VIRUS ENTOMOPATOGEN By Irda Safni Virus Virus berasal

VIRUS ENTOMOPATOGEN By Irda Safni

Virus

§ “Virus” berasal dari bahasa Latin, yang artinya racun dan selalu berhubungan dengan penyakit dan kematian. § Definisi virus (modern): biosistem paling dasar yang menunjukkan keragaman makromolekuler yang mampu berkembang biak dan membelah sendiri, tapi kurang mampu bereaksi terhadap faktor lingkungan, tidak dapat bergerak sendiri, dan tidak dapat menggunakan sumber energi metaboliknya sendiri bertolak belakang dengan definisi makhluk hidup

§ Kebanyakan biosistem dasar yang menunjukkan kompleksitas makromolekul dan mampu bereplikasi sendiri dan berevolusi, tetapi kekurangan iritabilitas (kemampuan untuk bereaksi terhadap faktor lingkungan), pergerakan sendiri, dan sumber energi metabolik sendiri, berlawanan dengan sifat dasar yang merupakan ciri-ciri makhluk hidup.

§ Setelah virus partikel masuk ke dalam sel yang peka, asam nukleatnya akan mengambil alih sistem metabolik sel dan bereplikasi menjadi virus-virus partikel yang baru, hingga isi sel inangnya habis dan akhirnya mati.
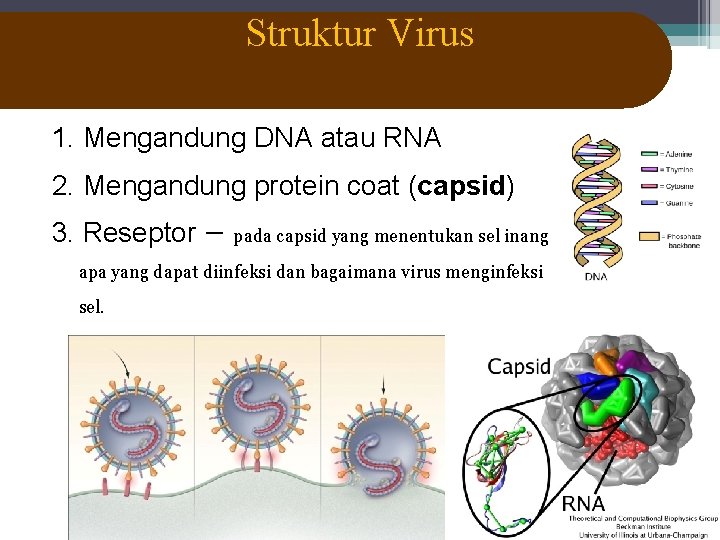
Struktur Virus 1. Mengandung DNA atau RNA 2. Mengandung protein coat (capsid) 3. Reseptor – pada capsid yang menentukan sel inang apa yang dapat diinfeksi dan bagaimana virus menginfeksi sel.
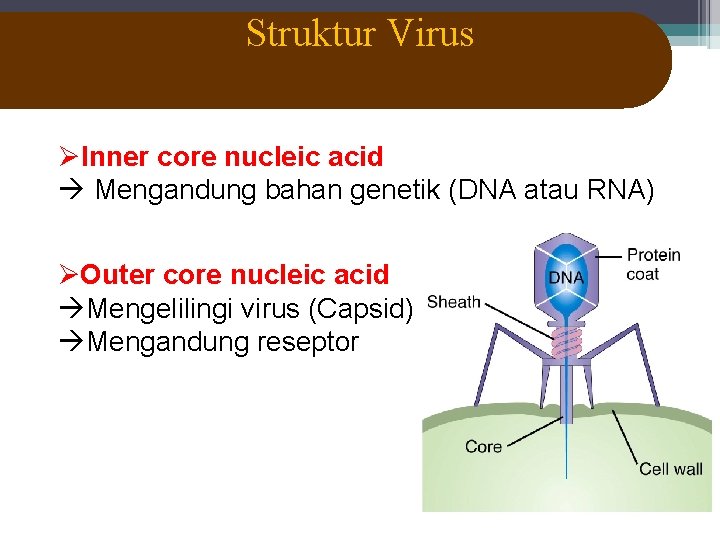
Struktur Virus ØInner core nucleic acid Mengandung bahan genetik (DNA atau RNA) ØOuter core nucleic acid Mengelilingi virus (Capsid) Mengandung reseptor
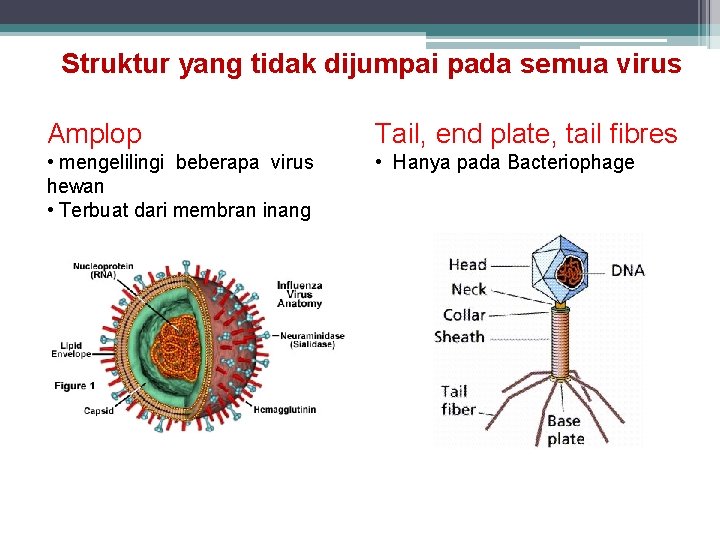
Struktur yang tidak dijumpai pada semua virus Amplop Tail, end plate, tail fibres • mengelilingi beberapa virus hewan • Terbuat dari membran inang • Hanya pada Bacteriophage

