Vitamins Carbohydrates Andy Howard Introductory Biochemistry Fall 2010

Vitamins; Carbohydrates Andy Howard Introductory Biochemistry, Fall 2010 29 September 2010 Biochem: Vitamins&Carbohydrates 1 09/29/2010

Now we’ll study vitamins and sugars! n n Vitamins are diet-derived micronutrients, many of them used as sources of coenzymes Carbohydrates are vital as energy sources, and they also serve as building blocks for lipidcarbohydrate and proteincarbohydrate complexes 09/29/2010 Biochem: Vitamins&Carbohydrates 2
What we’ll discuss n Vitamins n n n Definitions Coenzyme Precursors Ascorbate Fat-soluble vitamins n Sugar Concepts n n Monosaccharides Chirality Cyclization Sugars, cont’d n Sugar derivatives n Oligosaccharides n Glycosides Polysaccharides n Starch & glycogen n Cellulose and chitin 09/29/2010 Biochem: Vitamins&Carbohydrates 3
i. Clicker quiz, question 1 n Which coenzyme would you expect would be required for the reaction oxaloacetate + glutamate aspartate + -ketoglutarate? (a) ascorbate (b) PLP (c) thiamine pyrophosphate (d) NAD (e) none of the above 09/29/2010 Biochem: Vitamins&Carbohydrates 4

Vitamins: necessary micronutrients that cannot be synthesized internally n n n What’s a vitamin for one organism is not for another Primates and some rodents are the only vertebrates that don’t synthesize ascorbate E. coli can make almost everything given energy and sources of atoms 09/29/2010 Biochem: Vitamins&Carbohydrates 5

Why wouldn’t organisms make everything? n n n Complex metabolites require energy for synthesis Control of their synthesis is also metabolically expensive Cheaper in the long run to derive these nutrients from diet 09/29/2010 Biochem: Vitamins&Carbohydrates 6
Vitamins: broad classifications n Water-soluble vitamins n n n Coenzymes or coenzyme precursors Non-coenzymic metabolites Fat-soluble vitamins n n Antioxidants Other lipidic vitamins 09/29/2010 Biochem: Vitamins&Carbohydrates 7

Are all nutrients that we can’t synthesize considered vitamins? n n n No: If it’s required in large quantities, it’s not a vitamin By convention, essential fatty acids like arachidonate aren’t considered vitamins 09/29/2010 Biochem: Vitamins&Carbohydrates 8

Warning: ugly photos coming n n I have included some web-derived photos of patients with severe vitamin deficiencies If you’re squeamish, be prepared. 09/29/2010 Biochem: Vitamins&Carbohydrates 9
Coenzyme precursors n n We’ve already outlined the fact that most water-soluble coenzymes are derived from vitamins—typically B vitamins Typically the dietary form can be converted by a fairly short metabolic pathway into the coenzyme form, e. g. n niacin + glutamine nicotinamide + glutamate nicotinamide + ADP-ribose NAD Some coenzyme precursors are, in fact, lipidic 09/29/2010 Biochem: Vitamins&Carbohydrates 10

The B vitamins n n n n n All aqueous micronutrients Generally identified via pathologies associated with dietary deficiencies B 1: thiamin (produces TPP) B 2: riboflavin (produces FAD, FMN) B 3: niacin (produces NAD, NADP) B 5: pantothenate (produces Coenzyme A) B 6: pyridoxamine (produces PLP) B 9: folate: produces THF, THF derivatives B 12: cobalamin (produces adenosylcobalamin, methylcobalamin) 09/29/2010 Biochem: Vitamins&Carbohydrates 11
Deficiency of niacin n Pellagra: dermatitis, diarrhea, dementia Still found in some diets that are low in vitamin content Humans can actually synthesize nicotinamide from tryptophan; but that’s often in short supply too 09/29/2010 Biochem: Vitamins&Carbohydrates Image courtesy history. nih. gov 12
Deficiency of thiamine n n Beriberi: primary symptoms are in nervous system and musculature Polished rice is missing thiamine; rice hulls are rich in it 09/29/2010 Biochem: Vitamins&Carbohydrates Image courtesy answers. com 13

Deficiency of cobalamin n n Available sources of cobalamin are animal products Therefore vegans need to be careful to get cobalamin from supplements Symptoms of deficiency (pernicious anemia): weakness, fatigue, pallor, palpitations, dizziness Deficiency is common even in non-vegans: 5 -40% of the population? 09/29/2010 Biochem: Vitamins&Carbohydrates 14

How do herbivores get B 12? n n n Strict herbivores like cattle do require cobalamin: how do they get it? Answer: gut bacteria in the cattle produce enough for the cattle to use This illustrates the fact that even large animals don’t need much of this 09/29/2010 Biochem: Vitamins&Carbohydrates 15
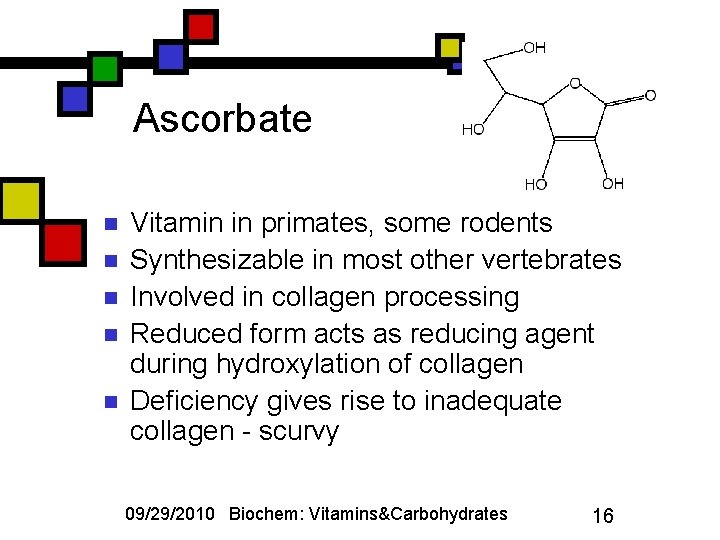
Ascorbate n n n Vitamin in primates, some rodents Synthesizable in most other vertebrates Involved in collagen processing Reduced form acts as reducing agent during hydroxylation of collagen Deficiency gives rise to inadequate collagen - scurvy 09/29/2010 Biochem: Vitamins&Carbohydrates 16
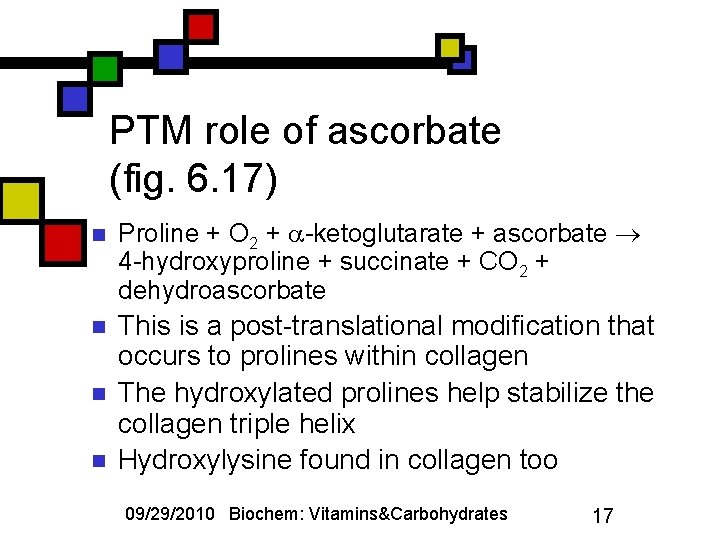
PTM role of ascorbate (fig. 6. 17) n Proline + O 2 + -ketoglutarate + ascorbate 4 -hydroxyproline + succinate + CO 2 + dehydroascorbate n This is a post-translational modification that occurs to prolines within collagen The hydroxylated prolines help stabilize the collagen triple helix Hydroxylysine found in collagen too n n 09/29/2010 Biochem: Vitamins&Carbohydrates 17

Dietary deficiency of ascorbate n n Primary sources of ascorbate are fruits, particularly citrus, and green vegetables Ascorbate deficiency’s first symptom involves collagen degradation, leading to scurvy Image courtesy U. Cincinnati Medical School 09/29/2010 Biochem: Vitamins&Carbohydrates 18

