vod do tmovho projektu Peter Ballo Katedra fyziky

Úvod do tímového projektu Peter Ballo Katedra fyziky Fakulta elektrotechniky a informatiky
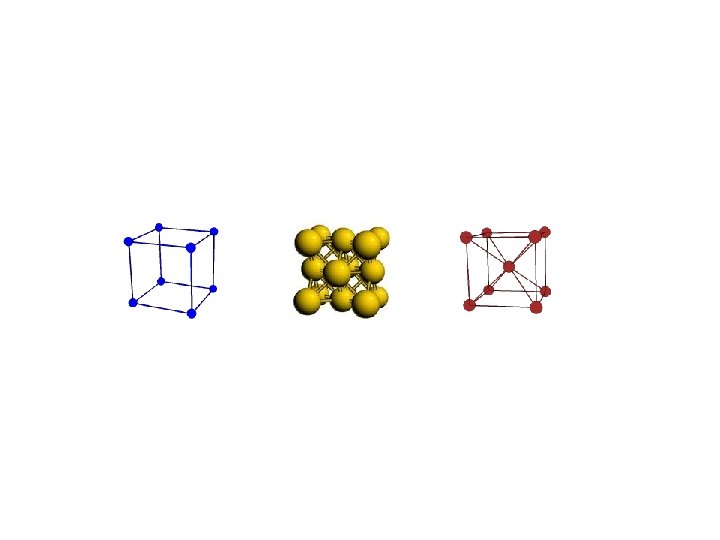

Interatomic Potentials • • Before we can start a simulation, we need the model! Interactions between atoms, molecules, … are determined by quantum mechanics: – Schrödinger Equation + Born-Oppenheimer (BO) approximation – BO: Because electrons T is so much higher (1 e. V=10, 000 K) than true T and they move so fast, we can get rid of electrons and consider interaction of nuclei in an effective potential “surface. ” V(R). – Approach does not work during chemical reactions. Crucial since V(R) determines the quality of result. But we don’t know V(R). – Semi-empirical approach: make a good guess and use experimental data to fix it up – Quantum chemistry approach: works in a real space. – Ab initio approach: it works really excellent but…

Semi-empirical potentials • Assume a functional form, e. g. 2 -body form. • Find some data: theory + experiment • Use theory + simulation to fit form to data. • What data? – Atom-atom scattering in gas phase – Virial coefficients, transport in gas phase – Low-T properties of the solid, cohesive energy, lattice constant, bulk modulus. – Melting temperature, critical point, triple point, surface tension, …. • Interpolation versus extrapolation. • Are results predictive?

Some tests -Lattice constant -Bulk modulus -Cohesive energy -Vacancy formation energy -Property of an impurity
![Lennard-Jones potential V(R) = i<jv(ri-rj) v(r) = 4 [( /r)12 - ( /r)6] = Lennard-Jones potential V(R) = i<jv(ri-rj) v(r) = 4 [( /r)12 - ( /r)6] =](http://slidetodoc.com/presentation_image_h2/625b06ceffb359c3dae5f4c54743e79b/image-8.jpg)
Lennard-Jones potential V(R) = i<jv(ri-rj) v(r) = 4 [( /r)12 - ( /r)6] = minimum = wall of potential Reduced units: – Energy in – Lengths in Good model for rare gas atoms Phase diagram is universal! (for rare gas systems).

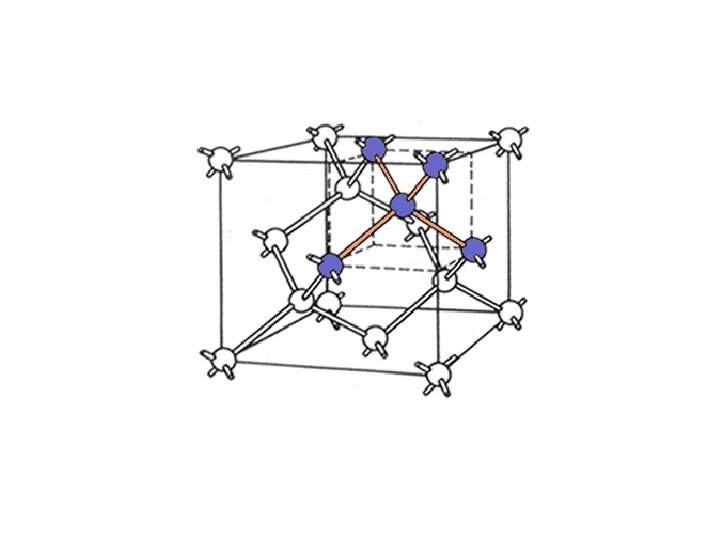
Silicon potential • • Solid silicon is NOT well described by a pair potential. Tetrahedral bonding structure caused by the partially filled p-shell: sp 3 hybrids (s+px+py+pz , s-px+py+pz , s+px-py+pz , s+px+py-pz) Stiff, short-ranged potential caused by localized electrons. Stillinger-Weber (1985) potential fit to: Lattice constant, cohesive energy, melting point, structure of liquid Si for r<a • rk Minimum at 109 o ri i rj

