Vitamin K and Vitamin K Deficiency Bleeding in

Vitamin K and Vitamin K Deficiency Bleeding in Infants What Providers Need to Know Lauren Marcewicz, MD Centers for Disease Control and Prevention Division of Blood Disorders July 10, 2014 National Center on Birth Defects and Developmental Disabilities Division of Blood Disorders
To hear this webinar, please also dial in to 1 -866 -694 -4541 and use participant code 13565573 National Center on Birth Defects and Developmental Disabilities Division of Blood Disorders

Learning Objectives § § Describe basic vitamin K physiology in infants Describe the different types of vitamin K deficiency (VKDB) bleeding and their associated severities Explain various concerns about vitamin K prophylaxis List resources for clinicians and families regarding vitamin K prophylaxis
What is Vitamin K? § Group of fat-soluble vitamins • • § Role • • • § K 1, found mainly in plants K 2, form stored in animals – multiple forms Blood coagulation: activates clotting factors Bone metabolism Vascular health Sources • • • Leafy green foods Cheeses and dairy, liver Made by intestinal bacteria
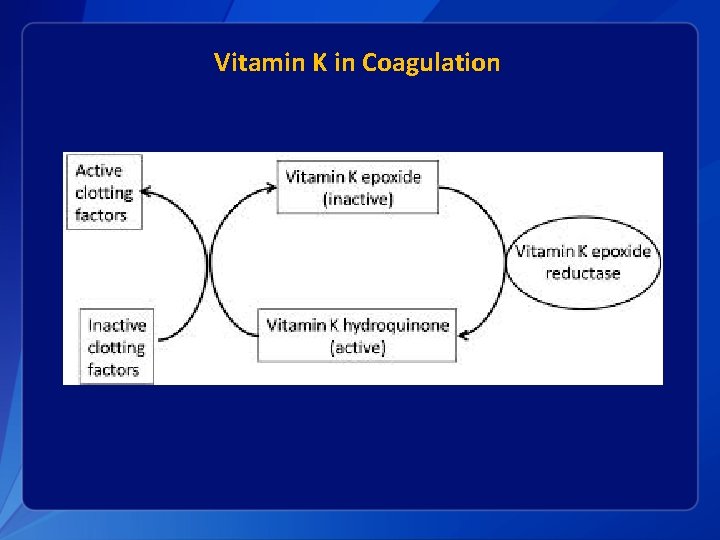
Vitamin K in Coagulation
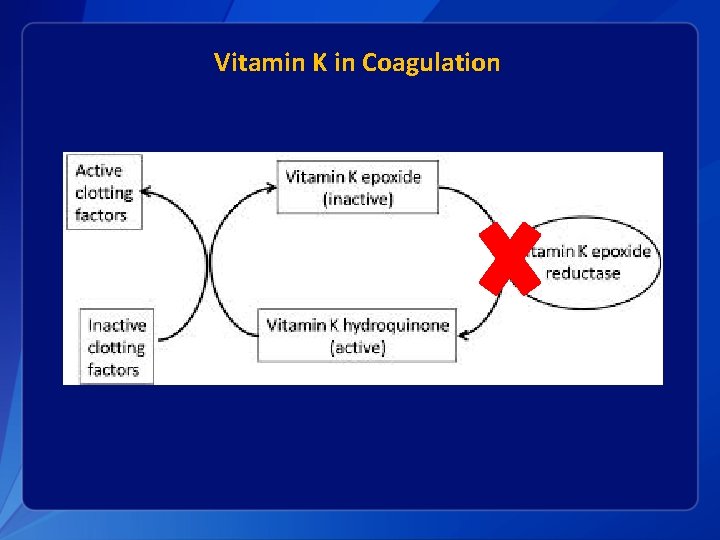
Vitamin K in Coagulation

Vitamin K in Infants § Infants are relatively vitamin K deficient when compared with adults: • • • § Plasma K 1 concentrations lower K 2 virtually absent in the liver Concentrations of plasma vitamin K higher in mothers than in their infants Lower levels of active vitamin K dependent factors PIVKA Deficiency probably intensifies in first few days: • • Prothrombin activity falls PIVKA levels increase

