Voltammetry Biosensors Voltammetry Biosensors electrochemical biological component in

Voltammetry Biosensors

Voltammetry Biosensors (electrochemical) biological component in the recognition layer enzymes DNA, RNA antigens, antibodies (immunosensors) tissues living organisms cells algae bacteria detection usually not with voltammetry but with amperometry measurement of a current at a constant potential potentiometric detection possible with some enzymes

Voltammetry - Biosensors biosensor design working electrodes GCE, CPE, SPE, Pt, Au immobilization of enzymes covalent immobilization to functional groups on surface to functional groups of SAMs bulk modification with heterogeneous materials embedding into bulk entrapment into films on electrode surface Nafion, PVA, Ethylacetate Hydrogels electropolymers entrapment barriers over enzyme layer e. g. , dialysis membrane, gel
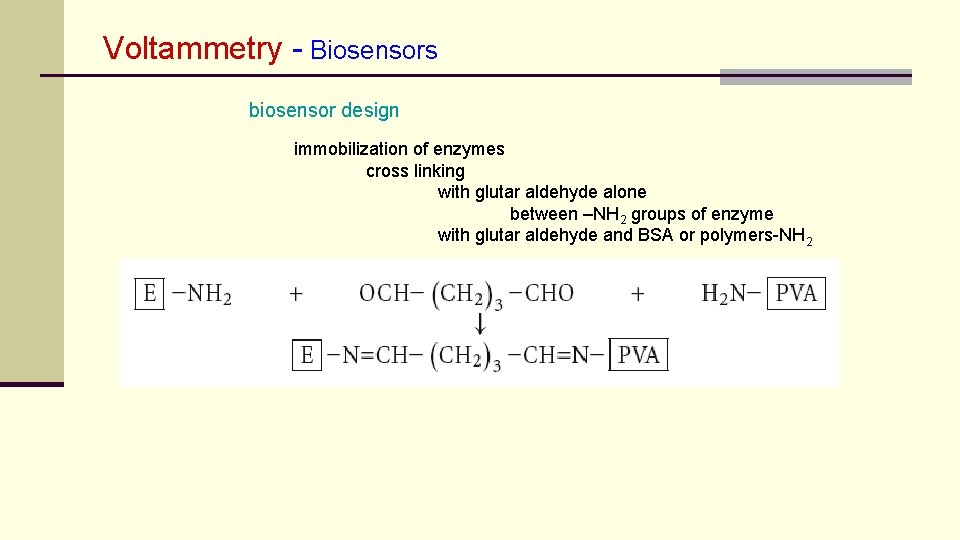
Voltammetry - Biosensors biosensor design immobilization of enzymes cross linking with glutar aldehyde alone between –NH 2 groups of enzyme with glutar aldehyde and BSA or polymers-NH 2
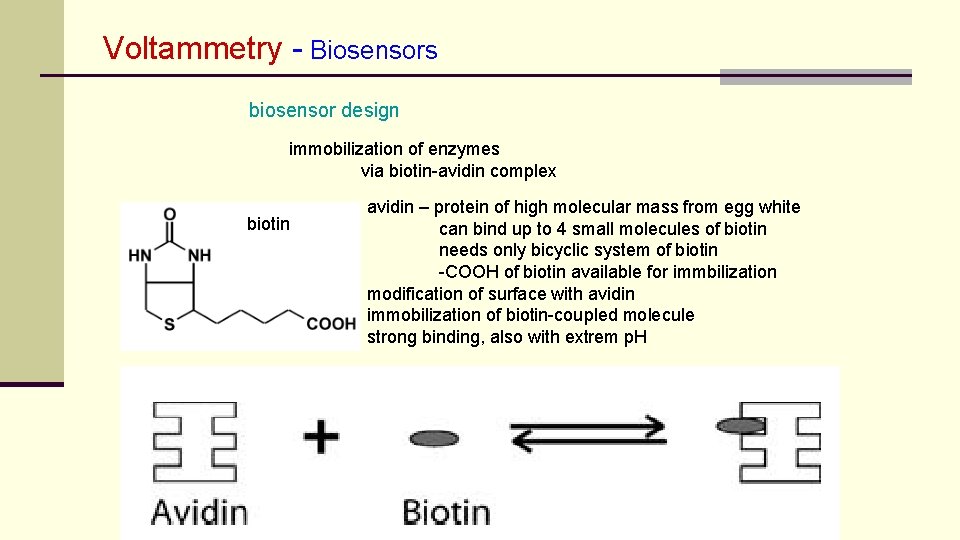
Voltammetry - Biosensors biosensor design immobilization of enzymes via biotin-avidin complex biotin avidin – protein of high molecular mass from egg white can bind up to 4 small molecules of biotin needs only bicyclic system of biotin -COOH of biotin available for immbilization modification of surface with avidin immobilization of biotin-coupled molecule strong binding, also with extrem p. H
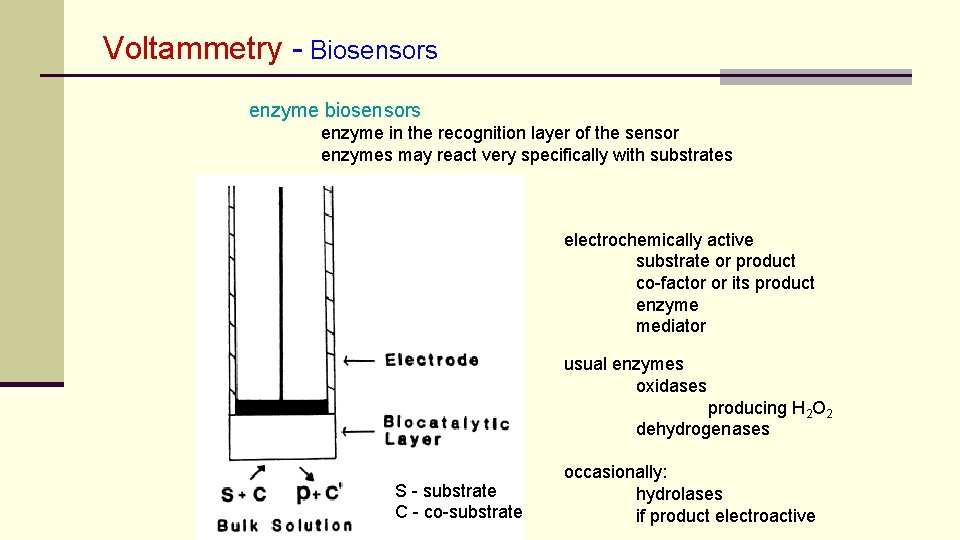