Scurvy in history n n Shortage of green vegetables in sailors’ diets meant scurvy was rampant on shipboard until the 18 th century Success of English navy over French 1760 -1800 was partly due to the introduction of limes in English sailors’ diets 50 years before the French caught on 09/29/2010 Biochem: Vitamins&Carbohydrates 19
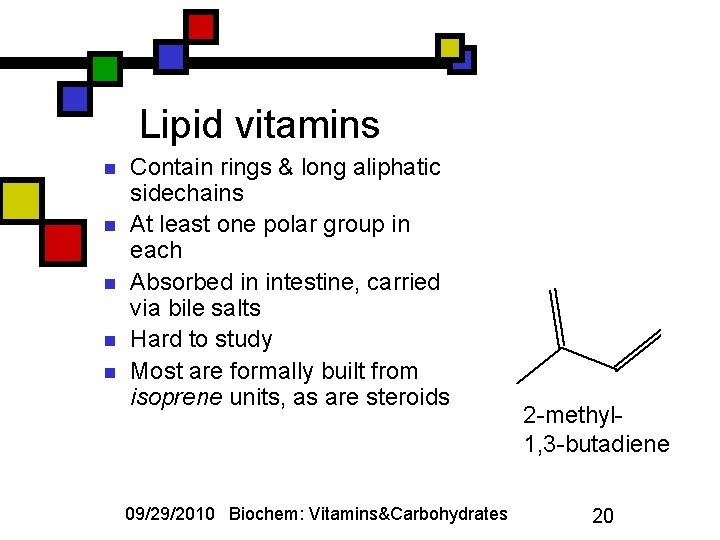
Lipid vitamins n n n Contain rings & long aliphatic sidechains At least one polar group in each Absorbed in intestine, carried via bile salts Hard to study Most are formally built from isoprene units, as are steroids 09/29/2010 Biochem: Vitamins&Carbohydrates 2 -methyl 1, 3 -butadiene 20

Vitamin A (retinol) n n n 3 forms varying in terminal polar group; based on four isoprene units Involved in signaling and receptors -carotene is nonpolar dimer 09/29/2010 Biochem: Vitamins&Carbohydrates 21

Vitamin A deficiency n n Produces night blindness because the retina and cornea dry out Most common cause: nursing infants whose mothers have vitamin A deficiency in their diet Corneal scarring due to vitamin A deficiency: www. cehjournal. org/ 09/29/2010 Biochem: Vitamins&Carbohydrates 22

Vitamin D n n Several related forms Cholesterol-derived C 30 (6 isoprenes) Hormones involved in Ca 2+ regulation Cancer chemoprevention! Figure courtesy Cyberlipid 09/29/2010 Biochem: Vitamins&Carbohydrates (cholecalciferol) 23
Vitamin D deficiency n n n Rickets in children: Bone disease, restlessness, slow growth One form of vitamin D is actually synthesizable from cholesterol given adequate sunlight; Therefore rickets is most common in densely settled urban environments www. thachers. org/rickets_photos. htm 09/29/2010 Biochem: Vitamins&Carbohydrates 24

Vitamin E ( -tocopherol) n n n phenol C 30 (6 isoprenes) nonsteroidal vitamin Phenol can undergo 1 eoxidation to moderately stable free radical Antioxidant activity prevents damage to fatty acids in membranes Fig. Courtesy UIC pharmacy program 09/29/2010 Biochem: Vitamins&Carbohydrates 25
Vitamin K (phylloquinone) n n n C 30 (6 isoprenes) non-steroidal vitamin Involved in synthesis of proteins involved in blood coagulation Reduced form involved as reducing agent in carboxylation reaction on glu sidechains Figure courtesy Cyberlipid 09/29/2010 Biochem: Vitamins&Carbohydrates 26

Carbohydrates and lipids! n n (deep breath): Now we will launch into a chemical exploration of carbohydrates and lipids These are the two major categories of macronutrients besides proteins Significant as intermediates, fuel, structural elements, cofactors, … 09/29/2010 Biochem: Vitamins&Carbohydrates 27

Carbohydrates n n n n These are polyhydroxylated aldehydes and ketones, many of which can exist in cyclic forms General monomeric formula (CH 2 O)m, 3 < m < 9 With one exception (dihydroxyacetone), they contain chiral centers Highly soluble Can be oligomerized and polymerized Oligomers may or may not be soluble Most abundant organic molecules on the planet 09/29/2010 Biochem: Vitamins&Carbohydrates 28

How do we measure solubility for very soluble compounds? n n n (Note: this is not a serious chemical topic: it’s an example of how statistics can be abused…) The assertion is that, with highly soluble compounds like sugars, it’s difficult to use conventional approaches to compare their solubilities The suggestion is that we might use the amount of time it takes to dissolve (for example) 50 g of solute in 100 m. L of cold water: if it’s fast, the solute is more soluble than if it’s slow. 09/29/2010 Biochem: Vitamins&Carbohydrates 29
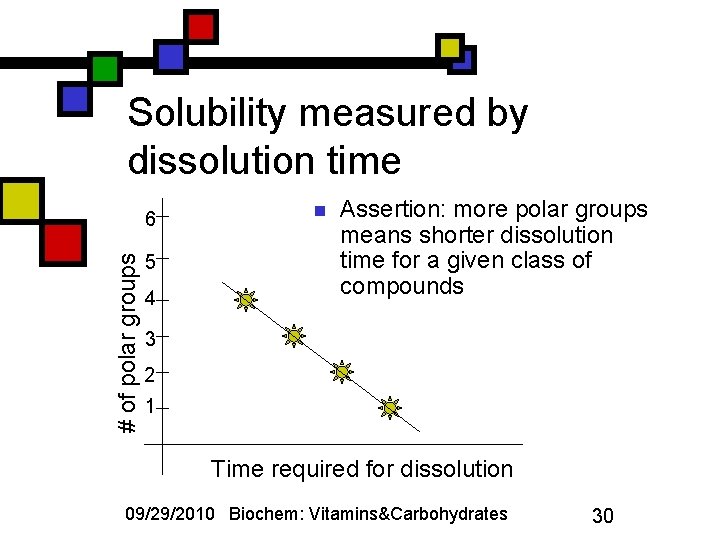
Solubility measured by dissolution time # of polar groups 6 5 4 n Assertion: more polar groups means shorter dissolution time for a given class of compounds 3 2 1 Time required for dissolution 09/29/2010 Biochem: Vitamins&Carbohydrates 30
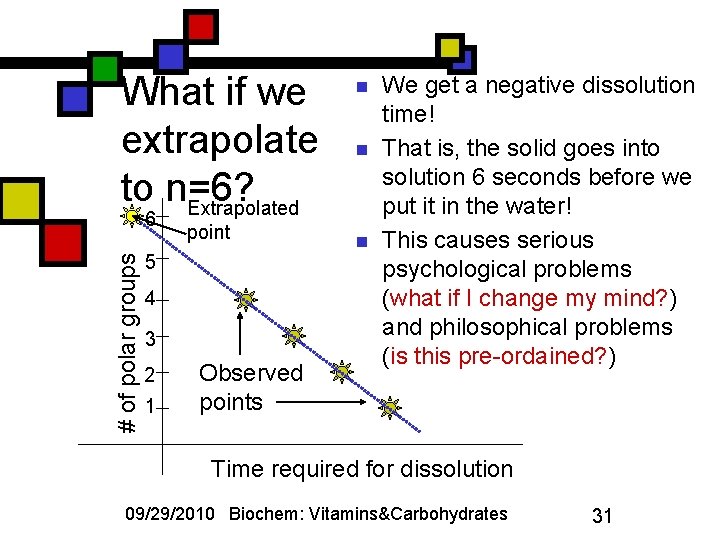
What if we extrapolate to n=6? Extrapolated # of polar groups 6 point 5 4 3 2 1 Observed points n n n We get a negative dissolution time! That is, the solid goes into solution 6 seconds before we put it in the water! This causes serious psychological problems (what if I change my mind? ) and philosophical problems (is this pre-ordained? ) Time required for dissolution 09/29/2010 Biochem: Vitamins&Carbohydrates 31

