Voltage of a Battery something about available energy
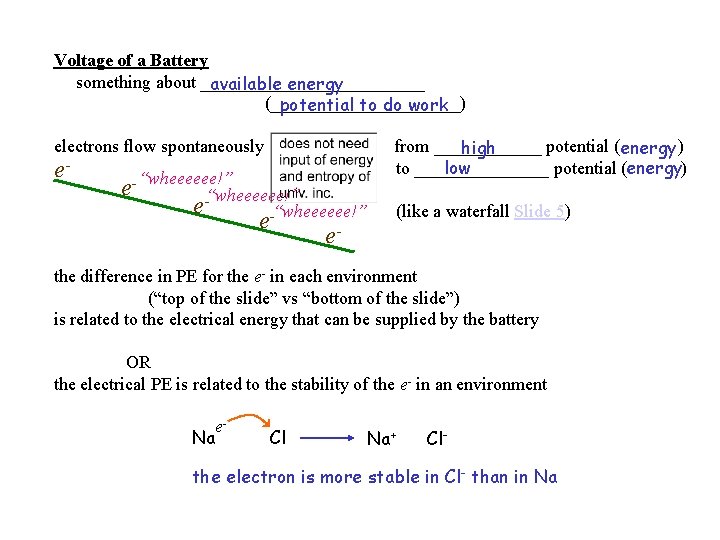
Voltage of a Battery something about _____________ available energy (___________) potential to do work electrons flow spontaneously e- e- “wheeeeee!” e-“wheeeeee!” e- from ______ potential (energy ) high low to ________ potential (energy) (like a waterfall Slide 5) the difference in PE for the e- in each environment (“top of the slide” vs “bottom of the slide”) is related to the electrical energy that can be supplied by the battery OR the electrical PE is related to the stability of the e- in an environment e. Na Cl Na+ Cl- the electron is more stable in Cl- than in Na
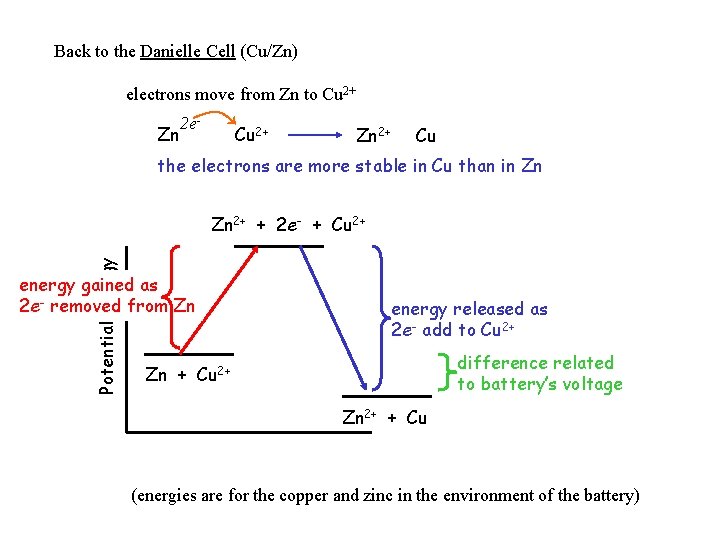
Back to the Danielle Cell (Cu/Zn) electrons move from Zn to Cu 2+ Zn 2 e- Cu 2+ Zn 2+ Cu the electrons are more stable in Cu than in Zn Potential Energy Zn 2+ + 2 e- + Cu 2+ energy gained as 2 e- removed from Zn energy released as 2 e- add to Cu 2+ difference related to battery’s voltage Zn + Cu 2+ Zn 2+ + Cu (energies are for the copper and zinc in the environment of the battery)
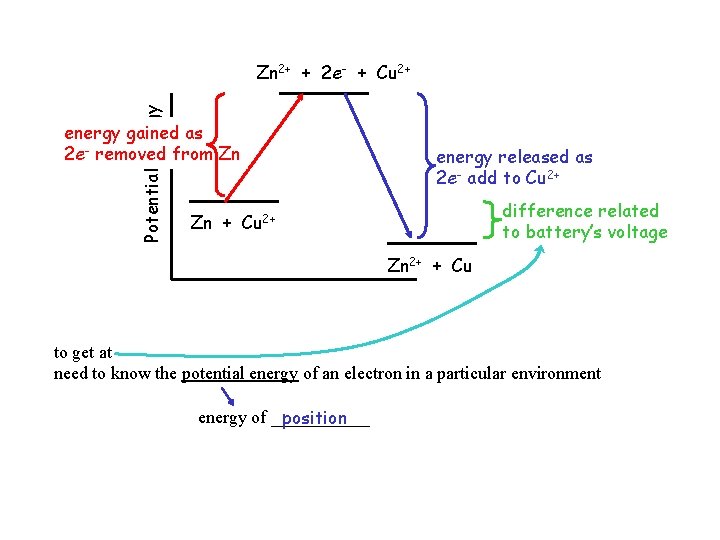
Potential Energy Zn 2+ + 2 e- + Cu 2+ energy gained as 2 e- removed from Zn energy released as 2 e- add to Cu 2+ difference related to battery’s voltage Zn + Cu 2+ Zn 2+ + Cu to get at need to know the potential energy of an electron in a particular environment energy of ______ position
