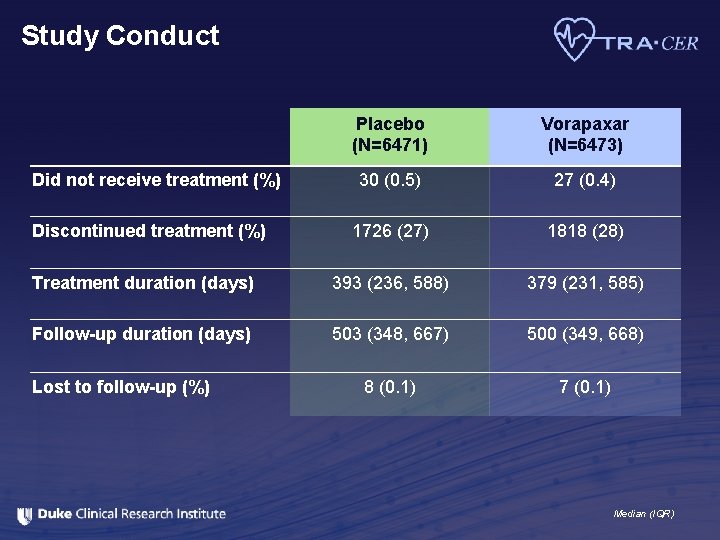
Study Conduct Placebo (N=6471) Vorapaxar (N=6473) 30 (0. 5) 27 (0. 4) 1726 (27) 1818 (28) Treatment duration (days) 393 (236, 588) 379 (231, 585) Follow-up duration (days) 503 (348, 667) 500 (349, 668) 8 (0. 1) 7 (0. 1) Did not receive treatment (%) Discontinued treatment (%) Lost to follow-up (%) Median (IQR)
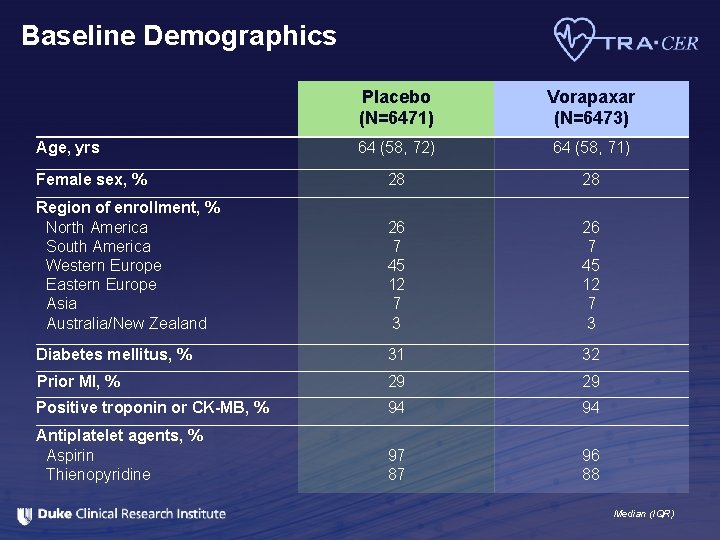
Baseline Demographics Placebo (N=6471) Vorapaxar (N=6473) 64 (58, 72) 64 (58, 71) Female sex, % 28 28 Region of enrollment, % North America South America Western Europe Eastern Europe Asia Australia/New Zealand 26 7 45 12 7 3 Diabetes mellitus, % 31 32 Prior MI, % 29 29 Positive troponin or CK-MB, % 94 94 Antiplatelet agents, % Aspirin Thienopyridine 97 87 96 88 Age, yrs Median (IQR)
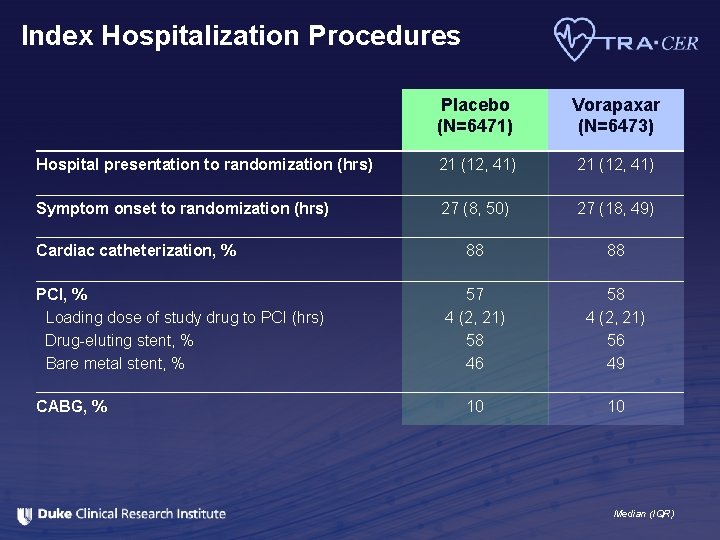
Index Hospitalization Procedures Placebo (N=6471) Vorapaxar (N=6473) Hospital presentation to randomization (hrs) 21 (12, 41) Symptom onset to randomization (hrs) 27 (8, 50) 27 (18, 49) 88 88 57 4 (2, 21) 58 46 58 4 (2, 21) 56 49 10 10 Cardiac catheterization, % PCI, % Loading dose of study drug to PCI (hrs) Drug-eluting stent, % Bare metal stent, % CABG, % Median (IQR)
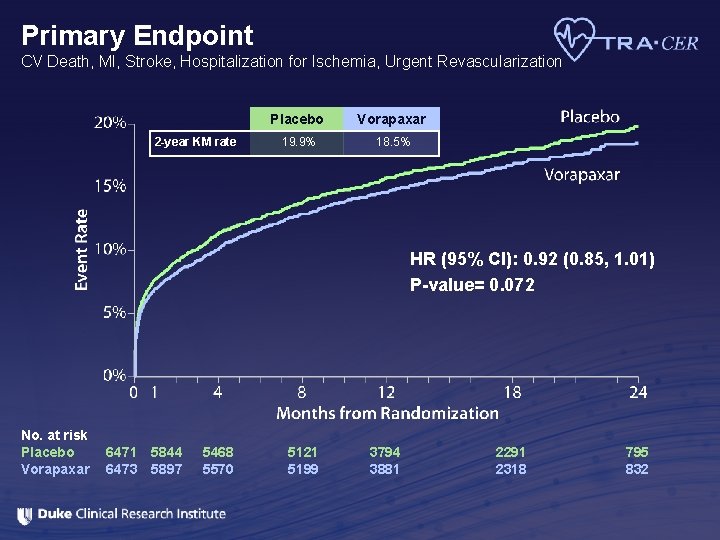