Whose idea is this? n n n Isaac Asimov, that’s who! “The endochronic properties of resublimated thiotimolene”: Astounding Science Fiction, March 1948 My point: extrapolations and other misuses of statistics are dangerous Benjamin Disraeli (popularized by Mark Twain): There are three kinds of untruth: lies, damn lies, and statistics. Okay: let’s get back to the science. 09/29/2010 Biochem: Vitamins&Carbohydrates 32
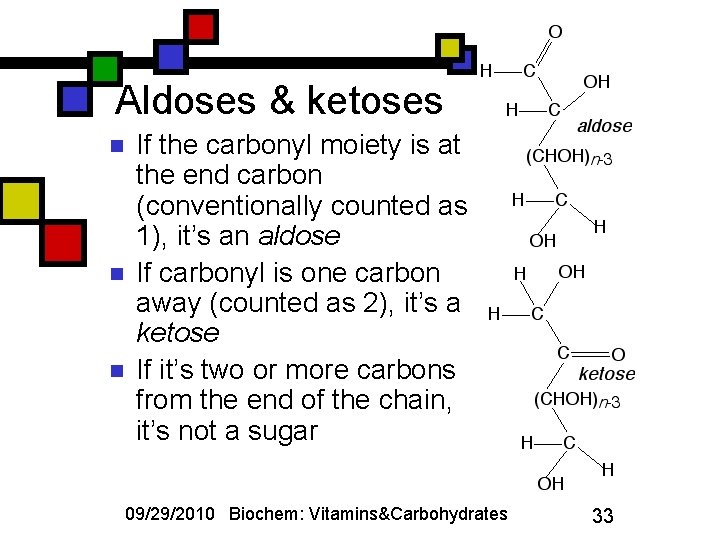
Aldoses & ketoses n n n If the carbonyl moiety is at the end carbon (conventionally counted as 1), it’s an aldose If carbonyl is one carbon away (counted as 2), it’s a ketose If it’s two or more carbons from the end of the chain, it’s not a sugar 09/29/2010 Biochem: Vitamins&Carbohydrates 33
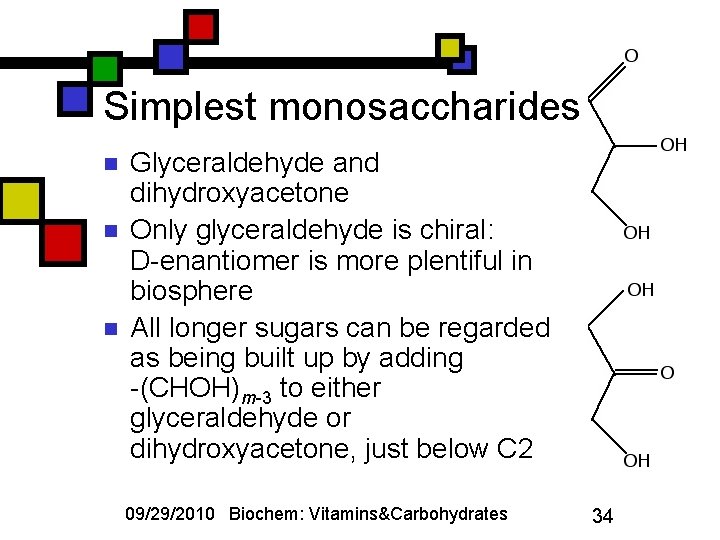
Simplest monosaccharides n n n Glyceraldehyde and dihydroxyacetone Only glyceraldehyde is chiral: D-enantiomer is more plentiful in biosphere All longer sugars can be regarded as being built up by adding -(CHOH)m-3 to either glyceraldehyde or dihydroxyacetone, just below C 2 09/29/2010 Biochem: Vitamins&Carbohydrates 34
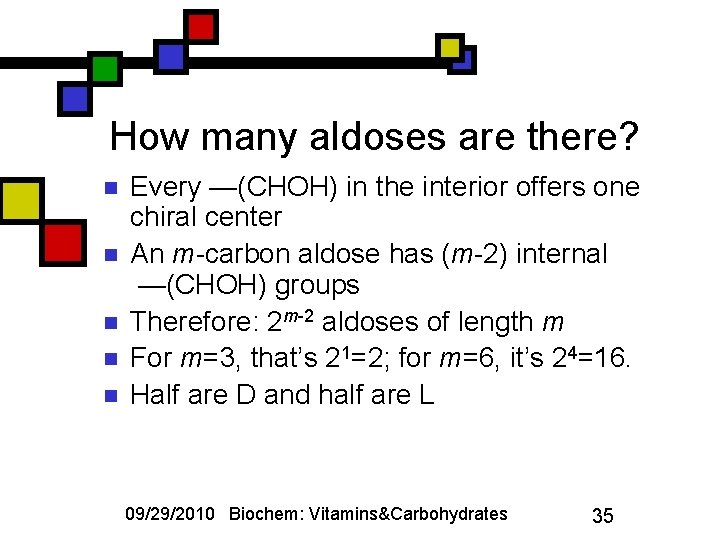
How many aldoses are there? n n n Every —(CHOH) in the interior offers one chiral center An m-carbon aldose has (m-2) internal —(CHOH) groups Therefore: 2 m-2 aldoses of length m For m=3, that’s 21=2; for m=6, it’s 24=16. Half are D and half are L 09/29/2010 Biochem: Vitamins&Carbohydrates 35
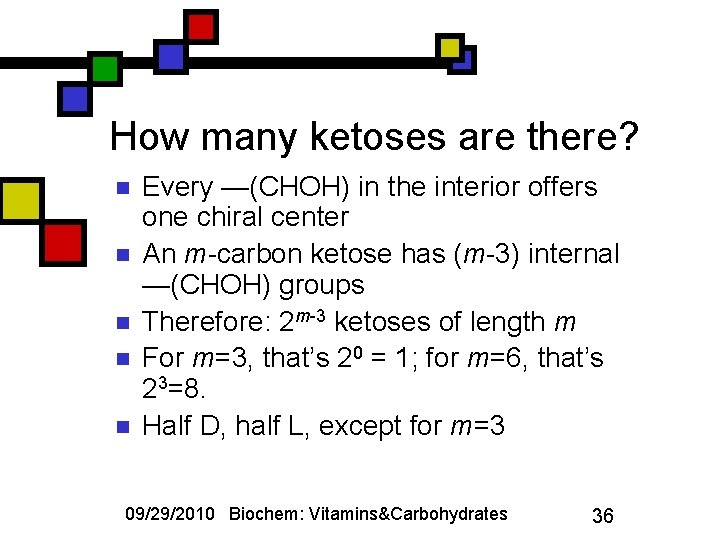
How many ketoses are there? n n n Every —(CHOH) in the interior offers one chiral center An m-carbon ketose has (m-3) internal —(CHOH) groups Therefore: 2 m-3 ketoses of length m For m=3, that’s 20 = 1; for m=6, that’s 23=8. Half D, half L, except for m=3 09/29/2010 Biochem: Vitamins&Carbohydrates 36

Review: stereochemical nomenclature n n n Stereoisomers: compounds with identical covalent bonding apart from chiral connectivity Enantiomers: compounds for which the opposite chirality applies at all chiral centers Epimers: compounds that differ in chirality at exactly one chiral center One chiral center: enantiomers are epimers. > 1 chiral center: enantiomers are not epimers. 09/29/2010 Biochem: Vitamins&Carbohydrates 37
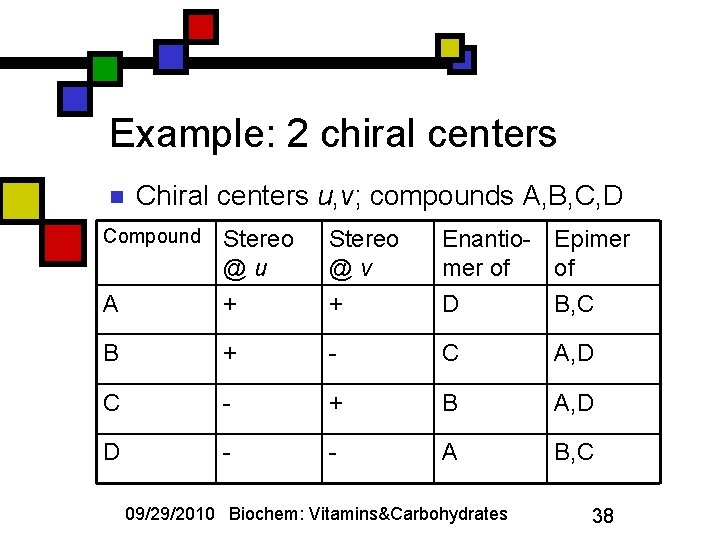
Example: 2 chiral centers n Chiral centers u, v; compounds A, B, C, D Compound Stereo @u Stereo @v Enantio- Epimer of of A + + D B, C B + - C A, D C - + B A, D D - - A B, C 09/29/2010 Biochem: Vitamins&Carbohydrates 38