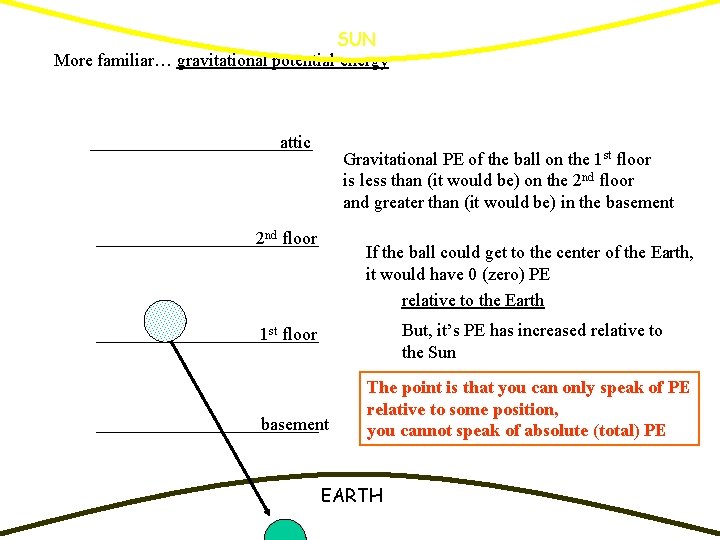
SUN More familiar… gravitational potential energy attic Gravitational PE of the ball on the 1 st floor is less than (it would be) on the 2 nd floor and greater than (it would be) in the basement 2 nd floor If the ball could get to the center of the Earth, it would have 0 (zero) PE relative to the Earth But, it’s PE has increased relative to the Sun 1 st floor basement The point is that you can only speak of PE relative to some position, you cannot speak of absolute (total) PE EARTH

The point is that you can only speak of PE relative to some position, you cannot speak of absolute (total) PE The same is true for electrical PE: can only speak of the relative potential energy of an electron in an environment Zn 2 e- Cu 2+ Zn 2+ Cu the electrons are more stable in Cu than in Zn lower electrical PE So, if the electrons are more stable in Cu than in Zn, which of Zn 2+ + 2 e- Zn Cu 2+ + 2 e- Cu the reduction of Cu 2+ is more likely to happen ? the relative potential energy of the electron in the metal reflects the relative tendency for a metal to be reduced (to its pure form) - the lower the potential energy of an electron in a metal, the greater the tendency for a metal to be reduced (to its pure form)