Primary Endpoint CV Death, MI, Stroke, Hospitalization for Ischemia, Urgent Revascularization 2 -year KM rate Placebo Vorapaxar 19. 9% 18. 5% HR (95% CI): 0. 92 (0. 85, 1. 01) P-value= 0. 072 No. at risk Placebo Vorapaxar 6471 5844 6473 5897 5468 5570 5121 5199 3794 3881 2291 2318 795 832
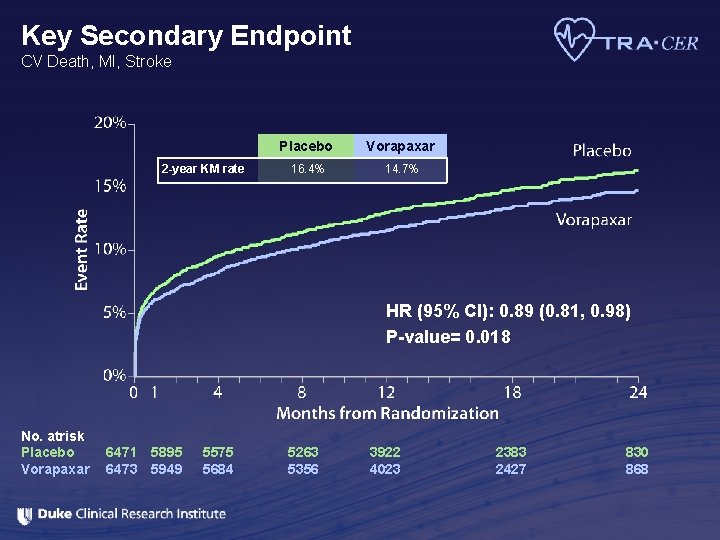
Key Secondary Endpoint CV Death, MI, Stroke 2 -year KM rate Placebo Vorapaxar 16. 4% 14. 7% HR (95% CI): 0. 89 (0. 81, 0. 98) P-value= 0. 018 No. atrisk Placebo Vorapaxar 6471 5895 6473 5949 5575 5684 5263 5356 3922 4023 2383 2427 830 868
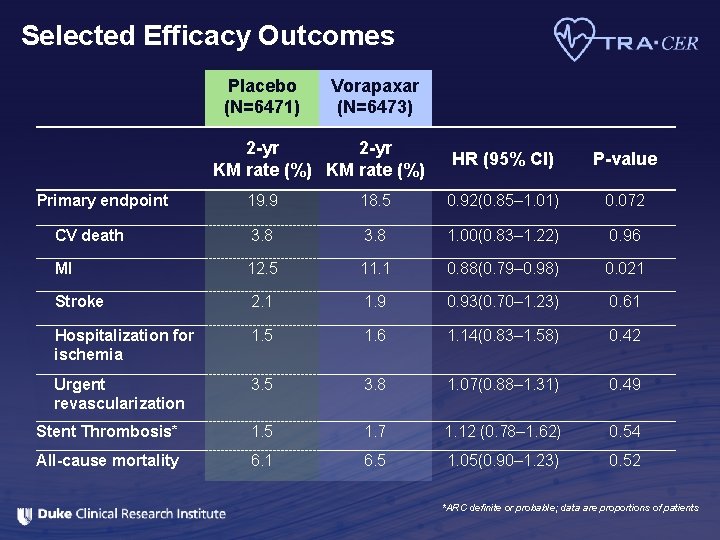
Selected Efficacy Outcomes Placebo (N=6471) Vorapaxar (N=6473) 2 -yr KM rate (%) Primary endpoint HR (95% CI) P-value 19. 9 18. 5 0. 92(0. 85– 1. 01) 0. 072 CV death 3. 8 1. 00(0. 83– 1. 22) 0. 96 MI 12. 5 11. 1 0. 88(0. 79– 0. 98) 0. 021 Stroke 2. 1 1. 9 0. 93(0. 70– 1. 23) 0. 61 Hospitalization for ischemia 1. 5 1. 6 1. 14(0. 83– 1. 58) 0. 42 Urgent revascularization 3. 5 3. 8 1. 07(0. 88– 1. 31) 0. 49 Stent Thrombosis* 1. 5 1. 7 1. 12 (0. 78– 1. 62) 0. 54 All-cause mortality 6. 1 6. 5 1. 05(0. 90– 1. 23) 0. 52 *ARC definite or probable; data are proportions of patients
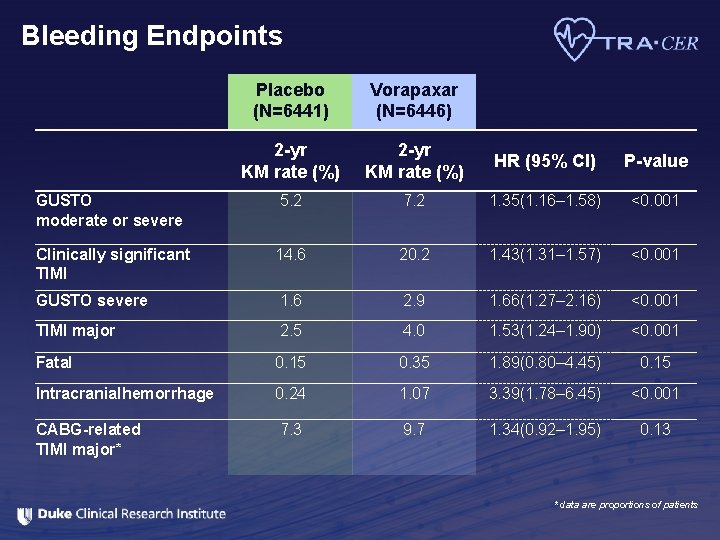
Bleeding Endpoints Placebo (N=6441) Vorapaxar (N=6446) 2 -yr KM rate (%) HR (95% CI) P-value GUSTO moderate or severe 5. 2 7. 2 1. 35(1. 16– 1. 58) <0. 001 Clinically significant TIMI 14. 6 20. 2 1. 43(1. 31– 1. 57) <0. 