Voltammetry - Biosensors enzyme biosensors enzyme in the recognition layer of the sensor enzymes may react very specifically with substrates electrochemically active substrate or product co-factor or its product enzyme mediator usual enzymes oxidases producing H 2 O 2 dehydrogenases S - substrate C - co-substrate occasionally: hydrolases if product electroactive

Voltammetry – Biosensors enzyme biosensors modification: isolated enzyme tissues oxidases, dehydrogenases change the oxidation state electrochemical conversion often not possible reactive site of enzyme hidden by protein shell co-factor necessary O 2 with oxygenases NAD+ with dehydrogenases may be replaced by mediators hydrogen peroxide intermediate of many oxidases with O 2 as electron acceptor may be detected with platinum electrodes high overpotential with carbon electrodes for ist oxidation to O 2 or its reduction to H 2 O mediated oxidation/reduction to decrease overpotential
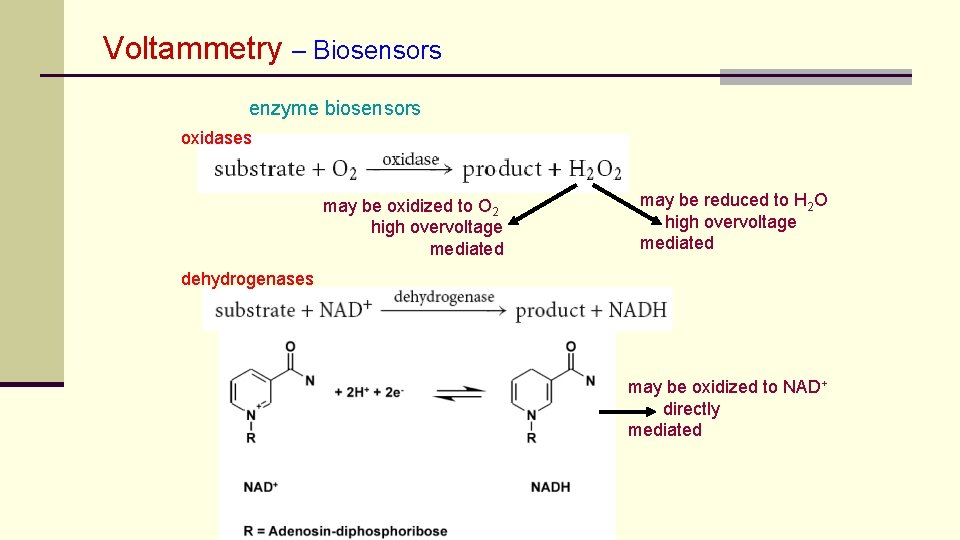
Voltammetry – Biosensors enzyme biosensors oxidases may be oxidized to O 2 high overvoltage mediated may be reduced to H 2 O high overvoltage mediated dehydrogenases may be oxidized to NAD+ directly mediated
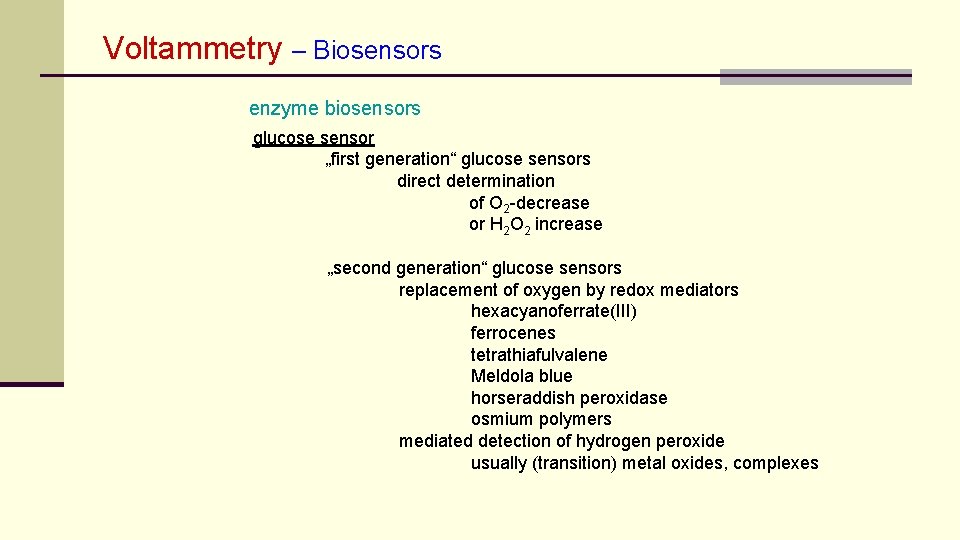
Voltammetry – Biosensors enzyme biosensors glucose sensor „first generation“ glucose sensors direct determination of O 2 -decrease or H 2 O 2 increase „second generation“ glucose sensors replacement of oxygen by redox mediators hexacyanoferrate(III) ferrocenes tetrathiafulvalene Meldola blue horseraddish peroxidase osmium polymers mediated detection of hydrogen peroxide usually (transition) metal oxides, complexes