Properties n n n Enantiomers have identical physical properties (MP, BP, solubility, surface tension…) except when they interact with other chiral molecules (Note!: water isn’t chiral!) Stereoisomers that aren’t enantiomers can have different properties; therefore, they’re given different names 09/29/2010 Biochem: Vitamins&Carbohydrates 39
Sugar nomenclature n n All sugars with m ≤ 7 have specific names apart from their enantiomeric (L or D) designation, e. g. D-glucose, L-ribose. The only 7 -carbon sugar that routinely gets involved in metabolism is sedoheptulose, so we won’t try to articulate the names of the others 09/29/2010 Biochem: Vitamins&Carbohydrates 40
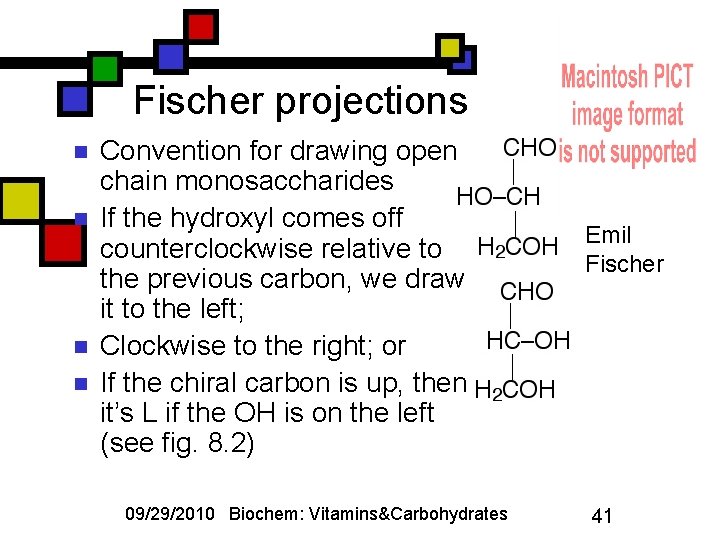
Fischer projections n n Convention for drawing openchain monosaccharides If the hydroxyl comes off counterclockwise relative to the previous carbon, we draw it to the left; Clockwise to the right; or If the chiral carbon is up, then it’s L if the OH is on the left (see fig. 8. 2) 09/29/2010 Biochem: Vitamins&Carbohydrates Emil Fischer 41

Cyclic sugars n n n Sugars with at least four carbons can readily interconvert between the openchain forms we have drawn and fivemembered(furanose) or six-membered (pyranose) ring forms in which the carbonyl oxygen becomes part of the ring Remember that 5 - and 6 -atom rings form with little or no strain; 4 - and 3 - are much harder There are no C=O bonds in the ring forms 09/29/2010 Biochem: Vitamins&Carbohydrates 42
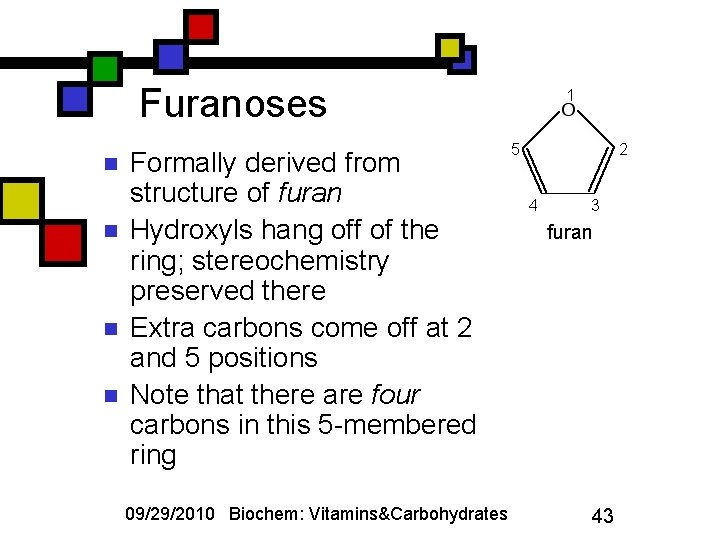
Furanoses n n Formally derived from structure of furan Hydroxyls hang off of the ring; stereochemistry preserved there Extra carbons come off at 2 and 5 positions Note that there are four carbons in this 5 -membered ring 09/29/2010 Biochem: Vitamins&Carbohydrates 1 5 2 4 3 furan 43
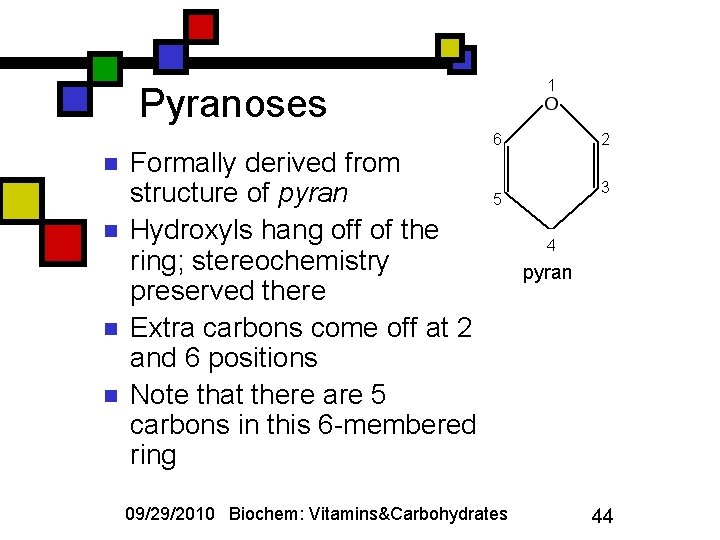
1 Pyranoses n n Formally derived from structure of pyran Hydroxyls hang off of the ring; stereochemistry preserved there Extra carbons come off at 2 and 6 positions Note that there are 5 carbons in this 6 -membered ring 6 2 3 5 09/29/2010 Biochem: Vitamins&Carbohydrates 4 pyran 44
How do we cyclize a sugar? n n n Formation of an internal hemiacetal or hemiketal (see a few slides from here) by conversion of the carbonyl oxygen to a ring oxygen Not a net oxidation or reduction; in fact it’s a true isomerization. The molecular formula for the cyclized form is the same as the open chain form 09/29/2010 Biochem: Vitamins&Carbohydrates 45
Family tree of aldoses n n Simplest: D-, L- glyceraldehyde (C 3) Add —CHOH: D, L-threose, erythrose (C 4) Add —CHOH: D, L- lyxose, xylose, arabinose, ribose (C 5) Add —CHOH: D, L-talose, galactose, idose, gulose, mannose, glucose, altrose, allose (C 6) 09/29/2010 Biochem: Vitamins&Carbohydrates 46
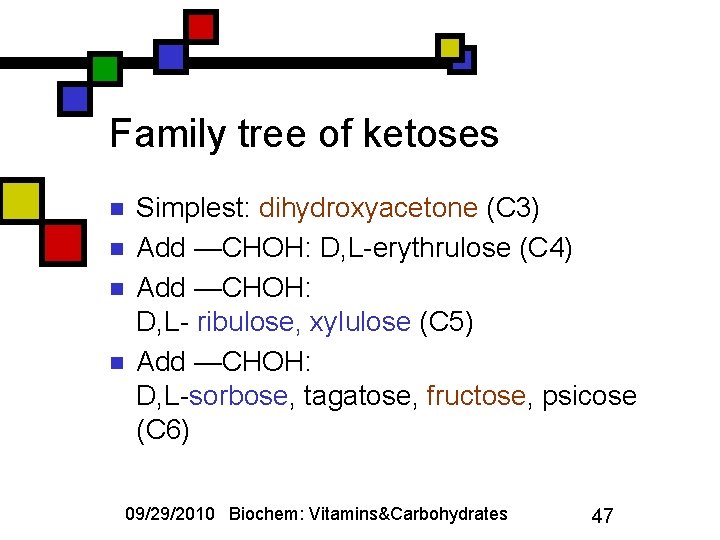
Family tree of ketoses n n Simplest: dihydroxyacetone (C 3) Add —CHOH: D, L-erythrulose (C 4) Add —CHOH: D, L- ribulose, xylulose (C 5) Add —CHOH: D, L-sorbose, tagatose, fructose, psicose (C 6) 09/29/2010 Biochem: Vitamins&Carbohydrates 47