Metallic potentials • • Have a inner core + valence electrons Valence electrons are delocalized. Pair potentials do not work very well. Strength of bonds decreases as density increases because of Pauli principle. EXAMPLE: at a surface, LJ potential predicts expansion but metals contract Embedded Atom Method (EAM) or glue models better. Daw and Baskes, PRB 29, 6443 (1984). Embedding function electron density pair potential • Good for spherically, closed-packed, symmetric atoms: FCC Cu, Al, Pb • Not so good for BCC.
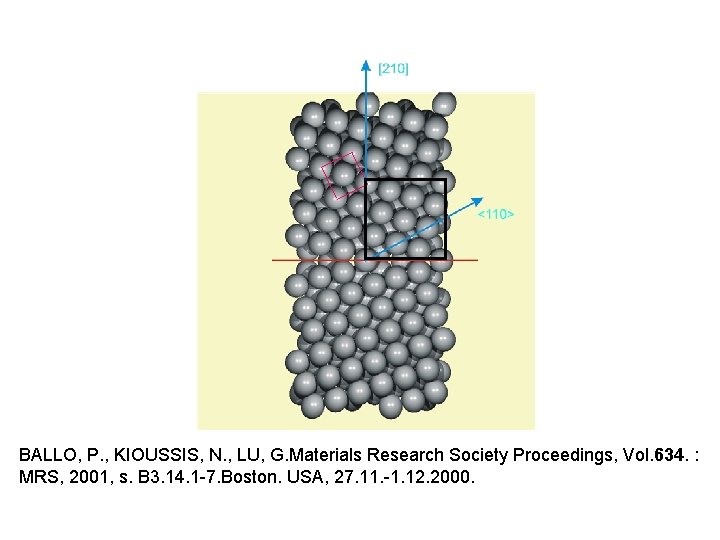
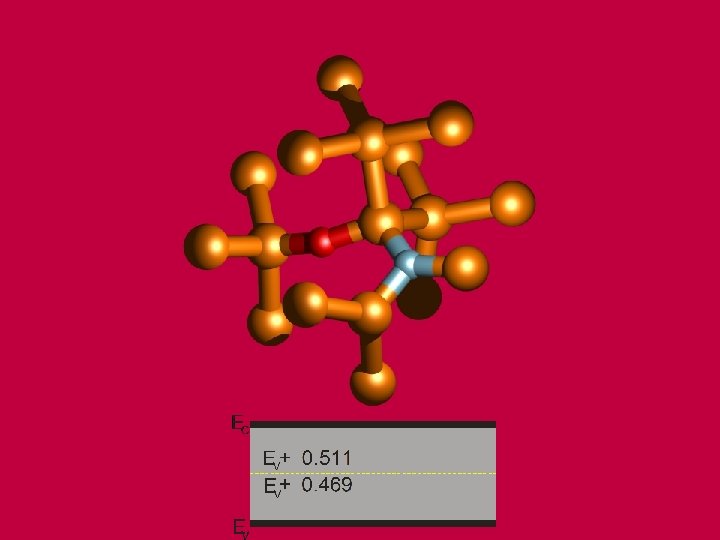