Physiology of Vitamin K Deficiency § Placental barrier • Not complete – some K makes it to fetus § At birth, infant is removed from placental source § No other source: • • • Colonic bacterial colonization incomplete Liver stores inadequate Breastmilk contains relatively low amounts of vitamin K
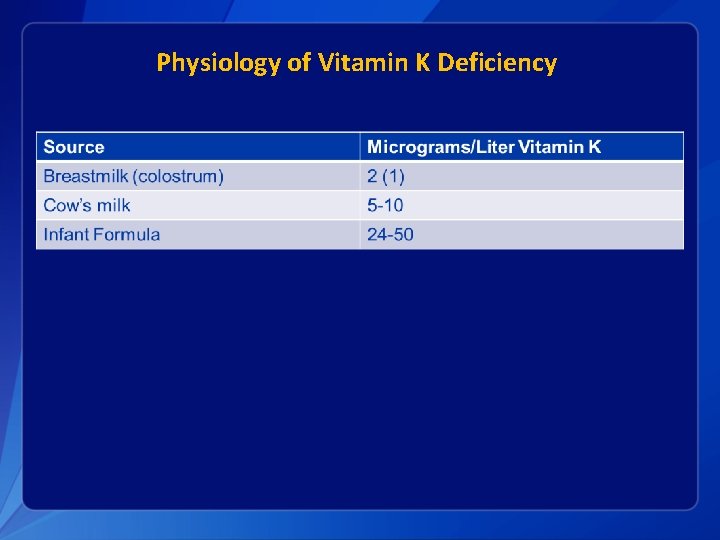
Physiology of Vitamin K Deficiency
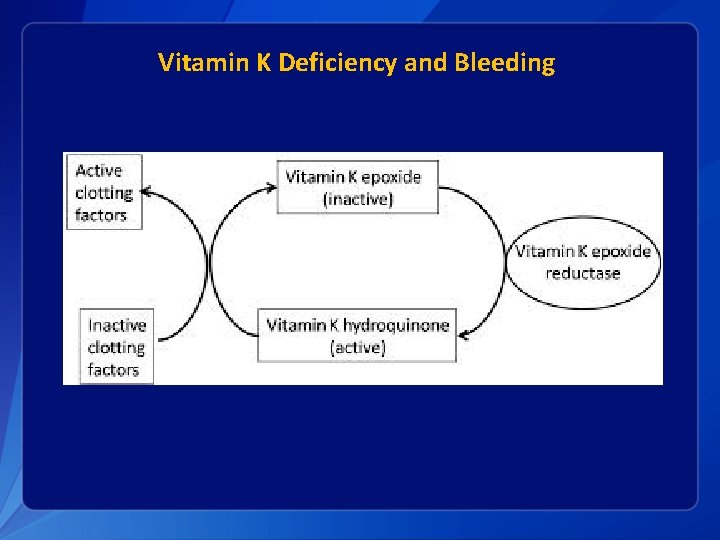
Vitamin K Deficiency and Bleeding
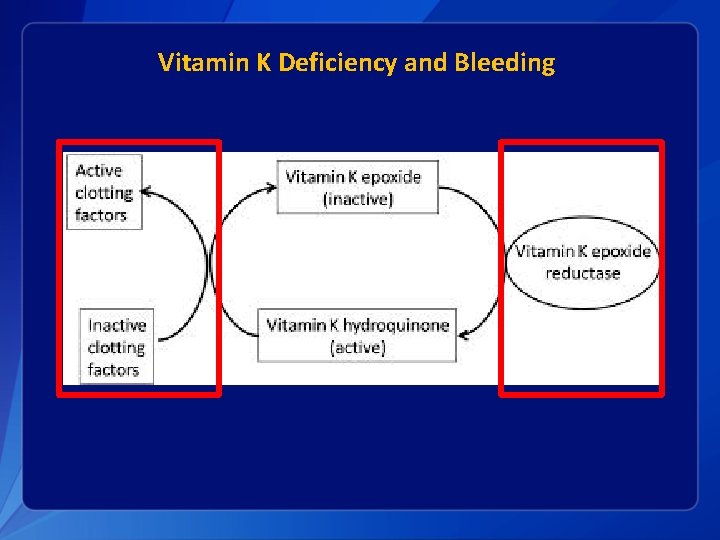
Vitamin K Deficiency and Bleeding

Vitamin K Deficiency Bleeding § For some infants, vitamin K deficiency leads to deficiency of active clotting factors and can result in hemorrhage: Vitamin K Deficiency Bleeding (VKDB) § VKDB classified by timing of presentation/diagnosis and etiology