Relative significance (? ) 09/29/2010 Biochem: Vitamins&Carbohydrates 48

Haworth projections n …provide a way of keeping track the chiral centers in a cyclic sugar, as the Fischer projections enable for straight-chain sugars 09/29/2010 Biochem: Vitamins&Carbohydrates Sir Walter Haworth 49
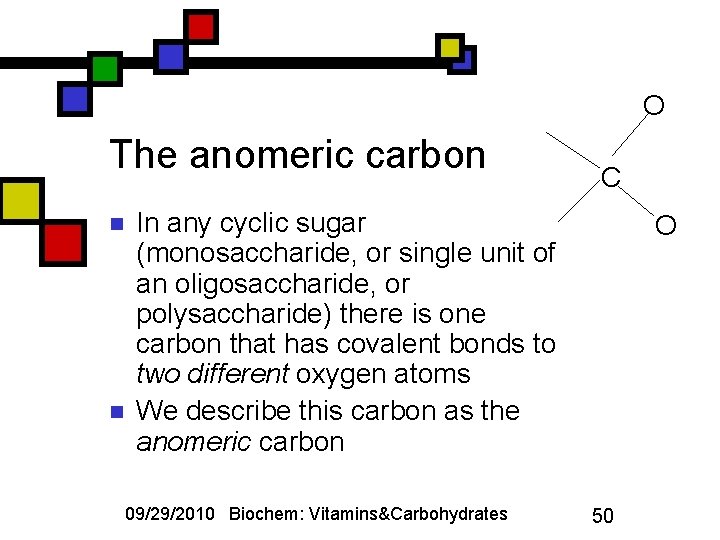
O The anomeric carbon n n C In any cyclic sugar (monosaccharide, or single unit of an oligosaccharide, or polysaccharide) there is one carbon that has covalent bonds to two different oxygen atoms We describe this carbon as the anomeric carbon 09/29/2010 Biochem: Vitamins&Carbohydrates O 50
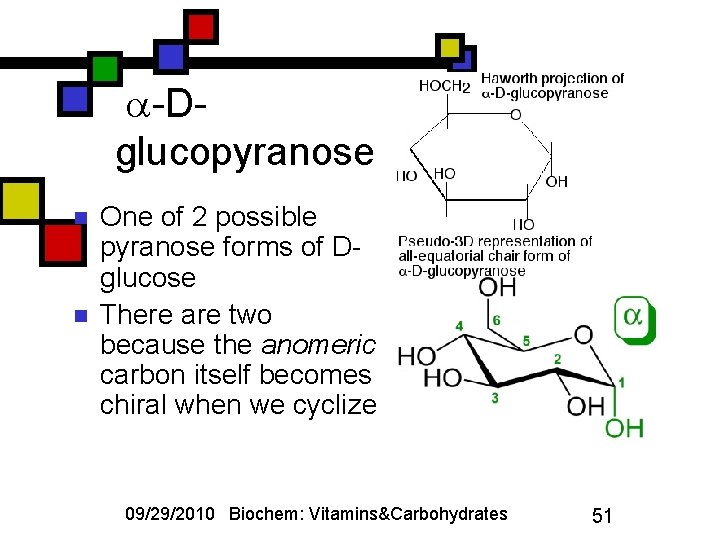
-Dglucopyranose n n One of 2 possible pyranose forms of Dglucose There are two because the anomeric carbon itself becomes chiral when we cyclize 09/29/2010 Biochem: Vitamins&Carbohydrates 51
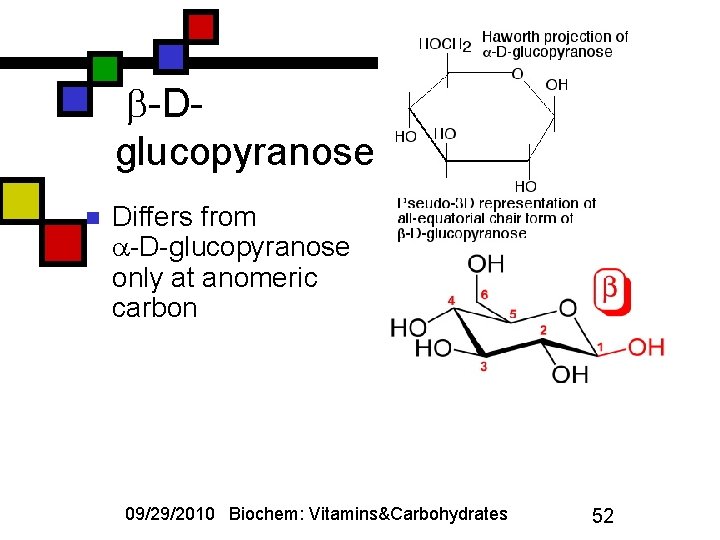
-Dglucopyranose n Differs from -D-glucopyranose only at anomeric carbon 09/29/2010 Biochem: Vitamins&Carbohydrates 52

Why is glucose special? n n All aldohexoses are fairly similar Glucose is the only aldohexose that, when built into the pyranose form, can be drawn with all its bulky substituents (–OH, –CH 2 OH) in equatorial positions This minimizes steric clashes This matters enough that some hexoses are actually marginally more stable as furanoses, e. g. fructose 09/29/2010 Biochem: Vitamins&Carbohydrates 53