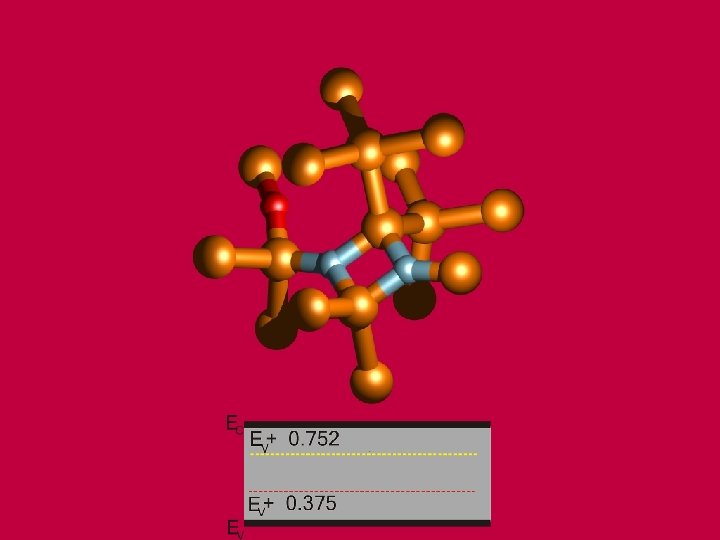
BALLO, P. , KIOUSSIS, N. , LU, G. Materials Research Society Proceedings, Vol. 634. : MRS, 2001, s. B 3. 14. 1 -7. Boston. USA, 27. 11. -1. 12. 2000.
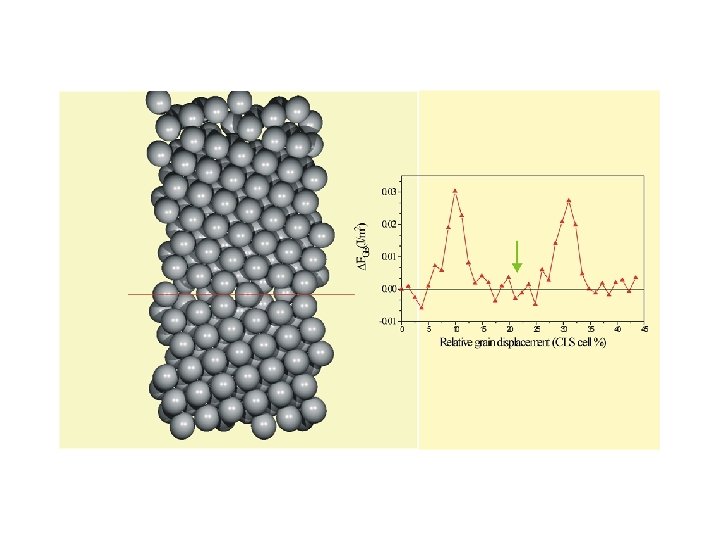
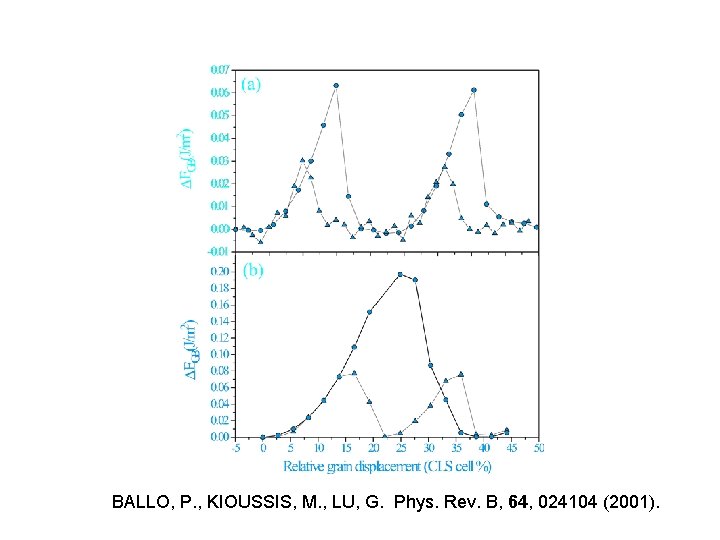
BALLO, P. , KIOUSSIS, M. , LU, G. Phys. Rev. B, 64, 024104 (2001).
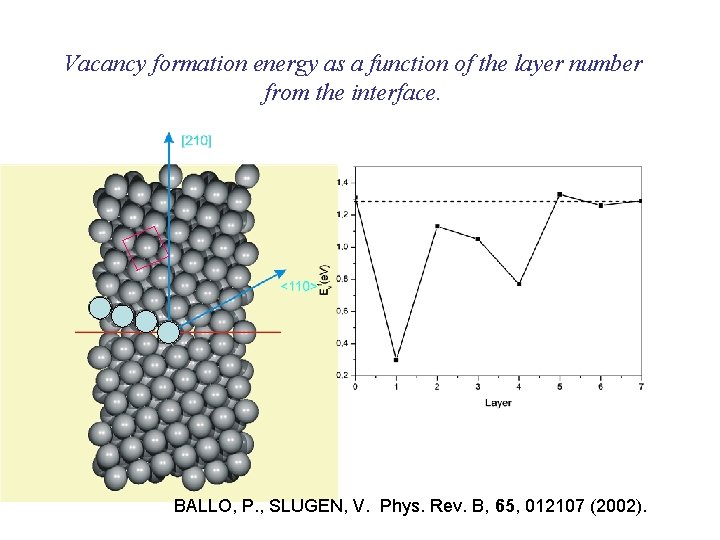
Vacancy formation energy as a function of the layer number from the interface. BALLO, P. , SLUGEN, V. Phys. Rev. B, 65, 012107 (2002).
BALLO, P. , HARMATHA, L. Phys. Rev. B, 68, 153201 (2003).
- Slides: 17