Vitek 2 Antimicrobial Susceptibility Testing on All Urine

Vitek 2 Antimicrobial Susceptibility Testing on All Urine Isolates - WASPLab and Removal of UF 1000 Automated Urine Microscopy John Mallon Technical Services Manager NHS GG&C Microbiology

INTRODUCTION • GG&C Microbiology consolidated onto 2 sites on 15 th January 2018 • WASPLab technology introduced in 2018 – delivered in May (GRI) and Sept (QEUH) • GG&C Microbiology receive 270, 000 urines per annum – 120, 000 GRI & 150, 000 QEUH • Funded for a 6 month pilot study in October 2018 to test ALL urine isolates on Vitek 2 • Pilot was to examine service improvements by testing all isolates with Vitek 2

PREVIOUS METHODOLOGY • All urines screened by UF 1000 (Day 0) • Microscopy positive specimens cultured onto CUTI • Disc Testing done on positive isolates (Day 1) • Vitek 2 testing done on some clinical categories (Day 1) and on resistant disc testing isolates (Day 2) • Full ESBL confirmation tests done on suspect organisms (Day 2) if screen positive on disc testing • Resistant organisms requiring Vitek 2 taking at least 3 days for a report to go out • Organism identification only done on isolates sent to the Vitek 2

PREVIOUS METHODOLOGY • Advantages: Ø Good turnaround times for negative samples Ø Ability to quickly report sensitive isolates to the standard urine antibiotics Ø Inexpensive Ø Targeting of additional expense on predominantly more resistant isolates

PREVIOUS METHODOLOGY • Disadvantages: Ø Poor turnaround times for positive samples with more complex resistance patterns Ø Isolates with disc testing only present a choice of a maximum of 5 antibiotics to the Consultants Ø Identification not performed on all isolates Ø Vitek “expert rules” not applied to detect intrinsic and inferred resistance patterns for ALL isolates Ø Inequality of service provision on urine reports to clinicians

URINE VITEK 2 PILOT • Vitek 2 Susceptibility to be performed on ALL urinary isolates • Full ESBL disc testing and Aztreonam / Mecillinam disc testing to be done as well as Vitek 2 • All organisms to be identified to species level on Vitek MS • Introduction of full electronic reporting and removal of paper worksheets