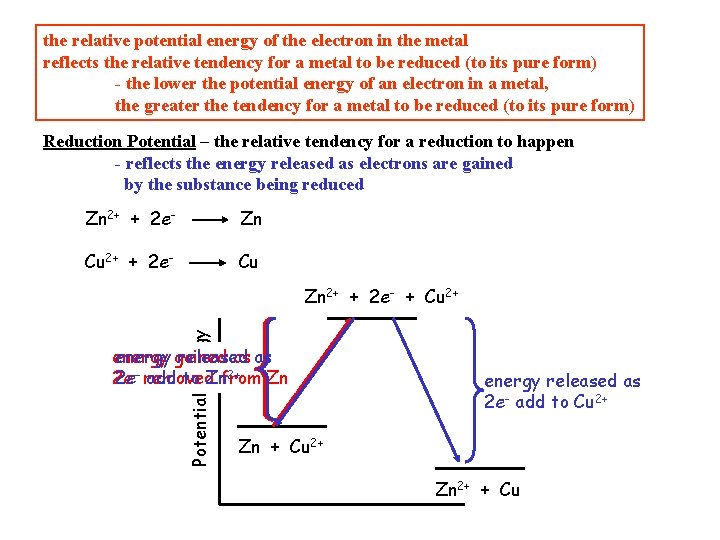
the relative potential energy of the electron in the metal reflects the relative tendency for a metal to be reduced (to its pure form) - the lower the potential energy of an electron in a metal, the greater the tendency for a metal to be reduced (to its pure form) Reduction Potential – the relative tendency for a reduction to happen - reflects the energy released as electrons are gained by the substance being reduced Zn 2+ + 2 e- Zn Cu 2+ + 2 e- Cu Potential Energy Zn 2+ + 2 e- + Cu 2+ energy gained released as as 2+ 2 e 2 e-- removed add to Znfrom Zn energy released as 2 e- add to Cu 2+ Zn + Cu 2+ Zn 2+ + Cu
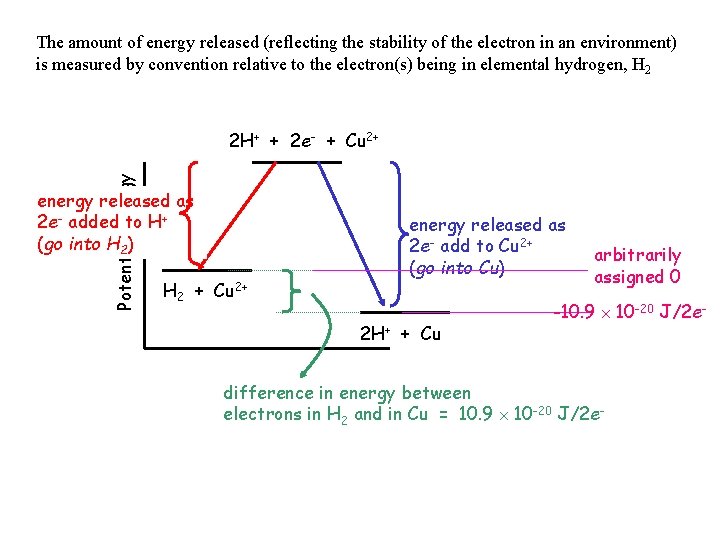
The amount of energy released (reflecting the stability of the electron in an environment) is measured by convention relative to the electron(s) being in elemental hydrogen, H 2 Potential Energy 2 H+ + 2 e- + Cu 2+ energy released as 2 e- added to H+ (go into H 2) H 2 + Cu 2+ energy released as 2 e- add to Cu 2+ (go into Cu) 2 H+ + Cu arbitrarily assigned 0 -10. 9 10 -20 J/2 e- difference in energy between electrons in H 2 and in Cu = 10. 9 10 -20 J/2 e-
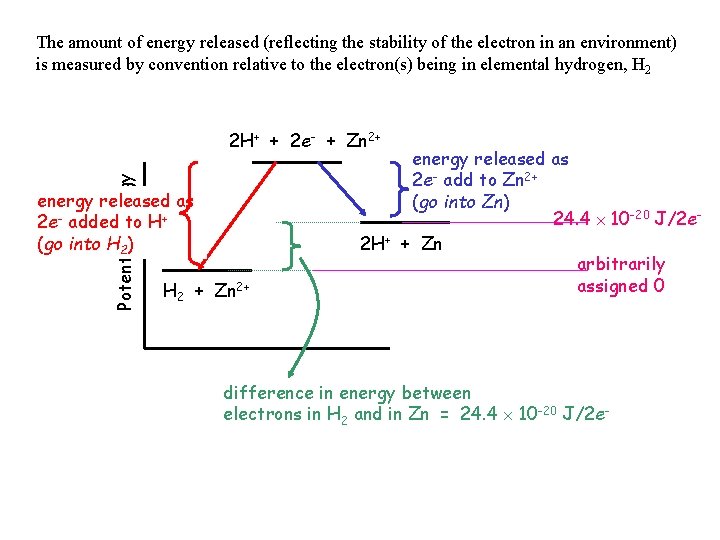