Siklus Hidup Virus • Virus adalah parasit obligat intracellular, yang artinya mereka tidak dapat bereplikasi atau mengekspresikan gennya tanpa bantuan sel hidup. • Setiap virus memiliki kisaran inang, sejumlah sel inang yang dapat diinfeksinya.

Tahap Perbanyakan Virus Setelah virus menginfeksi inangnya, komponen turunan virus dihasilkan oleh mesin sel inangnya Perakitan kapsid virus adalah proses non-enzimatik. Biasanya secara spontan. Biasanya virus hanya dapat menginfeksi sejumlah kecil inang (dikenal dengan nama "kisaran inang"). Mekanisme "gembok dan kunci" adalah prinsip utama kisaran inang virus. Protein tertentu pada partikel virus harus cocok pada tempat reseptor tertentu pada permukaan sel inang.

Siklus Hidup Virus • Untuk dapat bertahan hidup, virus harus mampu melakukan sebagai berikut: ▫ 1. Menemukan sel inang yang di dalamnya virus dapat bereplikasi ▫ 2. Mengikat pada sel ▫ 3. Memasuki sel ▫ 4. Melepas genomnya agar dapat bereplikasi ▫ 5. Mereplikasi genomnya ▫ 6. Transkripsi dan translasi protein virusnya ▫ 7. Membungkus genom dan proteinnya ▫ 8. keluar dari sel
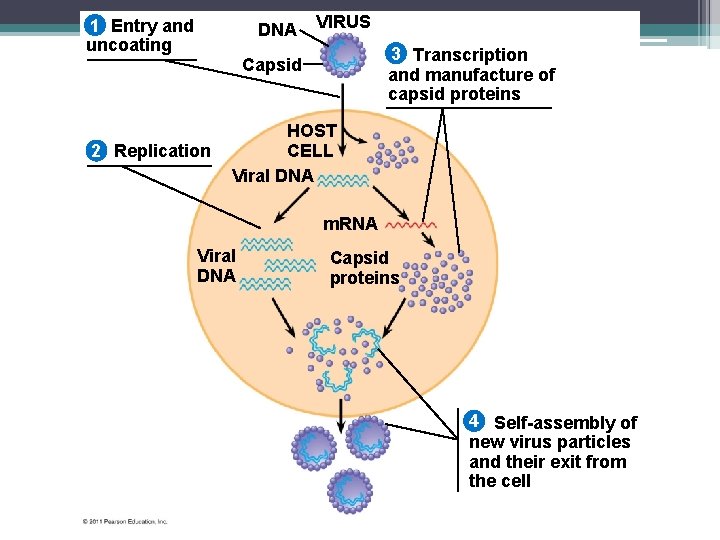
Figure 19. 4 1 Entry and uncoating DNA VIRUS 3 Transcription and manufacture of capsid proteins Capsid 2 Replication HOST CELL Viral DNA m. RNA Viral DNA Capsid proteins 4 Self-assembly of new virus particles and their exit from the cell

Virus Entomopatogen

§ Penyakit yang disebabkan virus entomopatogen mulai diketahui sejak abad ke-16. § Penyakit yang disebut Jaundice o graserrie, sekarang diidentifikasi sebagai nucleopolyhedrosis, ditemukan pada ulat sutra (Bobyx mory) oleh Vida pada tahun 1524 dan kemudian juga diisolasi dari lebah madu (Apis mellifera). § Pada tahun 1856, dua orang ahli Italia (Maestri dan Cornalia) menjelaskan occlusion bodies (OBs) ulat sutra nucleopolyhedrosis. § Pada tahun 1926 Paillot mendeskripsikan granulovirus (GVs) pertama sekali. § Pada tahun 1934 Ishimori menjelaskan jenis baru polyhedrosis di dalam ulat sutra OBs dibentuk didalam sitoplasma sel yang diinfeksi (bukan pada asam nukleat) sekarang dikenal dengan cypovirus.