URINE VITEK 2 PILOT - AIMS • Improved Antibiotic Stewardship – reduction of reporting of “non cdiffogenic” antibiotics (Cipro and Amoxy-clav) • Improved turnaround times – 13% of GP and 20% of inpatient have discs and Vitek 2 – 1 day delay • Quicker TAT will assist the inpatient areas with patient flow and potentially release beds sooner • Antimicrobial resistance monitoring – accurate resistance rates not fully known • Spending in laboratory will reduce prescribing costs • Examine accuracy of Vitek ESBL testing vs Mast Discs
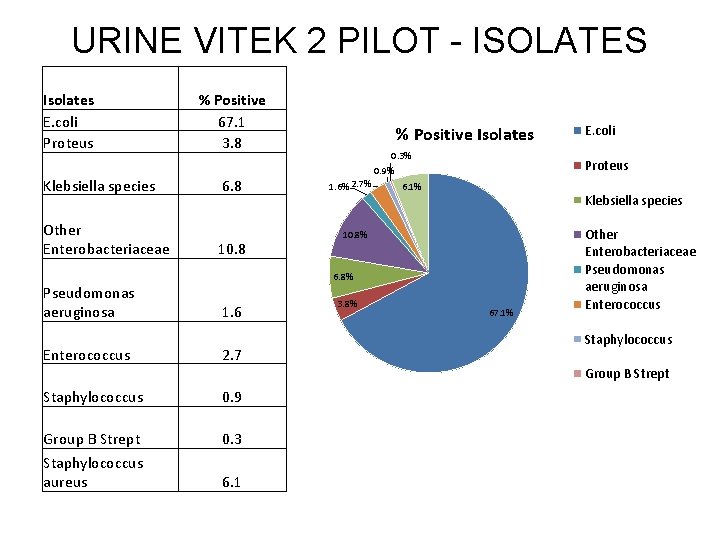
URINE VITEK 2 PILOT - ISOLATES Isolates E. coli Proteus Klebsiella species Other Enterobacteriaceae % Positive 67. 1 3. 8 6. 8 10. 8 % Positive Isolates 0. 3% 0. 9% 1. 6% 2. 7% Proteus 6. 1% Klebsiella species 10. 8% 6. 8% Pseudomonas aeruginosa Enterococcus 1. 6 2. 7 3. 8% E. coli 67. 1% Other Enterobacteriaceae Pseudomonas aeruginosa Enterococcus Staphylococcus Group B Strept Staphylococcus 0. 9 Group B Strept Staphylococcus aureus 0. 3 6. 1
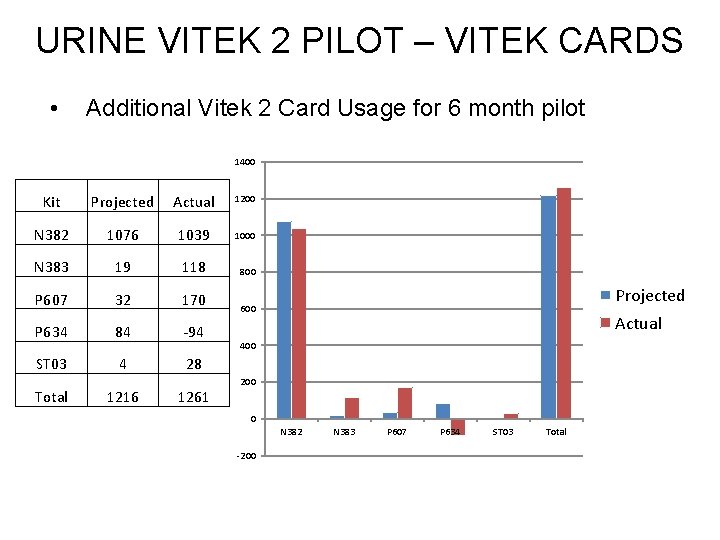
URINE VITEK 2 PILOT – VITEK CARDS • Additional Vitek 2 Card Usage for 6 month pilot 1400 Kit Projected Actual 1200 N 382 1076 1039 1000 N 383 19 118 800 P 607 32 170 P 634 84 -94 ST 03 4 28 Total 1216 1261 Projected 600 Actual 400 200 0 N 382 -200 N 383 P 607 P 634 ST 03 Total