The amount of energy released (reflecting the stability of the electron in an environment) is measured by convention relative to the electron(s) being in elemental hydrogen, H 2 Potential Energy 2 H+ + 2 e- + Zn 2+ energy released as 2 e- added to H+ (go into H 2) H 2 + Zn 2+ energy released as 2 e- add to Zn 2+ (go into Zn) 24. 4 10 -20 J/2 e 2 H+ + Zn arbitrarily assigned 0 difference in energy between electrons in H 2 and in Zn = 24. 4 10 -20 J/2 e-
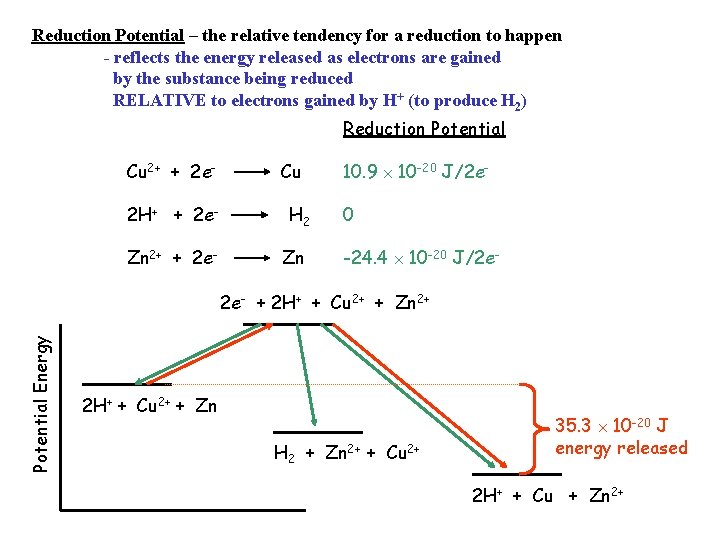
Reduction Potential – the relative tendency for a reduction to happen - reflects the energy released as electrons are gained by the substance being reduced RELATIVE to electrons gained by H+ (to produce H 2) Reduction Potential Cu 2+ + 2 e 2 H+ + 2 e. Zn 2+ + 2 e- Cu H 2 Zn 10. 9 10 -20 J/2 e 0 -24. 4 10 -20 J/2 e- Potential Energy 2 e- + 2 H+ + Cu 2+ + Zn 2+ 2 H+ + Cu 2+ + Zn H 2 + Zn 2+ + Cu 2+ 35. 3 10 -20 J energy released 2 H+ + Cu + Zn 2+

Standard Units – the energy available from a battery is measured in Volts 1 Volt (V) = 1 Joule (J) 1 Coulomb of charge (C) 96, 485 Coulomb: mole e- 1 Coulomb 1. 0364 10 -5 mole e- 6. 241 1018 e- Reduction Potential Cu 2+ + 2 e 2 H+ + 2 e. Zn 2+ + 2 e- Cu H 2 Zn 10. 9 10 -20 J/2 e- = x 6. 241 1018 e- = 0. 34 J 1 C 0 -24. 4 10 -20 J/2 e- = x 6. 241 1018 e- = -0. 76 J NOTE: the potential energy will depend on the environment standard reduction potentials are for : 25 C, 1 atm, and solutes at 1 M http: //www. google. com/ 1 C