§ Sejak tahun 1950 s/d 1970, Steinhaus dan koleganya menguji Baculovirus sebagai agens hayati di lapangan dengan mengaplikasi nucleopolyhedrovirus (NPV) untuk mengendalikan ulat alfalfa (Colias eurytheme Boisduval; Lepidoptera: Pieridae). § Bioinsektisida komersil berbahan aktif virus pertama dikembangkan pertama sekali pada tahun 1975 oleh Perusahaan Sandoz (dengan nama dagang Elcar) untuk mengendalikan Heliothis/Helicoverpa Lepidoptera: Noctuidae). § Selama tahun 1979 s/d 1980, penemuan penting pada genetika virus entomopatogen, khususnya Baculovirus. § Hingga saat ini studi genetika virus entomopatogen difokuskan pada studi genom lengkap telah ada 29 sekuensing genom lengkap virus entomopatogen.

§ Sama seperti bakteri, virus entomopatogen harus ditelan oleh serangga inangnya, sehingga efektif untuk mengendalikan serangga yang memiliki tipe mulut pengunyah. § Beberapa serangga Lepidoptera adalah inang penting Baculovirus, termasuk nucleopolyhedroviruses (NPV) dan Granuloviruses (GV).
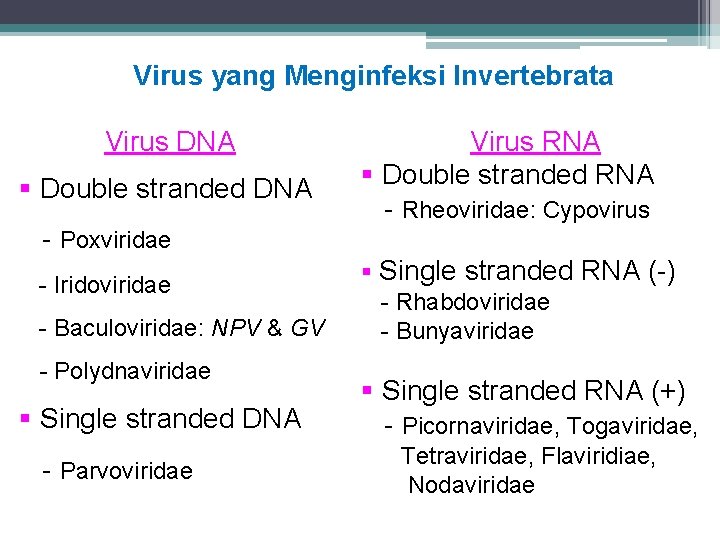
Virus yang Menginfeksi Invertebrata Virus DNA § Double stranded DNA - Poxviridae - Iridoviridae - Baculoviridae: NPV & GV - Polydnaviridae § Single stranded DNA - Parvoviridae Virus RNA § Double stranded RNA - Rheoviridae: Cypovirus § Single stranded RNA (-) - Rhabdoviridae - Bunyaviridae § Single stranded RNA (+) - Picornaviridae, Togaviridae, Tetraviridae, Flaviridiae, Nodaviridae
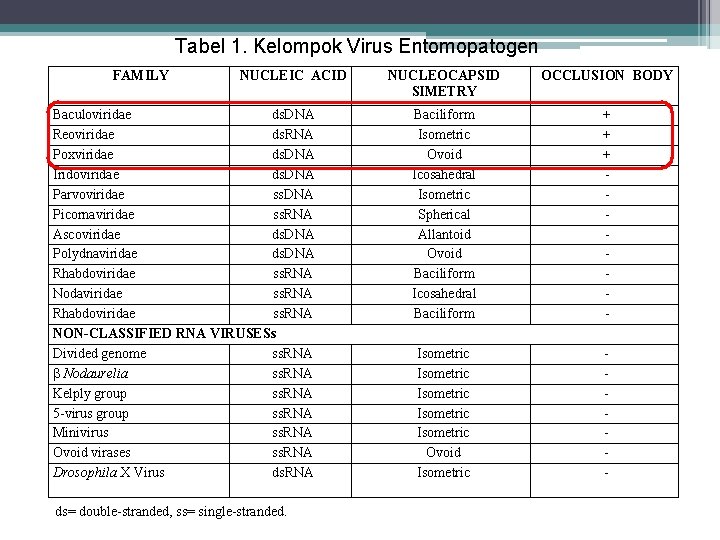
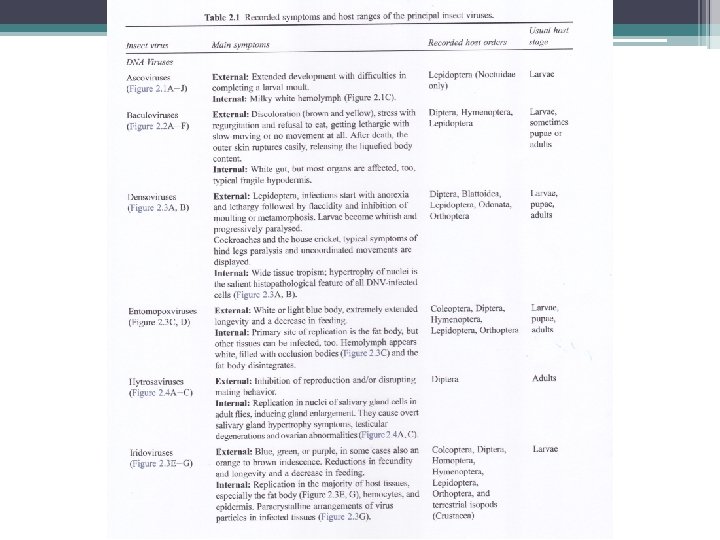
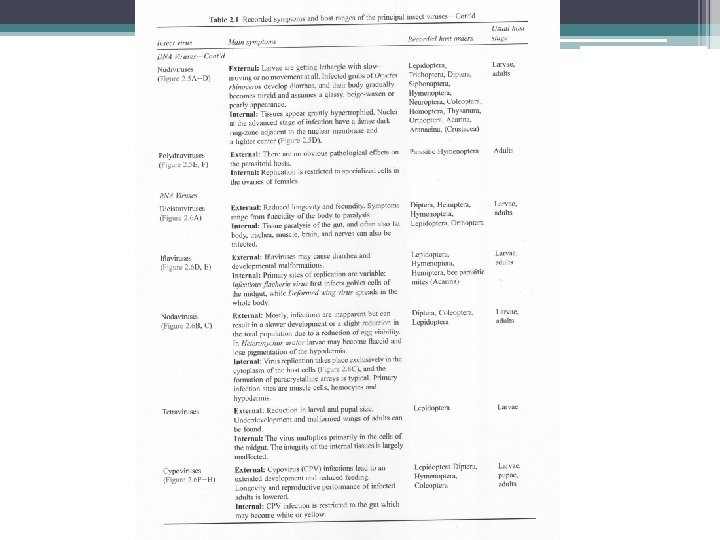
Tabel 1. Kelompok Virus Entomopatogen FAMILY NUCLEIC ACID Baculoviridae ds. DNA Reoviridae ds. RNA Poxviridae ds. DNA Iridoviridae ds. DNA Parvoviridae ss. DNA Picornaviridae ss. RNA Ascoviridae ds. DNA Polydnaviridae ds. DNA Rhabdoviridae ss. RNA Nodaviridae ss. RNA Rhabdoviridae ss. RNA NON-CLASSIFIED RNA VIRUSESs Divided genome ss. RNA Nodaurelia ss. RNA Kelply group ss. RNA 5 -virus group ss. RNA Minivirus ss. RNA Ovoid virases ss. RNA Drosophila X Virus ds. RNA ds= double-stranded, ss= single-stranded. s NUCLEOCAPSID SIMETRY OCCLUSION BODY Baciliform Isometric Ovoid Icosahedral Isometric Spherical Allantoid Ovoid Baciliform Icosahedral Baciliform + + + - Isometric Isometric Ovoid Isometric -