Count carefully! n n n It’s tempting to think that hexoses are pyranoses and pentoses are furanoses; But that’s not always true The ring always contains an oxygen, so even a pentose can form a pyranose In solution: pyranose, furanose, openchain forms are all present Percentages depend on the sugar 09/29/2010 Biochem: Vitamins&Carbohydrates 54
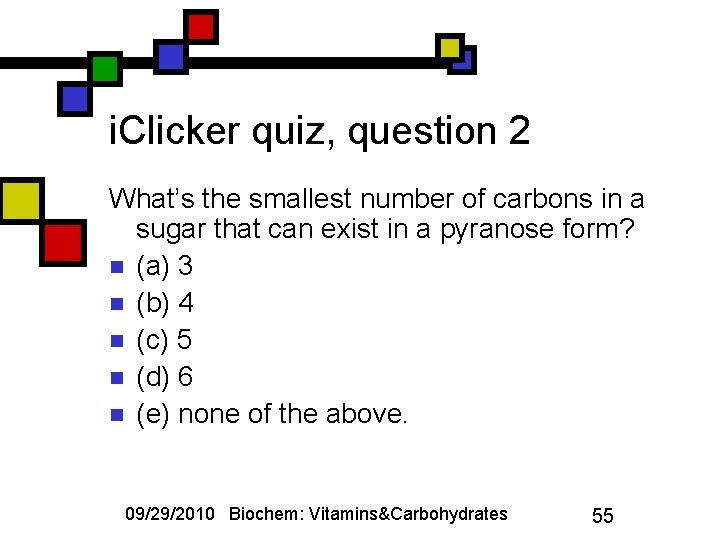
i. Clicker quiz, question 2 What’s the smallest number of carbons in a sugar that can exist in a pyranose form? n (a) 3 n (b) 4 n (c) 5 n (d) 6 n (e) none of the above. 09/29/2010 Biochem: Vitamins&Carbohydrates 55
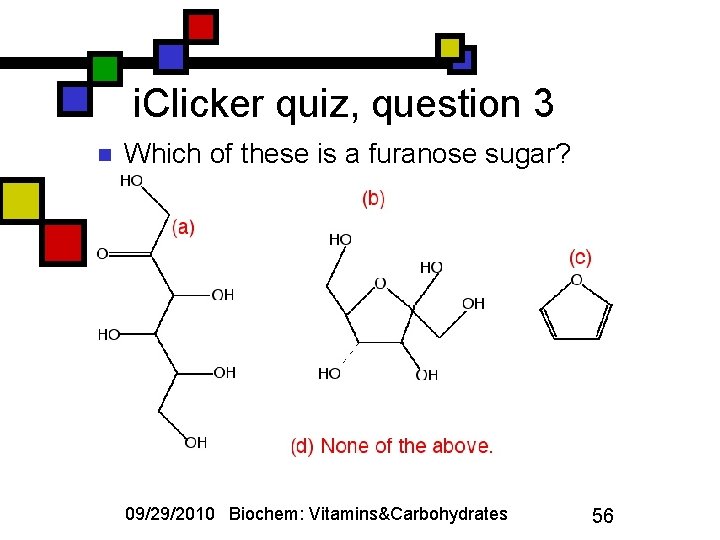
i. Clicker quiz, question 3 n Which of these is a furanose sugar? 09/29/2010 Biochem: Vitamins&Carbohydrates 56
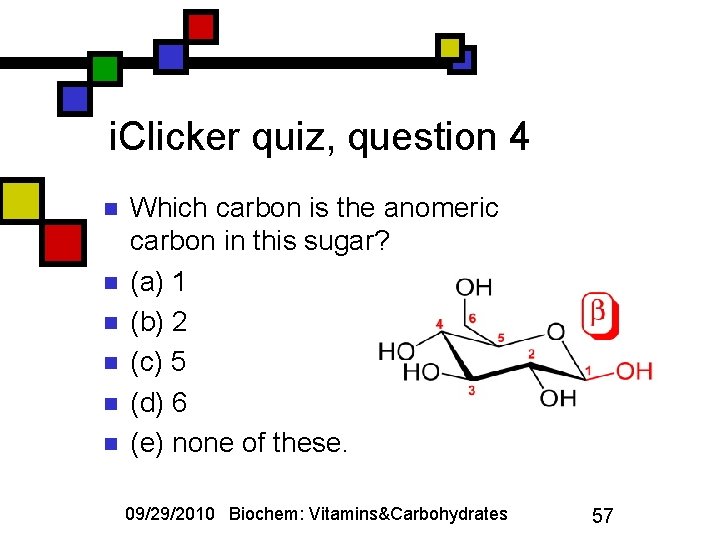
i. Clicker quiz, question 4 n n n Which carbon is the anomeric carbon in this sugar? (a) 1 (b) 2 (c) 5 (d) 6 (e) none of these. 09/29/2010 Biochem: Vitamins&Carbohydrates 57
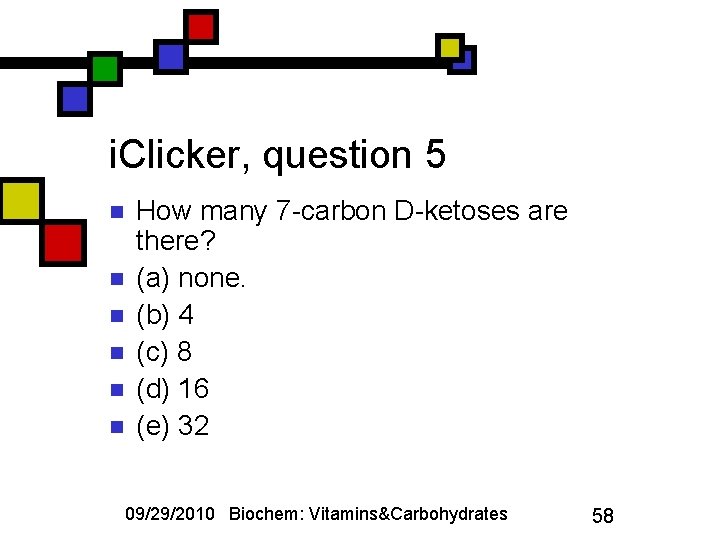
i. Clicker, question 5 n n n How many 7 -carbon D-ketoses are there? (a) none. (b) 4 (c) 8 (d) 16 (e) 32 09/29/2010 Biochem: Vitamins&Carbohydrates 58
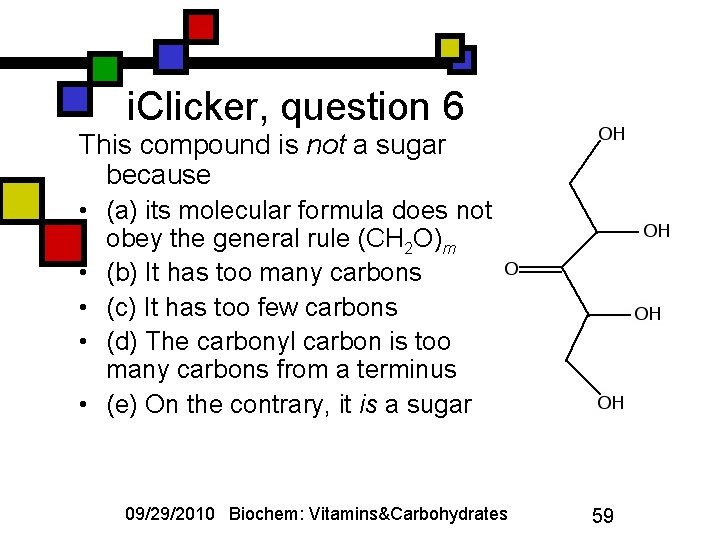
i. Clicker, question 6 This compound is not a sugar because • (a) its molecular formula does not obey the general rule (CH 2 O)m • (b) It has too many carbons • (c) It has too few carbons • (d) The carbonyl carbon is too many carbons from a terminus • (e) On the contrary, it is a sugar 09/29/2010 Biochem: Vitamins&Carbohydrates 59
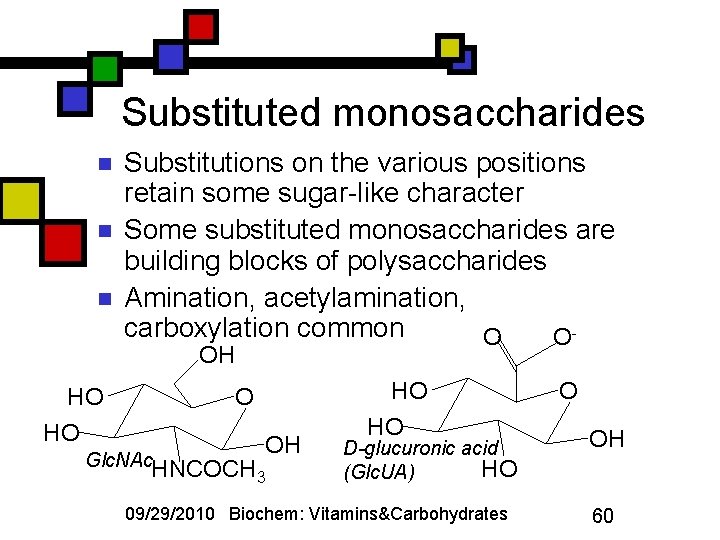
Substituted monosaccharides n n n Substitutions on the various positions retain some sugar-like character Some substituted monosaccharides are building blocks of polysaccharides Amination, acetylamination, carboxylation common O OOH HO HO O Glc. NAc. HNCOCH OH 3 HO HO D-glucuronic acid HO (Glc. UA) 09/29/2010 Biochem: Vitamins&Carbohydrates O OH 60