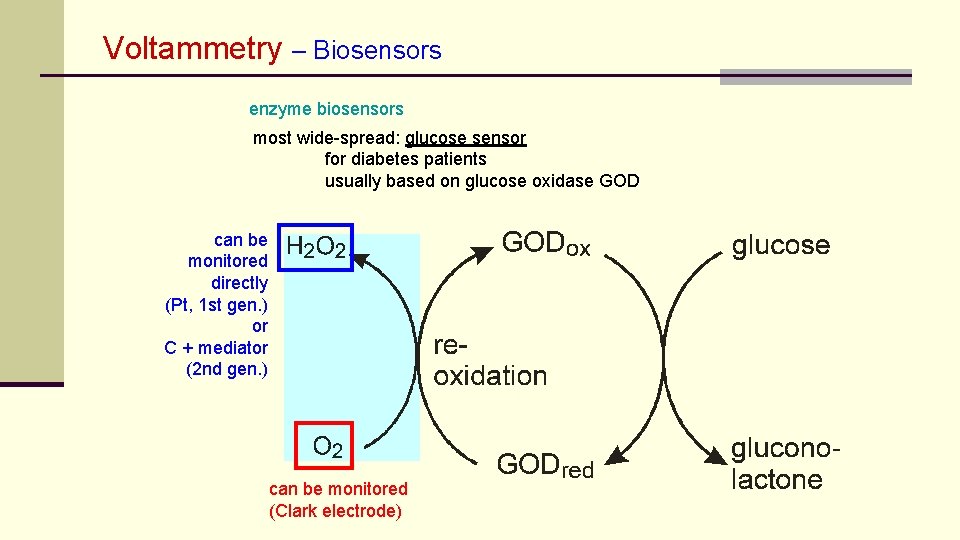
Voltammetry – Biosensors enzyme biosensors most wide-spread: glucose sensor for diabetes patients usually based on glucose oxidase GOD can be monitored directly (Pt, 1 st gen. ) or C + mediator (2 nd gen. ) can be monitored (Clark electrode)
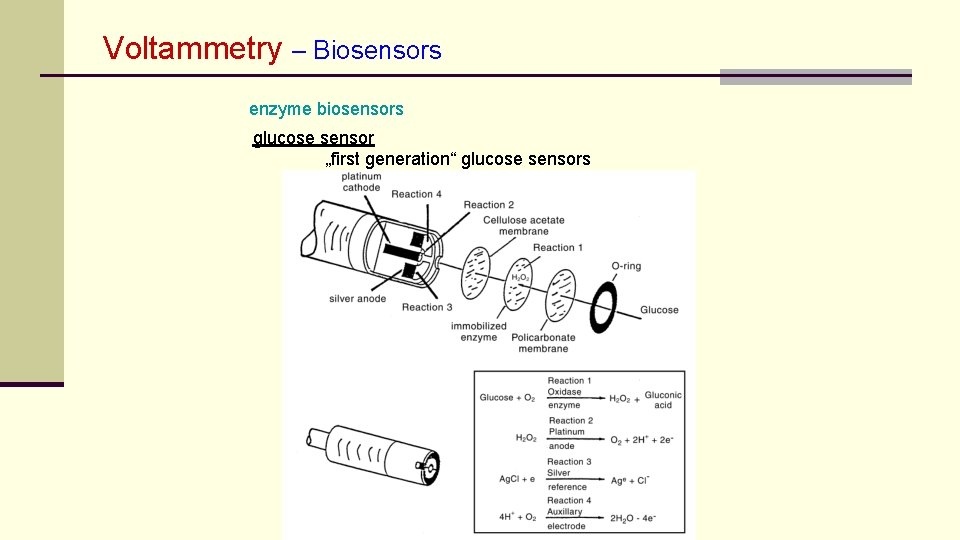
Voltammetry – Biosensors enzyme biosensors glucose sensor „first generation“ glucose sensors
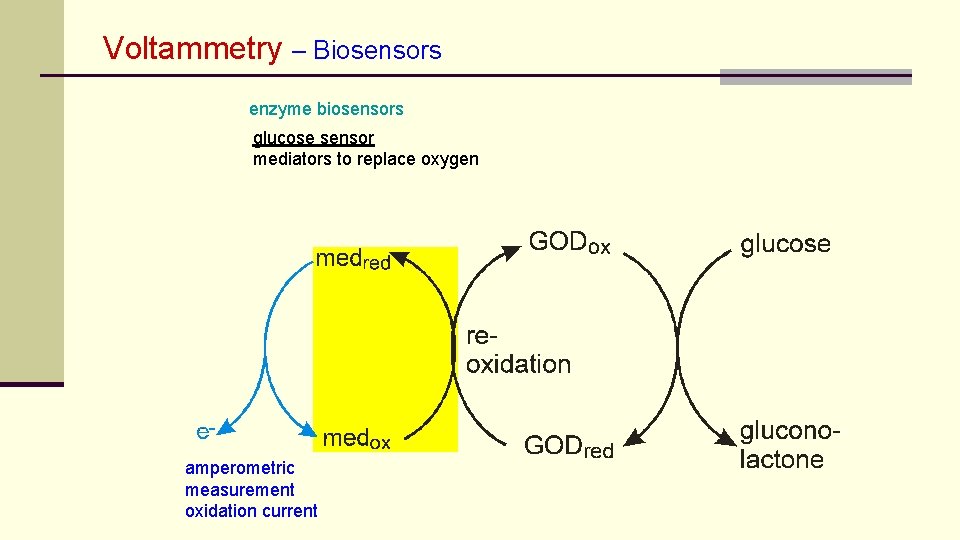
Voltammetry – Biosensors enzyme biosensors glucose sensor mediators to replace oxygen amperometric measurement oxidation current
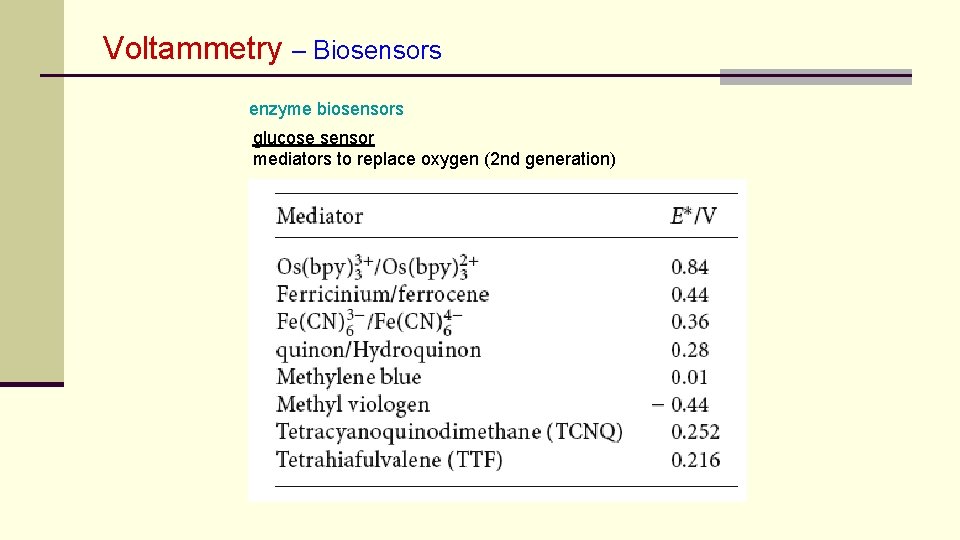
Voltammetry – Biosensors enzyme biosensors glucose sensor mediators to replace oxygen (2 nd generation)
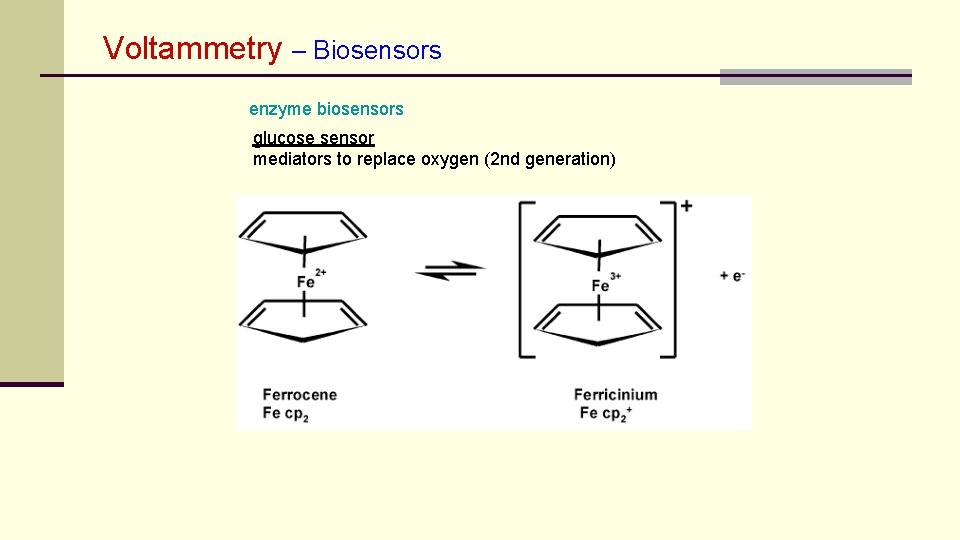