Klasifikasi Virus Entomopatogen v Klasifikasi virus entomopatogen dibuat sesuai dengan peraturan the International Commitee on Taxonomy of Viruses (ICTV). v Virus entomopatogen diklasifikasikan menjadi 12 famili. v Kriteria untuk menyusun klasifikasi disusun berdasarkan keragaman virus serangga, seperti: • Jenis bahan genetik (seperti single- atau double-stranded DNA, single- atau double-stranded RNA, positive atau negative strand) • Morfologi dan ukuran virion (seperti icosahedral, rodshaped, dll. ) • Kehadiran amplop di sekeliling virion. • Kehadiran occlusion body (OB) melingkupi virion. • Inang dan kisaran inang.

Klasifikasi Virus Entomopatogen v Kriteria utama sekuensing bahan genetik virus serangga bukan hanya menentukan diskriminasi antar spesies virus, akan tetapi juga membangun hubungan evolusioner antar virus dalam kelompok yang sama.

Penamaan Virus Entomopatogen § Virus entomopatogen diberi nama dengan cara singkatan (akronim). § Sesuai dengan inangnya, dan kelompok virusnya § Contoh: Autographa californica multiple nucleopolyhedrovirus Ac. MNPV § Semua nucleopolyhedroviruses dinamai NPV, granuloviruses dinamai GV, entomopoxviruses: EPV, iridoviruses: IV, dan cytoplasmic polyhedrosis viruses (cypoviruses) : CPV. § Virus entomopatogen sangat tinggi keragamannya, tapi hanya beberapa kelompok saja yang dijumpai pada populasi serangga dan menunjukkan potensi sebagai agen pengendali hayati.