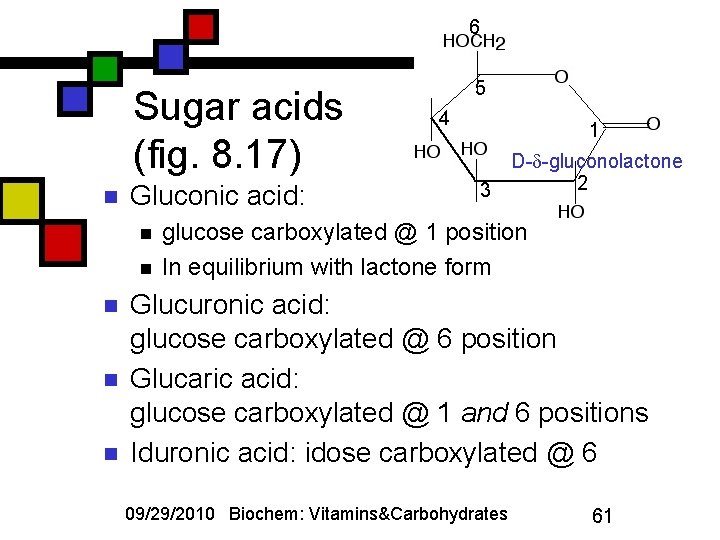
6 Sugar acids (fig. 8. 17) n Gluconic acid: n n n 5 4 1 3 D- -gluconolactone 2 glucose carboxylated @ 1 position In equilibrium with lactone form Glucuronic acid: glucose carboxylated @ 6 position Glucaric acid: glucose carboxylated @ 1 and 6 positions Iduronic acid: idose carboxylated @ 6 09/29/2010 Biochem: Vitamins&Carbohydrates 61
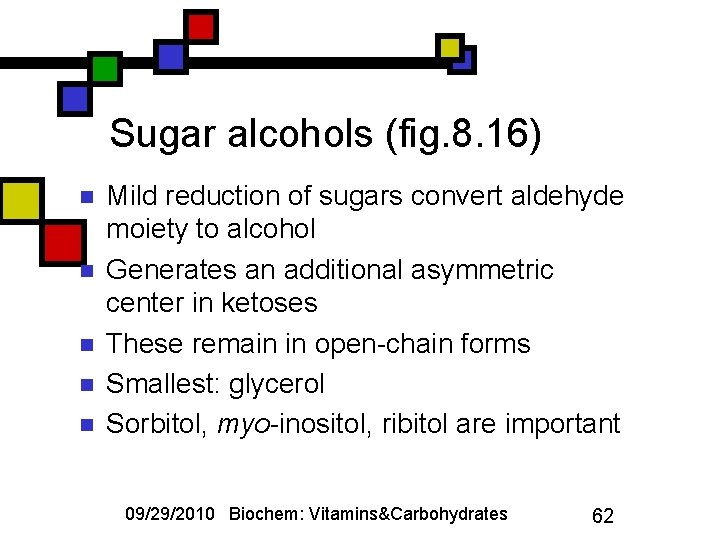
Sugar alcohols (fig. 8. 16) n n n Mild reduction of sugars convert aldehyde moiety to alcohol Generates an additional asymmetric center in ketoses These remain in open-chain forms Smallest: glycerol Sorbitol, myo-inositol, ribitol are important 09/29/2010 Biochem: Vitamins&Carbohydrates 62
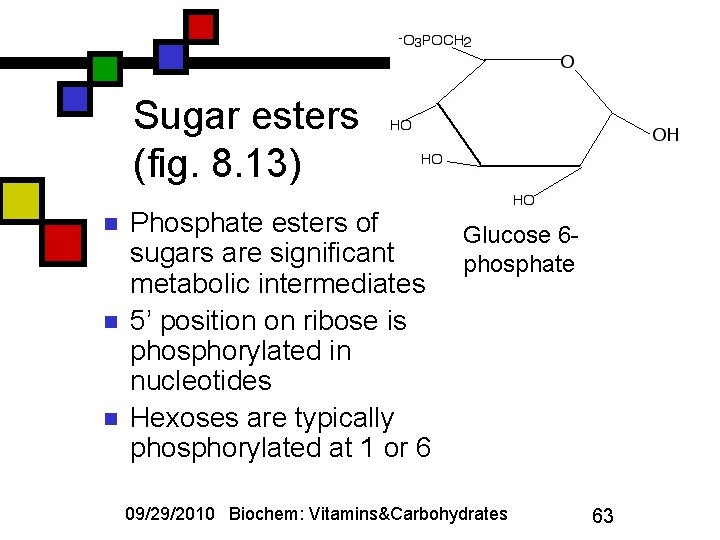
Sugar esters (fig. 8. 13) n n n Phosphate esters of sugars are significant metabolic intermediates 5’ position on ribose is phosphorylated in nucleotides Hexoses are typically phosphorylated at 1 or 6 Glucose 6 phosphate 09/29/2010 Biochem: Vitamins&Carbohydrates 63
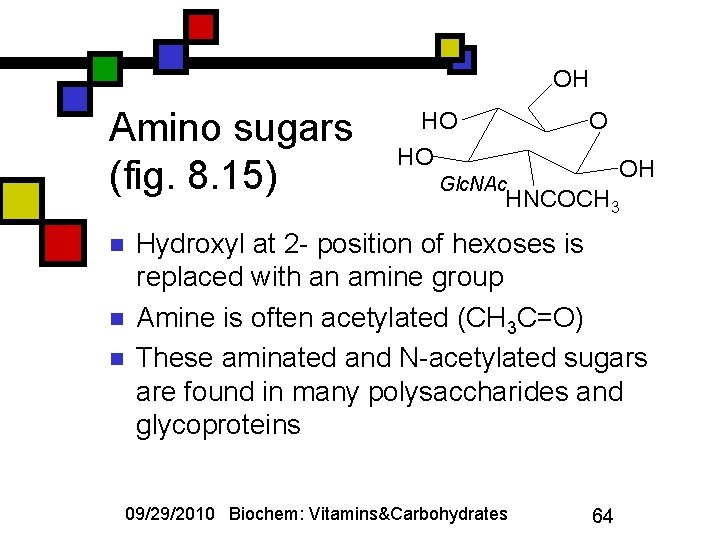
OH Amino sugars (fig. 8. 15) n n n HO OH Glc. NAc HNCOCH 3 Hydroxyl at 2 - position of hexoses is replaced with an amine group Amine is often acetylated (CH 3 C=O) These aminated and N-acetylated sugars are found in many polysaccharides and glycoproteins 09/29/2010 Biochem: Vitamins&Carbohydrates 64
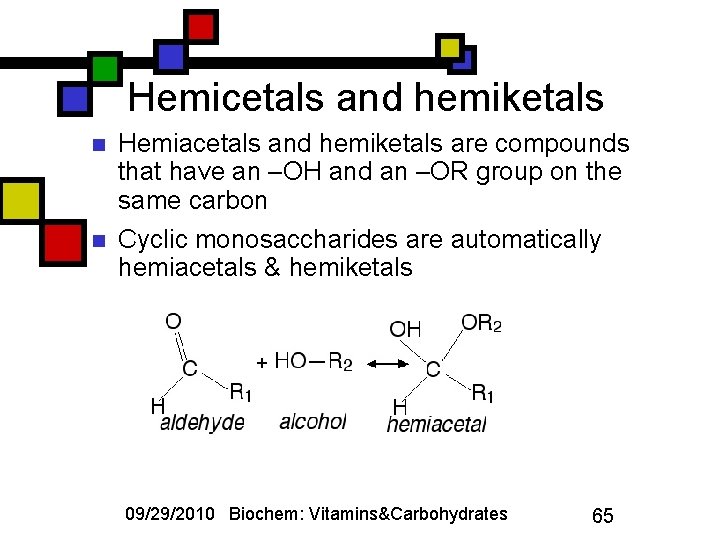
Hemicetals and hemiketals n n Hemiacetals and hemiketals are compounds that have an –OH and an –OR group on the same carbon Cyclic monosaccharides are automatically hemiacetals & hemiketals 09/29/2010 Biochem: Vitamins&Carbohydrates 65