001 GUSTO severe 1. 6 2. 9 1. 66(1. 27– 2. 16) <0. 001 TIMI major 2. 5 4. 0 1. 53(1. 24– 1. 90) <0. 001 Fatal 0. 15 0. 35 1. 89(0. 80– 4. 45) 0. 15 Intracranialhemorrhage 0. 24 1. 07 3. 39(1. 78– 6. 45) <0. 001 CABG-related TIMI major* 7. 3 9. 7 1. 34(0. 92– 1. 95) 0. 13 * data are proportions of patients
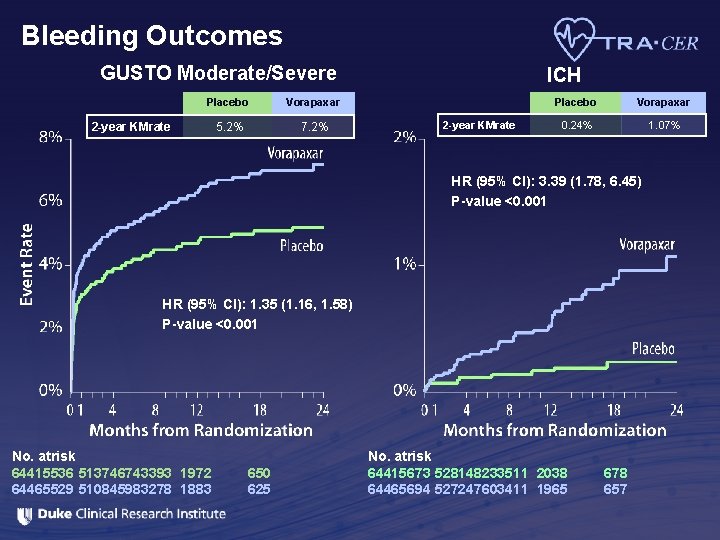
Bleeding Outcomes GUSTO Moderate/Severe Placebo Vorapaxar 5. 2% 7. 2% 2 -year KMrate ICH 2 -year KMrate Placebo Vorapaxar 0. 24% 1. 07% HR (95% CI): 3. 39 (1. 78, 6. 45) P-value <0. 001 HR (95% CI): 1. 35 (1. 16, 1. 58) P-value <0. 001 No. atrisk 64415536 513746743393 1972 64465529 510845983278 1883 650 625 No. atrisk 64415673 528148233511 2038 64465694 527247603411 1965 678 657
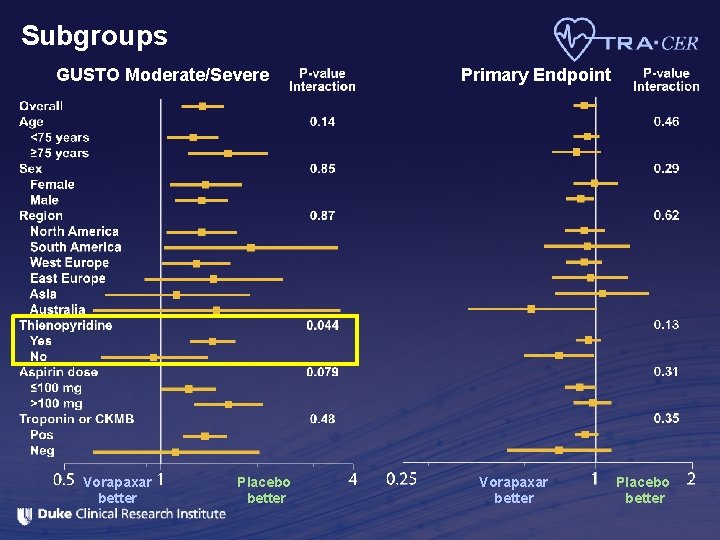
Subgroups GUSTO Moderate/Severe Vorapaxar better Placebo better Primary Endpoint Vorapaxar better Placebo better

Summary When added to standard of care in patients with NSTE ACS and high use of aspirin and P 2 Y 12 inhibition, vorapaxar: • Did not significantly reduce the composite of CV death, MI, stroke, hospitalization for ischemia, or urgent revascularization • Reduced CV death, MI, or stroke • Significantly increased bleeding, including major bleeding and intracranial hemorrhage Whether PAR-1 blockade improves outcomes with different medication strategies or in other patient populations with coronary artery disease requires further study.