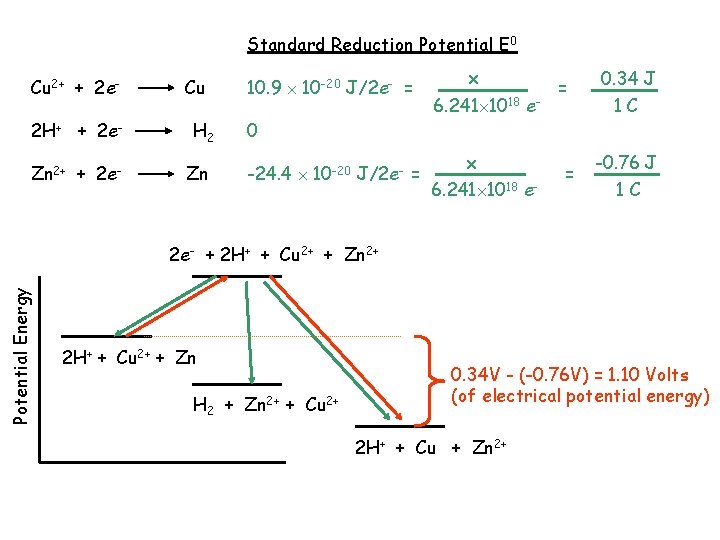
Standard Reduction Potential E 0 Cu 2+ + 2 e 2 H+ + 2 e. Zn 2+ + 2 e- Cu H 2 Zn 10. 9 10 -20 J/2 e- = x 6. 241 1018 e- = 0 -24. 4 10 -20 J/2 e- = x 6. 241 1018 e- = 0. 34 J 1 C -0. 76 J 1 C Potential Energy 2 e- + 2 H+ + Cu 2+ + Zn 2+ 2 H+ + Cu 2+ + Zn H 2 + Zn 2+ + Cu 2+ 0. 34 V - (-0. 76 V) = 1. 10 Volts (of electrical potential energy) 2 H+ + Cu + Zn 2+

Battery Voltage Zn(s) + Cu 2+(aq) Cu(s) + Zn 2+(aq) Zn(s) Zn 2+(aq) + 2 e. Cu Battery Voltage = (E 0 cell) = (net ionic equation) oxidation reduction potential for the oxidation 0. 34 V - (-0. 76 V) Determine Voltage for standard lead storage car battery Pb(s) + Pb. O 2(s) + H 2 SO 4(aq) hydrogen fuel cell H 2 (g) + O 2(g) H 2 O(l) Pb. SO 4(s) + H 2 O(l)
The Waterfall The Dam The Lake The Turbine The River
The Waterfall The Dam OPEN THE DAM!! The Lake The Turbine The River
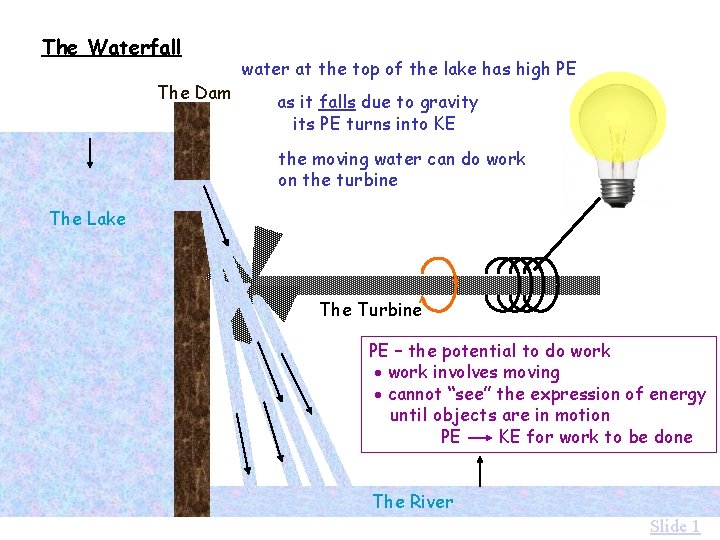
The Waterfall The Dam water at the top of the lake has high PE as it falls due to gravity its PE turns into KE the moving water can do work on the turbine The Lake The Turbine PE – the potential to do work involves moving cannot “see” the expression of energy until objects are in motion PE KE for work to be done The River Slide 1
- Slides: 15