Voltammetry – Biosensors enzyme biosensors glucose sensor mediators to replace oxygen (2 nd generation)
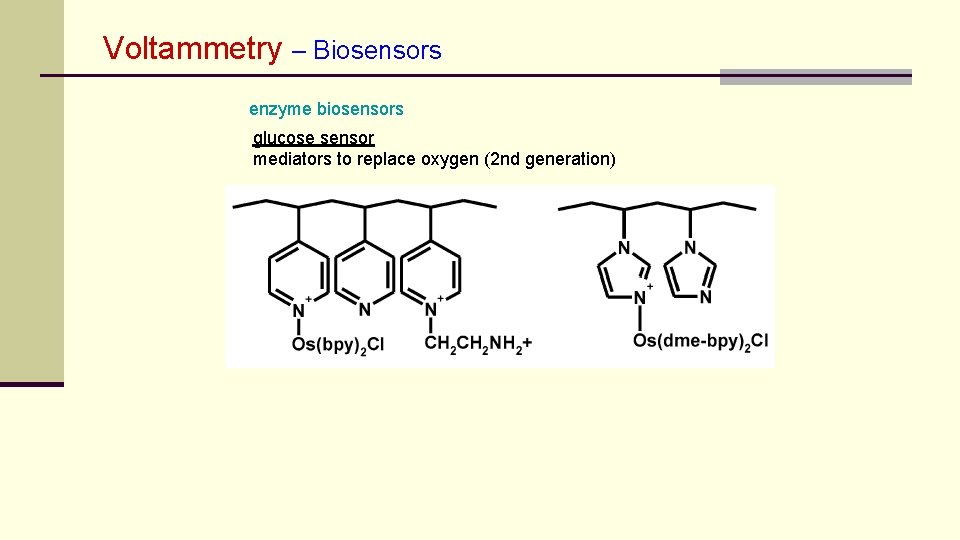
Voltammetry – Biosensors enzyme biosensors glucose sensor mediators to replace oxygen (2 nd generation)
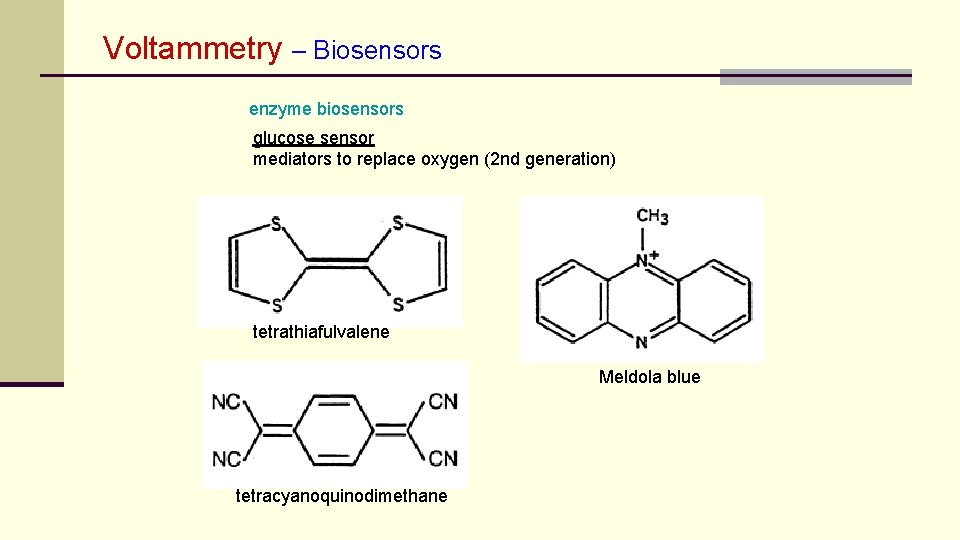
Voltammetry – Biosensors enzyme biosensors glucose sensor mediators to replace oxygen (2 nd generation) tetrathiafulvalene Meldola blue tetracyanoquinodimethane
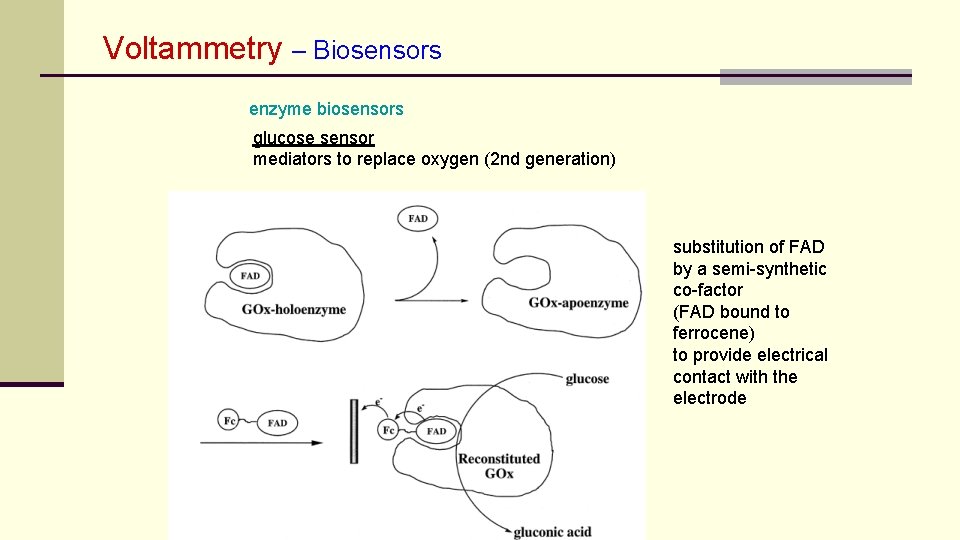
Voltammetry – Biosensors enzyme biosensors glucose sensor mediators to replace oxygen (2 nd generation) substitution of FAD by a semi-synthetic co-factor (FAD bound to ferrocene) to provide electrical contact with the electrode

Voltammetry – Biosensors enzyme biosensors glucose sensor example: detection of H 2 O 2 via mediated oxidation with Mn. O 2 double modified electrodes: Mn. O 2 and GOx