Study Organization Executive Committee Academic Research Organizations R Harrington (Chair), P Armstrong, P Aylward, E Chen, K Mahaffey, D Moliterno, J Strony, F Van de Werf, L Wallentin, H White DCRI: P Tricoci, T Rorick, S Leonardi, D Underwood, J Wrestler CVC: P Armstrong, C Sorochuck C 5: A Lincoff, D Mason Henry Ford: M Hudson, D Sydlowski Thomas Jefferson: D Whellan, B Gallagher Flinders: P Aylward, J Garrett Green Lane: H White, S Douglas Leuven: P Sinnaeve, A Beernaert Steering Committee G Ambrosio, A Betriu, C Bode, A Cequier, T Cheem, M Chen, J Cornel, A Dalby, R Diaz, A Erkan, P Grande, C Held, K Huber, M Hudson, Y Huo, D Isaza, J Jukema, M Laine, B Lewis, A Lincoff, J Lixin, G Montalescot, J Nicolau, J Nordrehaug, P Ofner, H Ogawa, S Park, M Pfisterer, J Prieto, L Providencia, W Ruzyllo, P Sinnaeve, R Storey, P Tricoci, M Valgimigli, D Whellan, P Widimsky, L Wong, T Yamaguchi Data & Safety Monitoring Board F Verheugt (Chair), R Frye, J Hochman, P Steg, K Bailey, J Easton Sponsor Merck: E Chen, R Harmelin–Kadouri, A Kilian, S Petrauskas, J Strony CEC Core Lab A Johnson J O’ Briant M Smith P Tricoci ECG: P Armstrong, H Siha Platelets: L Jennings, E Hord Angio: M Gibson, A Chirlin

The full article is now available online at www. nejm. org
- Slides: 17