Life Cycle of Insect Virus Generalized life-cycle of insect viruses. Figure credit: Jim Mc. Neil, Department of Entomology, Penn State University.

v Virus particles are usually found on the surface of plants or in the soil. v Insects become infected by consuming plant material with viral particles on the surface, although some pests of low-growing plants can be infected by contact with the soil. v Virus infection begins in the insect’s digestive system but spreads throughout the whole body of the host in fatal infections. v The body tissues of virus-killed insects are almost completely converted into virus particles. v The digestive system is among the last internal organ system to be destroyed, so the insects usually continue to feed until they die. Infected insects look normal until just prior to death, when they tend to darken in color and behave sluggishly. v They often develop more slowly than uninfected individuals.

v Most virus-infected insects die attached to the plant on which they feed. v Virus-killed insects break open and spill virus particles into the environment. These particles can infect new insect hosts. v Because of the destruction of the internal tissues, dead insects often have a “melted” appearance. v The contents of a dead insect can range from milky-white to dark brown or black. v While natural virus outbreaks tend to be localized, virus particles can be spread by the movement of infected insects, the movement of predators such as other insects or birds that come into contact with infected insects, or non-biological factors like water run-off, rain-splash or air-borne soil particles.

v Many virus-infected insects also climb to higher positions on their host plant before they die, which maximizes the spread of virus particles after the insect dies and disintegrates. v The number of virus infection cycles within a growing season depends heavily on the insect’s life cycle. v Insect pests with multiple generations per season or longer life cycles can be more heavily impacted by virus outbreaks since there is a greater opportunity for multiple virus infection cycles within a growing season.

Advantages and Disadvantages of Insect Viruses for Controlling Pests Advantages ØInsect viruses are unable to infect mammals, including humans, which makes them very safe to handle. Most insect viruses are relatively specific, so the risk of non-target effects on beneficial insects is very low. ØMany viruses occur naturally and may already be present in the environment. Even in cases where they are applied, successful infections can perpetuate the disease outbreak making repeat applications within a season unnecessary.

Disadvantages ØMost insect viruses take several days to kill their host insect, during which the pest is still causing damage. Insect death is also dose dependent, and very high doses are often necessary for adequate control. As insects age, they can become less susceptible to virus infection, so viruses are usually only effective against early larval life stages. ØAlthough viruses can persist in the environment for months or years, exposed virus particles, like those on the surface of plants, are quickly inactivated by direct sunlight or high temperatures, which can limit their persistence within a given season. Also, some agricultural practices can reduce persistence between seasons, such as tillage, which buries virus particles in the soil.

Suggestions for application of insect viruses v Viruses are usually not “stand alone” solutions to an insect pest problem, but are most effective in conjunction with other management strategies. v Insect viruses are fairly specific, be sure that the target pest is correctly identified. v Carefully scout fields before application and apply virus when the target pests are young but actively feeding.

v. Apply virus to maximize the longevity and effectiveness of virus particles: • Thoroughly coat plants to maximize coverage. Young plants can even be dipped in a solution of virus particles to completely cover the leaf area. • Apply in the morning or evening or on cloudy days when degradation from sunlight is reduced. • Avoid applying on rainy days, as rain will wash virus particles off the leaf surfaces. • Use formulations with ultraviolet (UV) light blockers and sticking agents to increase longevity. • Using mixed cropping and reduce soil disturbance after application. These help increase the persistence of virus particles in the system and may lead to better control within and between growing seasons.

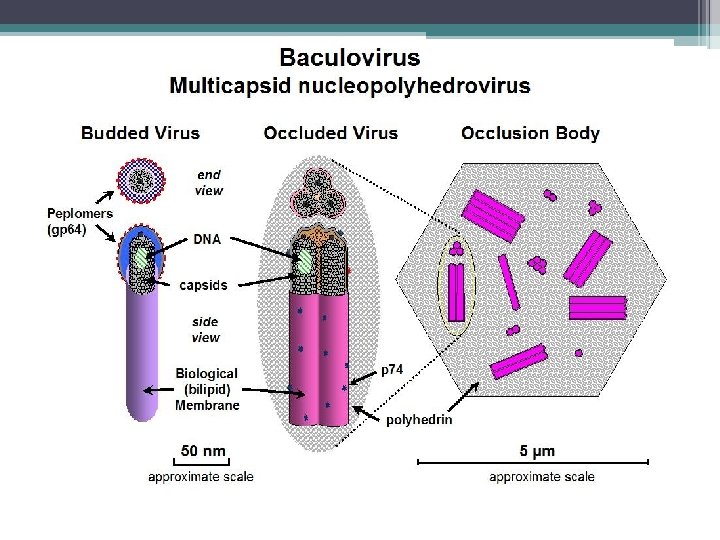
6 Baculoviruses Spodoptera littoralis 2 microns From Hunter-Fujita et al