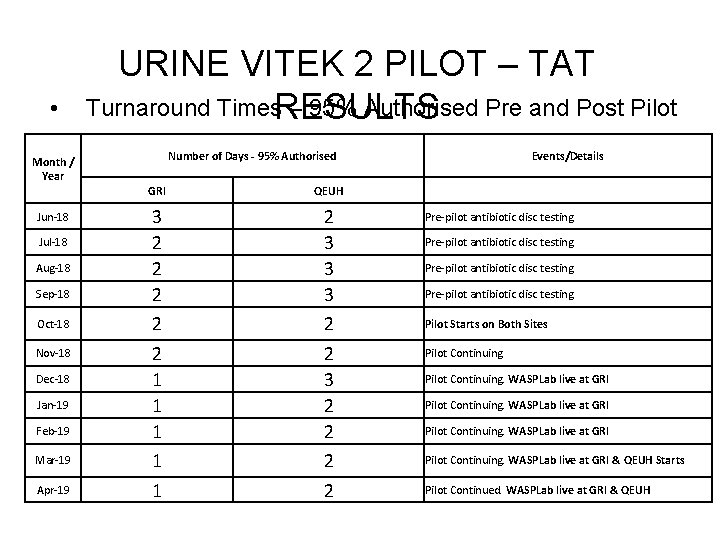
• Month / Year Jun-18 Jul-18 Aug-18 Sep-18 Oct-18 Nov-18 Dec-18 Jan-19 Feb-19 Mar-19 Apr-19 URINE VITEK 2 PILOT – TAT Turnaround Times. RESULTS – 95% Authorised Pre and Post Pilot Number of Days - 95% Authorised GRI QEUH 3 2 2 2 1 1 1 2 3 3 3 2 2 2 2 Events/Details Pre-pilot antibiotic disc testing Pilot Starts on Both Sites Pilot Continuing. WASPLab live at GRI & QEUH Starts Pilot Continued. WASPLab live at GRI & QEUH
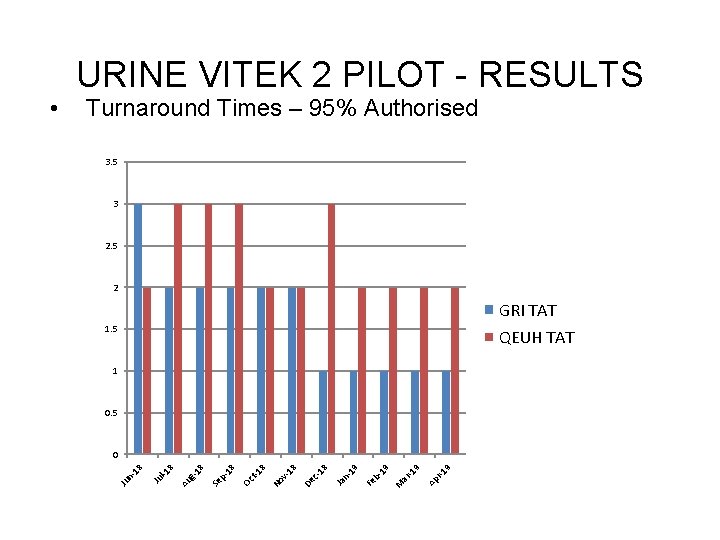
Turnaround Times – 95% Authorised 3. 5 3 2. 5 2 GRI TAT 1. 5 QEUH TAT 1 0. 5 9 r-1 Ap -1 9 ar M 19 b. Fe 19 n. Ja 18 c. De v 18 No 8 t-1 Oc 18 p. Se 18 Au g- 8 l-1 Ju n 18 0 Ju • URINE VITEK 2 PILOT - RESULTS
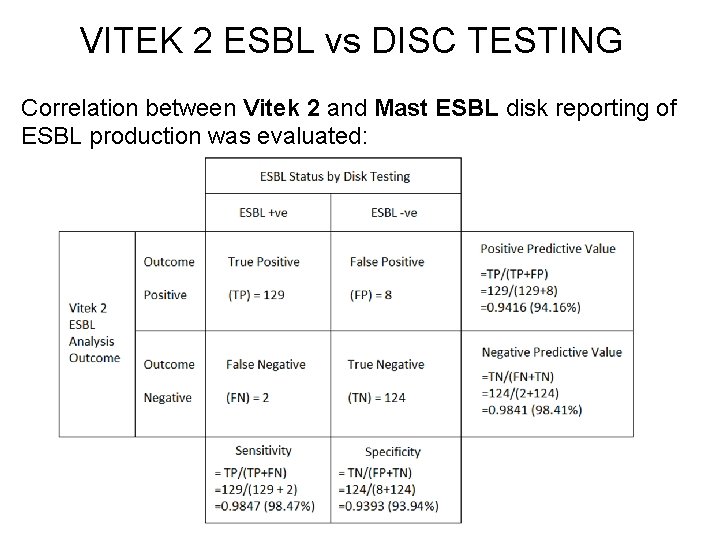
VITEK 2 ESBL vs DISC TESTING Correlation between Vitek 2 and Mast ESBL disk reporting of ESBL production was evaluated:
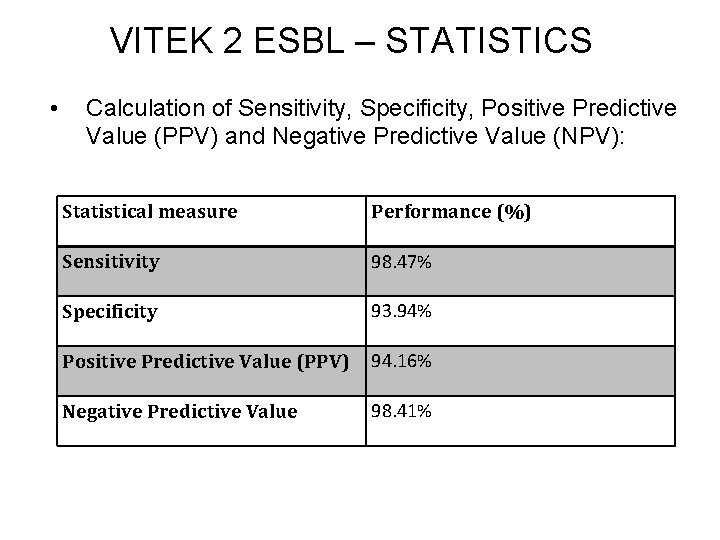
VITEK 2 ESBL – STATISTICS • Calculation of Sensitivity, Specificity, Positive Predictive Value (PPV) and Negative Predictive Value (NPV): Statistical measure Performance (%) Sensitivity 98. 47% Specificity 93. 94% Positive Predictive Value (PPV) 94. 16% Negative Predictive Value 98. 41%