Etiologies and Risk Factors § Idiopathic – no particular reason why this particular child developed VKDB • Associated with breast feeding • Slight male predominance • Summer months? § Secondary – predisposition to vitamin K deficiency itself • Cholestatic liver disease • Malabsorption • Diarrheal illness
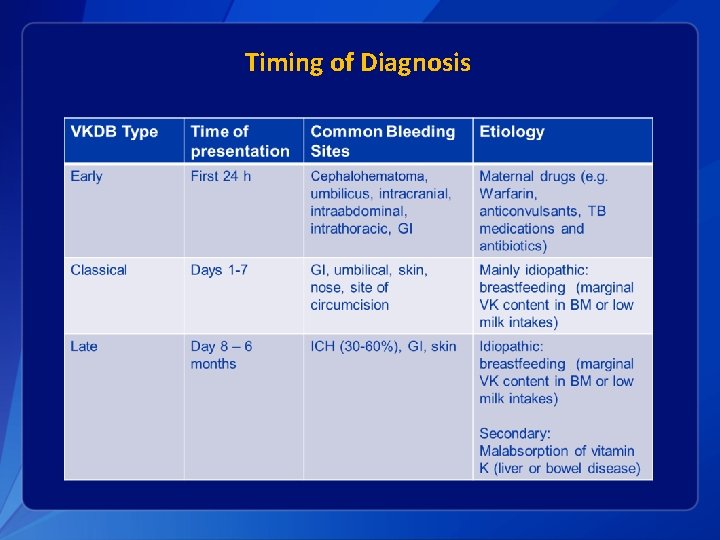
Timing of Diagnosis

How Common is VKDB? § Early estimated to occur in 6 -12% of women who take particular drugs § Classical estimated to occur in 0. 25 – 1% of infants § Late • • • Incidence typically quoted is 4 -7/100, 000: based on a 1993 review that applied specific parameters and re-calculated previous estimates – also leaves out a 10. 5/100, 000 number from Japan 116/100, 000 in Vietnam (based only on intracranial hemorrhages) and 72/100, 000 in Thailand Practical problems with both the at-risk population estimate and case ascertainment

Preventive Strategies • Early: supplement mothers with vitamin K • Classic/Late: o Intramuscular vitamin K, 0. 5– 1 mg o Recommended since 1961 o Oral vitamin K not as effective: need doses for 3 months

Vitamin K Works! § § Incidence falls to <1/100, 000 with intramuscular prophylaxis Relative risk for VKDB without injected prophylaxis is 81 times greater than if it is given Basically an unknown entity in U. S. and developed countries Higher incidences, large cohorts and case series reported from countries without prophylaxis programs (Ethiopia, Turkey, Vietnam and Thailand)

Investigation § Cluster in Tennessee: 5 cases, 1 near miss • 4 intracranial bleeds, one gastrointestinal bleed • Parents had opted out of vitamin K at birth in each case § Objectives included looking at incidence of VKDB and risk factors, but also understanding why parents opted out – and how common this is
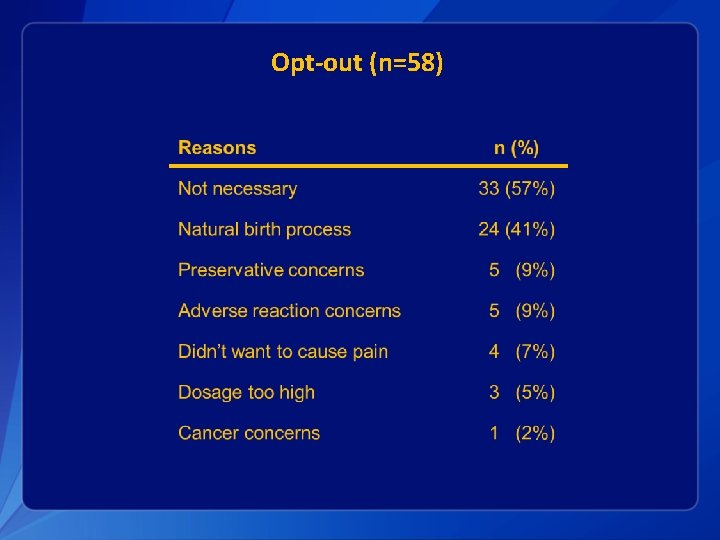
Opt-out (n=58)

How Prevention Works § Sufficiently severe and/or widespread condition • If condition is rare, should be severe and costly § Means of prevention is effective and does not cause more harm than the condition it prevents