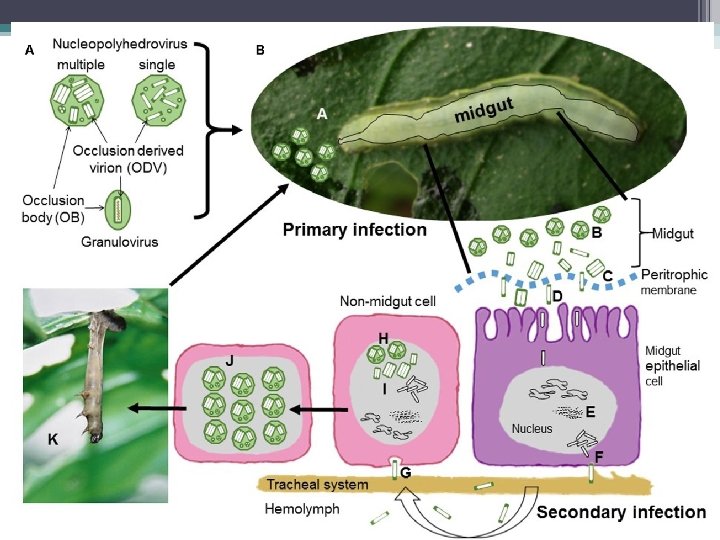
§ Kelompok Baculovirus yang penting sebagai virus entomopatogen adalah nucleopolyhedroviruses (NPV) and granuloviruses (GV). § Virus ini memiliki jenis inclusion bodies yang berbeda, di mana virus partikel (virion) menempel. § Parikel virus menyerang inti sel usus kecil, lemak tubuh atau sel-sel jaringan lainnya dan cairan tubuh serangga yang sudah mati. § Sebelum mati, larva yang terinfeksi memanjat bagian yang lebih tinggi dari tanaman, yang membantu diseminasi parikel virus dari tubuh larva yang sudah mati ke bagian tanaman yang lebih rendah. § Kebiasaan ini membantu menyebarkan virus untuk menginfeksi larva yag sehat.

§ Virus sangat spesifik inang dan dapat menyebabkan pengurangan populasi inang secara nyata. § Contoh virus yang dijual secara komersil: - Helicoverpa zea single-enveloped nucleopolyhedrovirus (Hz. SNVP) - Spodoptera exigua multi-enveloped nucleopolyhedrovirus (Se. MNPV) - Cydia pomonella granulovirus (Cp. GV).
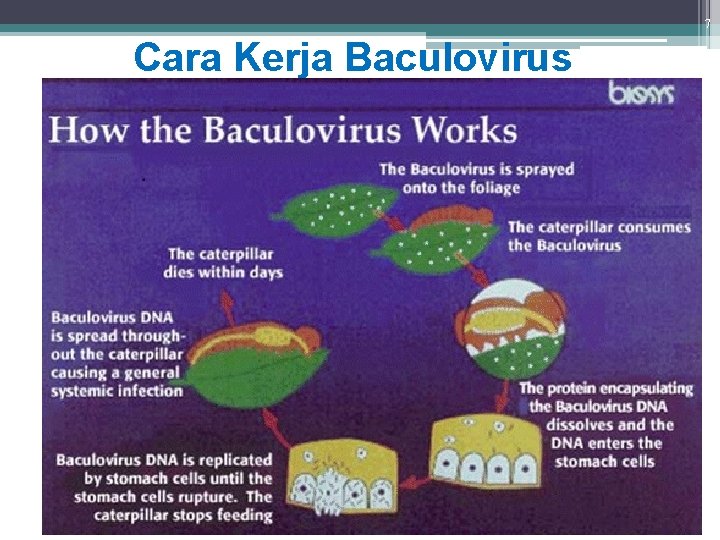
7 Cara Kerja Baculoviruses Mode of action From Hunter-Fujita et al

8 Baculoviruses Susceptibility of Alternative Hosts Found only in invertebrates No member of the family is known to infect plant or vertebrate Most have narrow host insect range, and infectivity is restricted to the original host genus or family

9 Baculoviruses Toxicity studies - mammals Toxicity test results from 1970 s/80 s of 29 NPVs indicated no toxicity or pathogenicity. Doses were generally 10 – 100 x the “per acre” (1 acre = 0. 45 ha) field rate equated to a 70 kg person. Heliothis zea NPV most extensively tested for toxicity in humans and led to registration of “Elcar” by Sandoz in USA.

10 Baculoviruses Toxicity studies – mammals cont. No effects of Hz. NPV found in: Acute toxicity-pathogenicity tests in mouse, rat, guinea pig, rabbit, monkey and man at 6 x 109 – 3 x 1012 OB / kg. Skin irritation sensitivity tests in guinea pigs, rabbits and man at 106 and 107 OB / mm 2 skin. Eye irritation tests in rabbits with 105 and 2 x 106 OB / eye Subacute toxicity-pathogenicity tests and subcutaneous injection into mice, rats, dogs and rhesus monkeys. Teratogenicity and carcenogenicity studies in rats and mice at 109 – 3. 5 x 1012 OB / kg. Similar but less extensive results for many other NPVs from the 1970 s/80 s

11 Baculoviruses Toxicity studies – wildlife Birds Able to pass NPV through the alimentary tract unaffected No deleterious effects Aquatic organisms No adverse effects Beneficial insects No direct effect on parasitoids, predators and pollinators Indirect effects on parasitoids resulting from host death

12 Baculoviruses Pathology studies Toxicity tests designed for testing effects of chemicals on vertebrates are insufficient Results reported in Gröner (1986) indicate no virus induced antibody production in test mammals and chicken. No cytogenetic effects of baculoviruses in mammalian cells either in vivo or in vitro.