Oligosaccharides and other glycosides n n A glycoside is any compound in which the hydroxyl group of the anomeric carbon is replaced via condensation with an alcohol, an amine, or a thiol All oligosaccharides are glycosides, but so are a lot of monomeric sugar derivatives, like nucleosides 09/29/2010 Biochem: Vitamins&Carbohydrates 66

Sucrose: a glycoside n n A disaccharide Linkage is between anomeric carbons of contributing monosaccharides, which are glucose and fructose 09/29/2010 Biochem: Vitamins&Carbohydrates 67

Other disaccharides n Maltose n n n Cellobiose n n n glc-glc with -glycosidic bond from left-hand glc Produced in brewing, malted milk, etc. -glc Breakdown product from cellulose Lactose: -gal-glc n n Milk sugar Lactose intolerance caused by absence of enzyme capable of hydrolyzing this glycoside 09/29/2010 Biochem: Vitamins&Carbohydrates 68

Reducing sugars n n Sugars that can undergo ring-opening to form the open-chain aldehyde compounds that can be oxidized to carboxylic acids We describe those as reducing sugars because they can reduce metal ions or amino acids in the presence of base Benedict’s test: 2 Cu 2+ + RCH=O + 5 OH- Cu 2 O + RCOO- + 3 H 2 O Cuprous oxide is red and insoluble 09/29/2010 Biochem: Vitamins&Carbohydrates 69
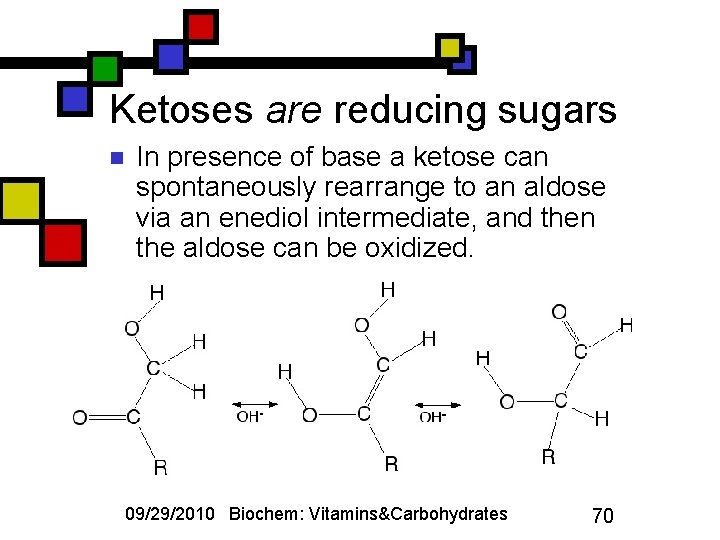
Ketoses are reducing sugars n In presence of base a ketose can spontaneously rearrange to an aldose via an enediol intermediate, and then the aldose can be oxidized. 09/29/2010 Biochem: Vitamins&Carbohydrates 70

Sucrose: not a reducing sugar n n Both anomeric carbons are involved in the glycosidic bond, so they can’t rearrange or open up, so it can’t be oxidized Bottom line: only sugars in which the anomeric carbon is free are reducing sugars 09/29/2010 Biochem: Vitamins&Carbohydrates 71

Why does this matter? n n Partly historical: this cuprate reaction was one of the first well-characterized tools for characterizing these otherwise very similar compounds But it also gives us a convenient way of distinguishing among types of glycosidic arrangements, even if we never really use Cu 2+ ions in experiments 09/29/2010 Biochem: Vitamins&Carbohydrates 72

Reducing & nonreducing ends n n Typically, oligo and polysaccharides have a reducing end a nonreducing end Non-reducing end is the sugar moiety whose anomeric carbon is involved in the glycosidic bond Reducing end is sugar whose anomeric carbon is free to open up and oxidize Enzymatic lengthening and degradation of polysaccharides occurs at nonreducing end or ends 09/29/2010 Biochem: Vitamins&Carbohydrates 73

Nucleosides n n n Anomeric carbon of ribose (or deoxyribose) is linked to nitrogen of RNA (or DNA) base (A, C, G, T, U) Generally ribose is in furanose form This is an example of an N-glycoside Diagram courtesy of World of Molecules 09/29/2010 Biochem: Vitamins&Carbohydrates 74
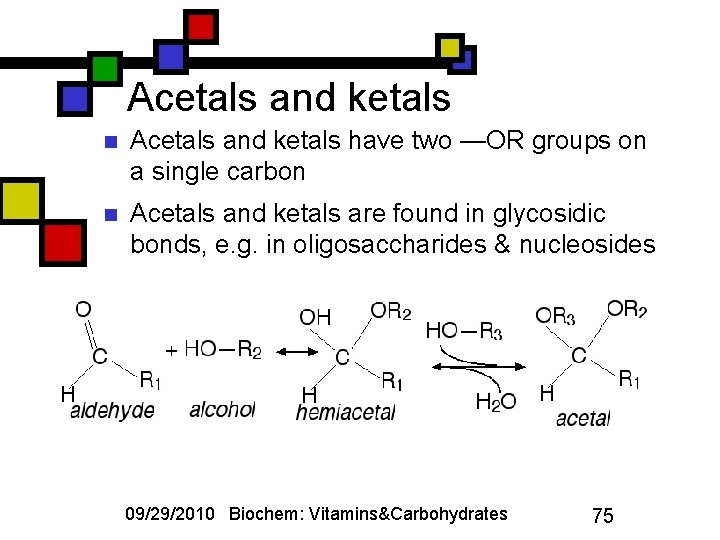
Acetals and ketals n Acetals and ketals have two —OR groups on a single carbon n Acetals and ketals are found in glycosidic bonds, e. g. in oligosaccharides & nucleosides 09/29/2010 Biochem: Vitamins&Carbohydrates 75

Polysaccharides n n n Homoglycans: all building blocks same Heteroglycans: more than one kind of building block No equivalent of genetic code for carbohydrates, so long ones will be heterogeneous in length and branching, and maybe even in monomer identity 09/29/2010 Biochem: Vitamins&Carbohydrates 76
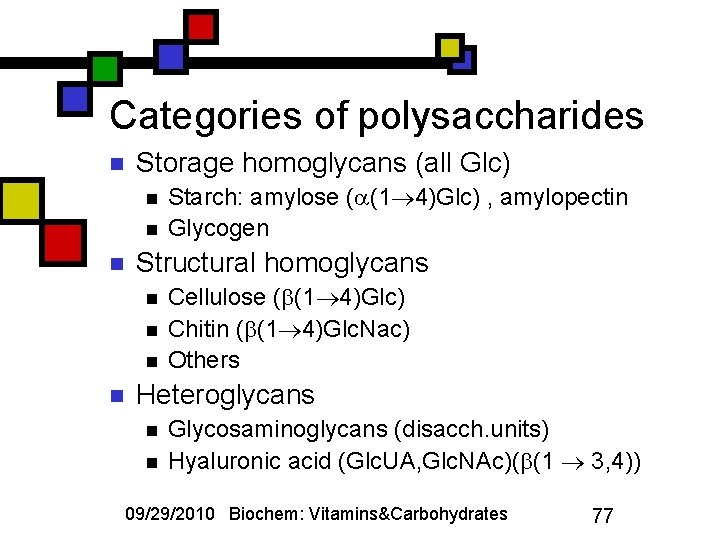
Categories of polysaccharides n Storage homoglycans (all Glc) n n n Structural homoglycans n n Starch: amylose ( (1 4)Glc) , amylopectin Glycogen Cellulose ( (1 4)Glc) Chitin ( (1 4)Glc. Nac) Others Heteroglycans n n Glycosaminoglycans (disacch. units) Hyaluronic acid (Glc. UA, Glc. NAc)( (1 3, 4)) 09/29/2010 Biochem: Vitamins&Carbohydrates 77

Storage polysaccharides n n Available sources of glucose for energy and carbon Long-chain polymers of glucose n n n Starch (amylose and amylopectin): in plants, it’s stored in 3 -100 µm granules Glycogen Branches found in all but amylose 09/29/2010 Biochem: Vitamins&Carbohydrates 78

Amylose n n Unbranched, -1 4 linkages Typically 100 -1000 residues Not soluble but can form hydrated micelles and may be helical Amylases hydrolyze -1 4 linkages Diagram courtesy Langara College 09/29/2010 Biochem: Vitamins&Carbohydrates 79
Amylopectin n n Mostly -1 4 linkages; 4% -1 6 Each sidechain has 15 -25 glucose moieties -1 6 linkages broken down by debranching enzymes 300 -6000 total glucose units per amylopectin molecule One reducing end, many nonreducing ends 09/29/2010 Biochem: Vitamins&Carbohydrates 80

Glycogen n n n Principal storage form of glucose in human liver; some in muscle Branched ( -1 4 + a few -1 6) More branches (~10%) Larger than starch: 50000 glucose One reducing end, many nonreducing ends Broken down to G-1 -P units Built up from G-6 -P G-1 -P UDP-Glucose units 09/29/2010 Biochem: Vitamins&Carbohydrates 81

Glycogen structure 09/29/2010 Biochem: Vitamins&Carbohydrates 82

Structural polysaccharides I n n Insoluble compounds designed to provide strength and rigidity Cellulose: glucose -1 4 linkages n n n Rigid, flat structure: each glucose is upside down relative to its nearest neighbors 300 -15000 glucose units Found in plant cell walls Resistant to most glucosidases Cellulases found in termites, ruminant gut bacteria Chitin: Glc. NAc -1 4 linkages: exoskeletons, cell walls (fig. 7. 26) 09/29/2010 Biochem: Vitamins&Carbohydrates 83
Structural polysaccharides II n Alginates: poly( -D-mannuronate), poly( -L-guluronate), linked 1 4 n n n Agarose: alternating D-gal, 3, 6 -anhydro-L-gal, with 6 -methyl-D-gal side chains n n n Cellulose-like structure when free Complexed to metal ions: 3 -fold helix (“egg-carton”) Forms gels that hold huge amounts of H 2 O Can be processed to use in the lab for gel exclusion chromatography Glycosaminoglycans: see next section 09/29/2010 Biochem: Vitamins&Carbohydrates 84
- Slides: 84