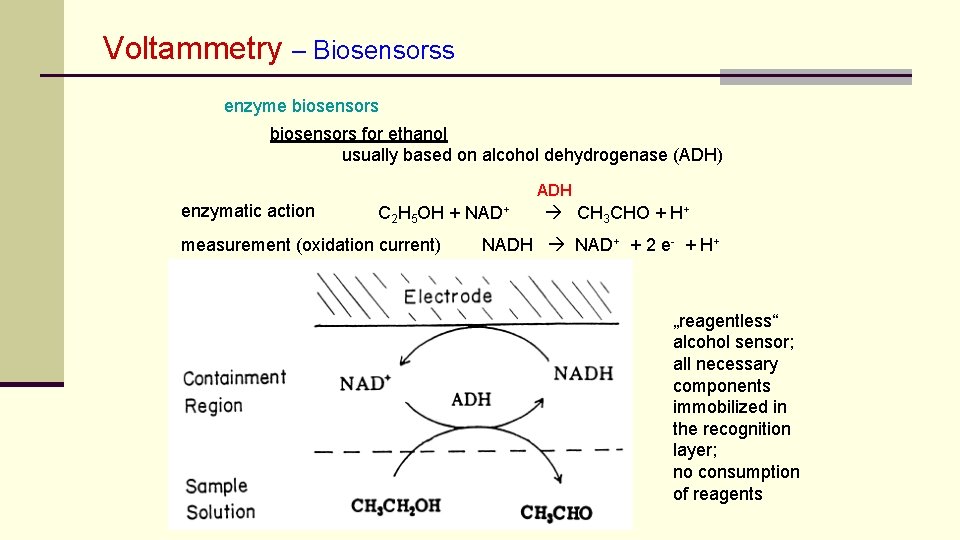
Voltammetry – Biosensorss enzyme biosensors for ethanol usually based on alcohol dehydrogenase (ADH) ADH enzymatic action C 2 H 5 OH + NAD+ measurement (oxidation current) CH 3 CHO + H+ NADH NAD+ + 2 e- + H+ „reagentless“ alcohol sensor; all necessary components immobilized in the recognition layer; no consumption of reagents
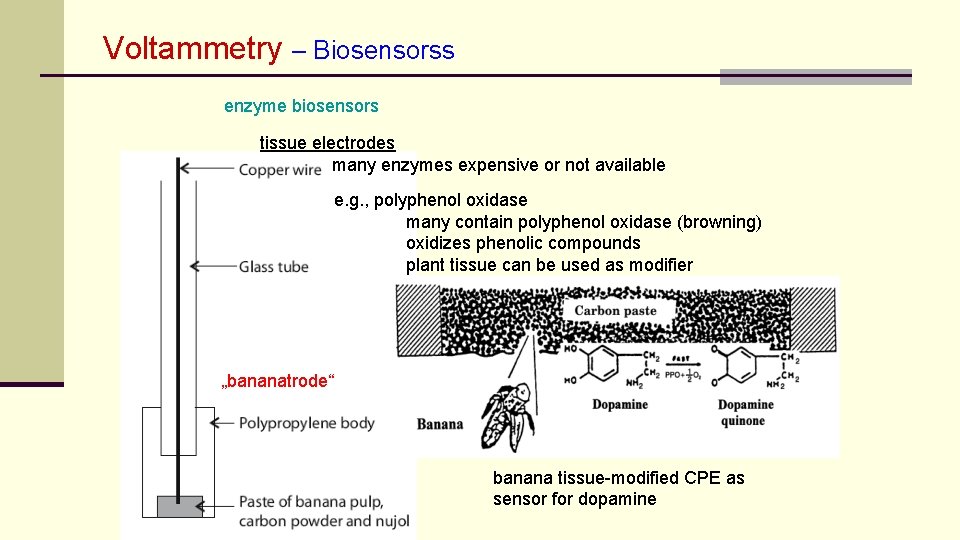
Voltammetry – Biosensorss enzyme biosensors tissue electrodes many enzymes expensive or not available e. g. , polyphenol oxidase many contain polyphenol oxidase (browning) oxidizes phenolic compounds plant tissue can be used as modifier „bananatrode“ banana tissue-modified CPE as sensor for dopamine
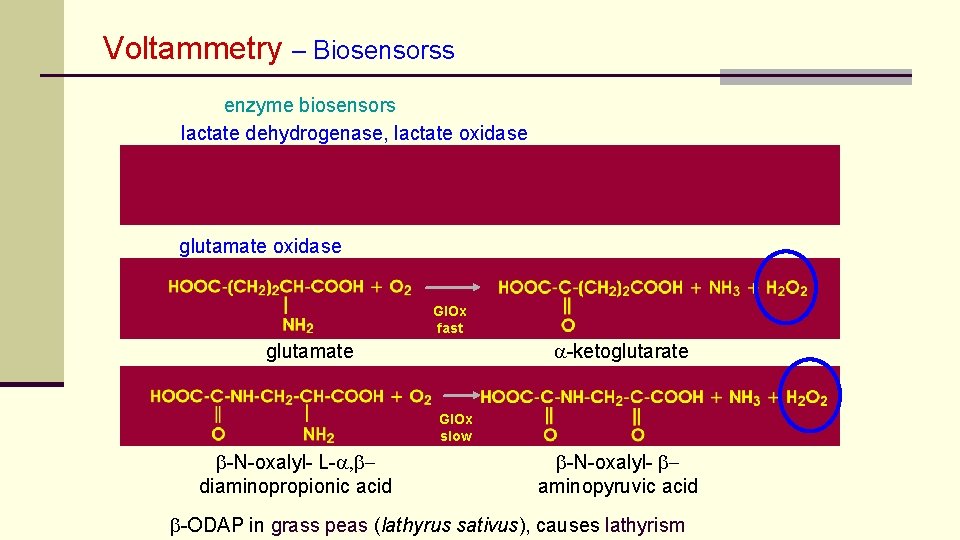