13 Baculoviruses Virus-cell interactions in vitro Ac. NPV inoculated into vertebrate cells can be takenup and the degree of up-take depends on cell type, temperature, time and viral phenotype. BUT, none of the human and nonhuman vertebrate lines tested showed evidence of viral replication. NPVs unable to activate retroviruses in mammalian cell lines

Baculoviridae: Granulosis viruses (GV)

15 Baculoviruses a list of the baculoviruses regulated as pesticide active ingredients by the US EPA Office of Pesticide Programs as of May 2005 Anagrapha falcifera NPV Cydia pomonella GV Douglas fir tussock moth NPV Gypsy moth NPV Helicoverpa zea NPV Indian meal moth GV Mamestra configurata NPV (pending) Spodoptera exigua NPV

16 Baculoviruses – US EPA fact sheet III. ASSESSING RISKS TO HUMAN HEALTH These viruses infect only the target insect larvae and closely related species. Toxicity tests show that the viruses pose no risk to the public. Workers wear protective clothing to prevent possible irritation from handling and applying the product. IV. ASSESSING RISKS TO THE ENVIRONMENT Tests show that the GV and NPVs that EPA has registered as pesticide active ingredients specifically infect only certain species of moth larvae. The viruses do not harm other organisms, including plants, beneficial insects, other wildlife, or the environment. These viruses occur naturally in their insect hosts.

17 Cypoviruses: Mode of action Polyhedra ingested and dissolved in larval midgut Virions released and attach to midgut columnar cells Viral core enters cell cytoplasm RNA transcription and replication RNA occluded in capsules Virus capsules occluded by virogenic stroma to form occlusion bodies

18 Cypoviruses (Rheoviridae) No CPV has been found infecting vertebrates or plants (Belloncik, 1989) Dendrolimus spectabilis CPV registered in Japan in 1974. Safety test results generally negative. – Katagiri, K. (1981) Pest control by cytoplasmic polyhedrosos viruses. In: Microbial control of pests and plant diseases 1970 -1980. (Ed Burges, H. D. ) Academic Press.

Poxviridae: Entomopoxviruses NPV GV GV NPV

§ Member of the family of Poxviridae has a wide host, including vertebrates and invertebrates. §Chicken pox and Small pox virus belong to this family. § The show allantoid – to brick-shaped virions, occluded within ovoid OBs called Spheroids. § Entomopoxvirus has been isolated from 27 orthopterans, lepidopterans, dipterans and coleopterans. § The subfamily Poxvirinae includes three genera, i. e. Entomopoxvirus A, Entomopoxvirus B, and Entomopoxvirus C § Entomopoxvirus A infects only coleopteran species; Entomopoxvirus B infects lepidopteran and coleopteran species; Entomopoxvirus C infects only dipteran species. § The fourth group, group D, has been proposed by ICTV which only attacks hymenopterans.

Poxviridae: Entomopoxvirus Anomala cuprea (Coleoptera) larvae infected with an entomopoxvirus show the symptoms of the infection such as a whitish appearance and underdevelopment (left, infected larva; right, healthy one).
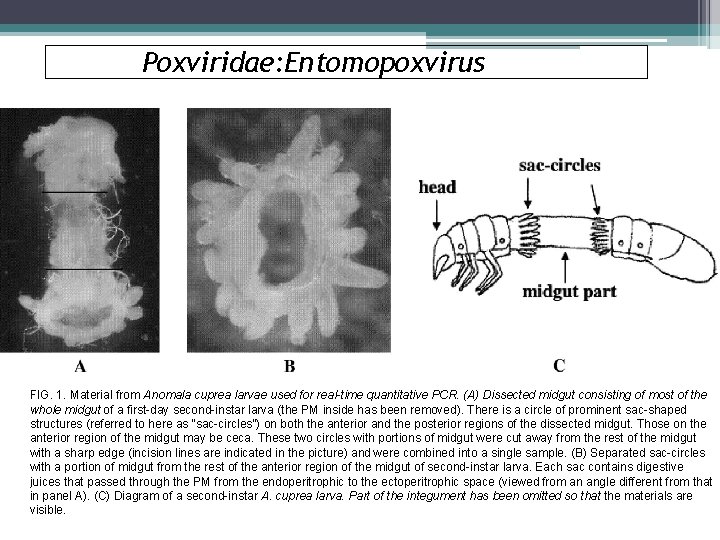
Poxviridae: Entomopoxvirus FIG. 1. Material from Anomala cuprea larvae used for real-time quantitative PCR. (A) Dissected midgut consisting of most of the whole midgut of a first-day second-instar larva (the PM inside has been removed). There is a circle of prominent sac-shaped structures (referred to here as “sac-circles”) on both the anterior and the posterior regions of the dissected midgut. Those on the anterior region of the midgut may be ceca. These two circles with portions of midgut were cut away from the rest of the midgut with a sharp edge (incision lines are indicated in the picture) and were combined into a single sample. (B) Separated sac-circles with a portion of midgut from the rest of the anterior region of the midgut of second-instar larva. Each sac contains digestive juices that passed through the PM from the endoperitrophic to the ectoperitrophic space (viewed from an angle different from that in panel A). (C) Diagram of a second-instar A. cuprea larva. Part of the integument has been omitted so that the materials are visible.