VITEK 2 ESBL STATISTICAL ANALYSIS • Focus on the common ESBLs at the urine bench • For urinary enterobacteriacaea isolates, the detection of ESBLs by the Vitek 2 appears Sensitive and Specific • Vitek susceptibility pattern is effective at eliminating False Negative isolates • False-negatives may be due to poor antibiotic responses within the test or Amp. C-masking of ESBLs • The Vitek overcalls ESBL producers for complex resistance patterns. • The clinical outcomes are unlikely to be severely affected by these false positive results

VITEK 2 ESBL – CLINICAL ANALYSIS • Evaluation of 263 urinary Enterobacteriaceae isolates found discrepant reporting of ESBL production in 3. 8% of cases • Due to interpretation of the full antibiogram this is unlikely to have affected patient management. • For non-sterile sites, it is reasonable to report ESBL production as per Vitek 2 results • In severe cases, and in sterile sites, ESBL disk testing will continue as a failsafe in the serious clinical cases

URINE VITEK 2 PILOT CONCLUSIONS • Improved Turnaround Times • Better antibiotic stewardship with more antibiotics reported clinically e. g. Amoxycillin and more resistant organisms released • ESBL disc testing not routinely required – Vitek 2 Fine • Aztreonam disc testing no longer required • Significant additional cost to fund extension of pilot • More work for BMS staff as setting up disc testing and Vitek 2 more work than previous methodology

Urine Microscopy – UF 1000 GG&C Microbiology performed urine microscopy on ALL specimens on UF 1000 instruments • 270, 000 requests per year with 4 instruments at QEUH (150, 000) and 3 instruments at GRI (120, 000) • Urine microscopy used as a screening method and negative microscopy reported out as final • Approximately 30% of urine samples were microscopy negative and authorised on the same day (Day 0) • Urine microscopy for Red Blood Cells was “switched off” due to the inaccurate results obtained • Specimens requiring mandatory culture manually identified and culture set up manually

SIGN Guidelines - Adults SIGN guidelines recommend urine specimen are Cultured Guidelines indicated that Microscopy alone lacks Sensitivity Microscopy indicated in: Ø Patients who are post renal transplant Ø Patients with Glomerulonephritis-

NICE Guidelines - Paediatrics Urine microscopy in children is indicated in the following: Ø Children under 3 years of age – Easy for the lab to triage Ø Children aged 3 or over - on dipstick testing if leucocyte esterase is positive and nitrite is negative, then a urine specimen should be sent for microscopy and culture - requires the clinical users to contact the lab

Culturing All Urines § Blind Culture has been performed before as a result of UF 1000 unavailability § Culturing ALL urine specimens is a similar amount of work for CSW staff § Culturing all urine specimens creates additional work for the BMS staff as there are 30% more CUTI plates to be read the following day § The majority of the additional plates set up with blind culture are culture negative

WASPLab • Moving to Blind Culture is less work for CSW staff as UF 1000 is not used and WASPLab loaded directly • WASPLab able to blind culture all specimens with throughput and capacity not an issue • Ability to use WASPLab for rapid screening of negative results no significant increase in BMS work for the additional CUTI plates • Additional plates are contained in WASPlab negative plates automatically discarded with

Service Benefits: Urines Vitek 2 • Improving urine culture turnaround times especially for complex resistant isolates • Greater range of antibiotics reported and less reporting of Cdiffogenic antibiotics in over 65’s • Able to report less broad spectrum antibiotics in sensitive isolates • Better epidemiology and information on local resistance patterns and trends • Equity of service for the patient and requestors.

Service Benefits Urine Microscopy Change • All specimens cultured and compliant with SIGN and NICE guidelines • Manual Microscopy performed on approximately 3% of specimens • Costs involved in overall service improvement identified from existing budget • Use of WASPLab for blind culture minimises additional work for BMS and CSW staff.

CONCLUSION Ø GG&C Microbiology to perform urine susceptibility testing on Vitek 2 on ALL urine isolates Ø Urine microscopy service reviewed to ensure microscopy offered as appropriate – evidence based practise Ø WASPLab technology employed to assist the laboratory to make the change Ø Financial impact contained within existing budget Ø Improved Antibiotic stewardship and urine specimen TAT to support better and faster patient care

QUESTIONS
- Slides: 25