Voltammetry – Biosensorss enzyme biosensors lactate dehydrogenase, lactate oxidase glutamate oxidase Gl. Ox fast a-ketoglutarate glutamate Gl. Ox slow b-N-oxalyl- L-a, bdiaminopropionic acid b-N-oxalyl- baminopyruvic acid b-ODAP in grass peas (lathyrus sativus), causes lathyrism
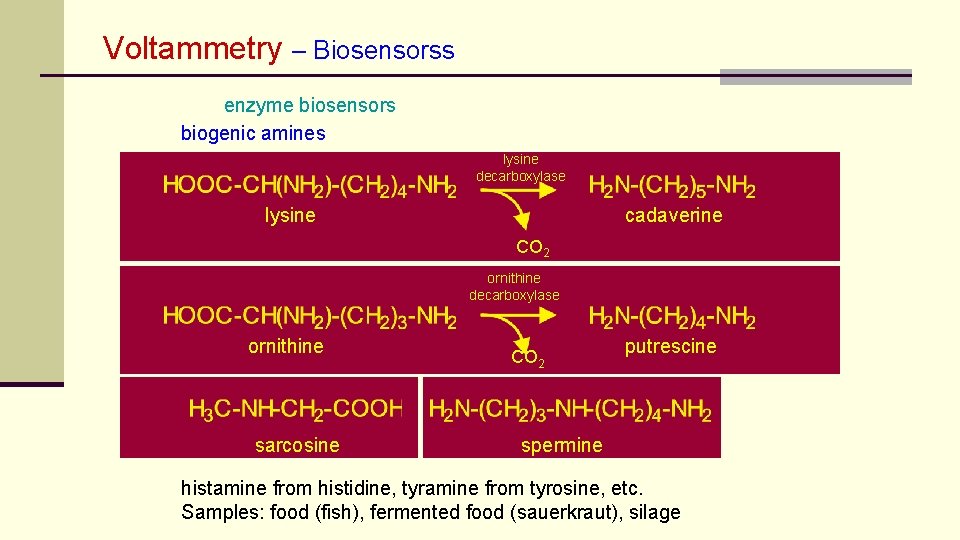
Voltammetry – Biosensorss enzyme biosensors biogenic amines lysine decarboxylase lysine cadaverine CO 2 ornithine decarboxylase ornithine sarcosine CO 2 putrescine spermine histamine from histidine, tyramine from tyrosine, etc. Samples: food (fish), fermented food (sauerkraut), silage
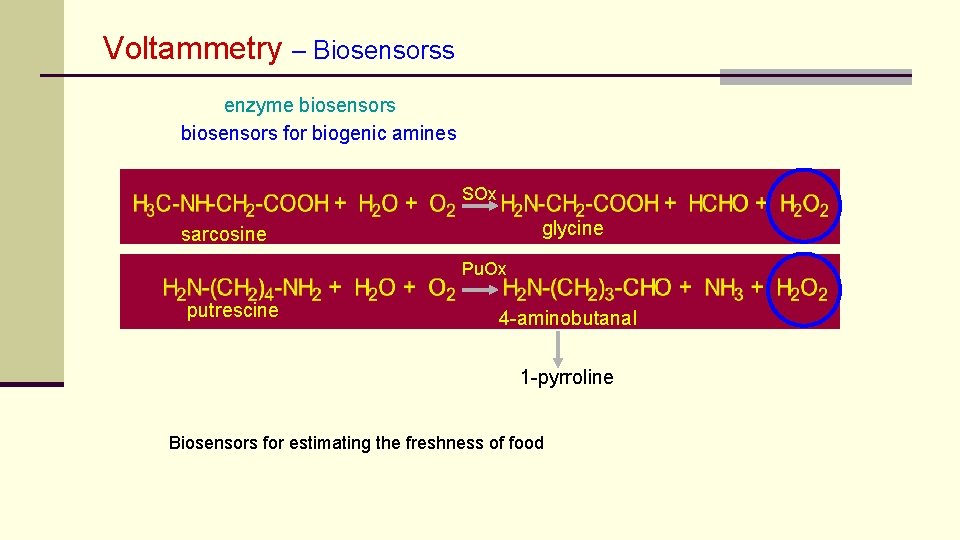
Voltammetry – Biosensorss enzyme biosensors for biogenic amines SOx glycine sarcosine Pu. Ox putrescine 4 -aminobutanal 1 -pyrroline Biosensors for estimating the freshness of food
- Slides: 24