The Vitamin K Story § Prior to 1961, massive doses of vitamin K were used (up to 80 mg/kg ) • • § Severe hemolytic anemia, hyperbilirubinemia sufficient to cause kernicterus 1961 AAP recommendation in response 1977 – cord blood rarely contained PIVKA; administration of vitamin K not supported • • 1978 Lancet editorial endorsed “selective vitamin K prophylaxis” Many hospitals began giving vitamin K only to “high risk” infants

Link With Cancer § 1990’s: Golding • 1 st paper – drugs given to infant was associated with development of childhood cancer (OR 2. 6) • 2 nd paper – intramuscular vitamin K-specific risk for cancer, OR 1. 97, specifically leukemia, OR 2. 65 § Association has not been replicated reliably • Subgroup analyses – 1 -6 years, 12 -17 months • Unclear administration • Largest case-control using clear recording of injection administration – no increased risk

Preservatives § Benzyl alcohol • Linked to severe toxicity in neonates in early 80’s • Used in multiple medications, including fluids – large doses administered • Once recognized, removed from fluids and minimized in neonatal medicine § Preservative-free vitamin K available

Dosage § Optimal dosage for an infant not well-established: • Formula-fed infants appear to be protected with about 50 mcg/ml • Slightly higher dose might be adequate for children with absorptive issues § § § Plasma levels with injection reach a peak about 40 times this Likely depot effect No syndrome of hypervitaminosis K

Granular Details § § § “People have been giving birth for centuries without a problem” “Unfortunately, someone’s palms get greased…money is involved in all the interventions” “We won’t know for years about what we’re putting into our bodies” “I just couldn’t understand why they needed vitamin K if it was something external” “Children get what they need in the first few days from their mother”

Two Broad Lines of Thought § Unnecessary § Multiple negative consequences of the shot

Two Broad Lines of Thought § Unnecessary • Sufficiently severe and/or widespread condition o If condition is rare, should be severe and costly § Multiple negative consequences of the shot

Two Broad Lines of Thought § Unnecessary • Sufficiently severe and/or widespread condition o If condition is rare, should be severe and costly § Multiple negative consequences of the shot • Means of prevention is effective and does not cause more harm than the condition it prevents

How Common is Opt-Out? § Hospitals: • 5 Nashville hospitals, 1 in an outlying county where 3 cases had come from • Random sample of 2013 births + chart review • 1 hospital recorded opt-out § • 2. 9% infants did not receive vitamin K Birthing centers: • 5 across the state; requested data • 31. 3% infants did not receive vitamin K

Reality Is:

Resources for Parents § Website: cdc. gov/ncbddd/vitamink • • § Basic information FAQs addressing concerns Parent stories Parent factsheet Evidencebasedbirth. com

Resources for Clinicians § Website: cdc. gov/ncbddd/vitamink • Podcast • Medscape video • Provider factsheet § Partner messaging • • AAFP ACOG AAP AWOHNN

American Academy of Pediatrics § Front page story in AAP News § Policy statement § Healthychildren. org

More Data Needed § Surveillance proposal with ASPHO: cases of VKDB • 1 year retrospective, 2 years prospective • Provider survey § Surveillance proposal for institution-based review • 10 year retrospective, 2 year prospective • Tracking form/EHR addition § Student/provider knowledge assessment

Acknowledgements § § § EIS Officers • Joshua Clayton • Matthew Maenner EIS Office • Danice Eaton • Randolph Daley • Rachel Avchen NCBDDD • Coleen Boyle • Lisa Richardson • Mike Soucie • Craig Hooper • Althea Grant • Ekwutosi Okoroh • Erika Odom • Daisy Christensen § § • Alyson Goodman • Gretchen Simmons Tennessee Department of Health • Tim Jones • John Dunn • Michael Warren • Julie Traylor • Angela Miller • Yinmei Li • George Wade Vanderbilt University • Robert Sidonio • Anna Morad • Stacey Morgan Schlafly
Questions? For more information about this webinar series, please contact Cynthia Sayers at cay 1@cdc. gov. Today’s slide presentation will be accessible through CDC’s website at http: //www. cdc. gov/ncbddd/blooddisorders/index. html Centers for Disease Control and Prevention 1600 Clifton Road NE, Atlanta, GA 30333 Telephone: 1 -800 -CDC-INFO (232 -4636)/TTY: 1 -888 -232 -6348 Visit: www. cdc. gov | Contact CDC at: 1 -800 -CDC-INFO or www. cdc. gov/info The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention. National Center on Birth Defects and Developmental Disabilities Division of Blood Disorders

The Experts
- Slides: 37