Ascoviridae: Ascovirus § Members of the family of Ascoviridae are double stranded DNA (ds. DNA) viruses that infect lepidopteran insects and cause the unique pathology of forming virion containing vesicles in the hemolymph of infected hosts. § The presence of the vesicles gives the hemolymph a milky white appearance, which is a major characteristic of the disease. § A few species of Ascovirus has been isolated only from insects, specifically from Lepidopterans (Noctuidae). § Enveloped virions of ascoviruses are bacilliform, ovoid or allantoid in shape, and occluded within vesicle-like OBs.

An ascovirus-infected caterpillar
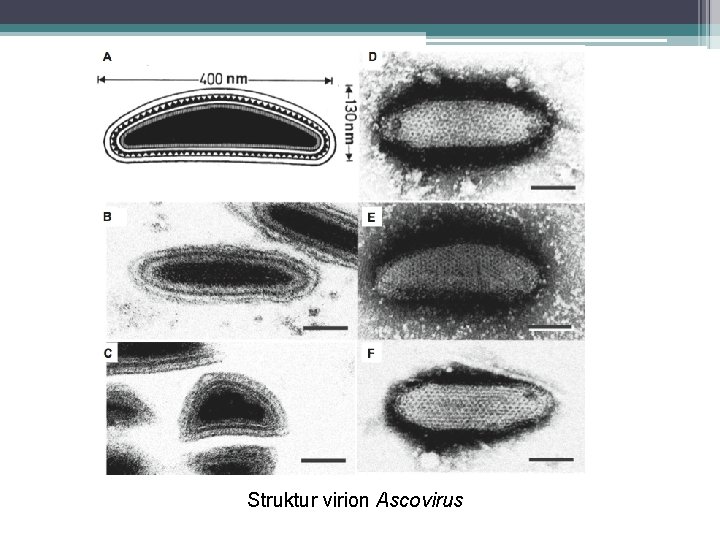
Struktur virion Ascovirus

Ascoviridae: Ascovirus Lifecycle Ø Ascovirus establishes in the Helicoverpa population through spring-summer. Ø The disease is transmitted from caterpillar to caterpillar by wasps (such as Microplitis). Ø Ascovirus could be transmitted directly from one caterpillar to another by spitting - for example, when caterpillars encounter each other on the plant. Ø When ascovirus particles enter the caterpillar´s body they multiply in tissue cells, eventually infecting the haemolymph (blood). This causes the haemolymph to change from clear to milky. The caterpillar stops eating, but may not die for several days or weeks, surviving in a lethargic state.

Ascoviridae: Ascovirus symptoms § In cases where a Microplitis wasp has both parasitised a caterpillar and infected it with ascovirus, the symptoms seen are those of the disease rather than of the parasitoid. When ascovirus kills the caterpillar, it also kills the developing Microplitis larva. § Caterpillars infected with ascovirus will generally stop eating within two days. They stop growing, but can live for weeks in a lethargic state before they die. § The blood of an ascovirus-infected caterpillar is white and creamy, whereas the blood of a healthy caterpillar is clear. Blood colour gives the best diagnosis in the laboratory and can be tested by splitting or pricking the caterpillar.

Iridoviridae: Iridovirus § Invertebrate Iridescent Viruses (IIVs) (family Iridoviridae) are known to infect a number of agricultural pests, medically important insect vectors, and terrestrial isopods that live in damp or aquatic habitats. §The major characteristic of this family is the presence of iridescent blue, green, orange, or purple coloration in heavily infected individuals. § The small iridovirus tend to display colors from violet to turquoise. § The viral structure is non-enveloped, non-occluded, isocahedral viral particles.

Iridoviridae: Iridovirus § Although some iridoviruses infect frogs and fishes, those infecting insects belong to two genera: Iridovirus, whose viral particles fluctuate between 120 to 130 nm in size. § They mostly infect arthropods, particularly insects, in damp or aquatic habitats worldwide (see complete list of invertebrate hosts. § They are highly infectious by injection but have low infectivity by ingestion. § Horizontal transmission can occur by cannibalism or predation of patently infected individuals, or the virus may even be vectored by nematodes and parasitoid wasps that introduce viral particles into the host insect during the act of penetration or oviposition.
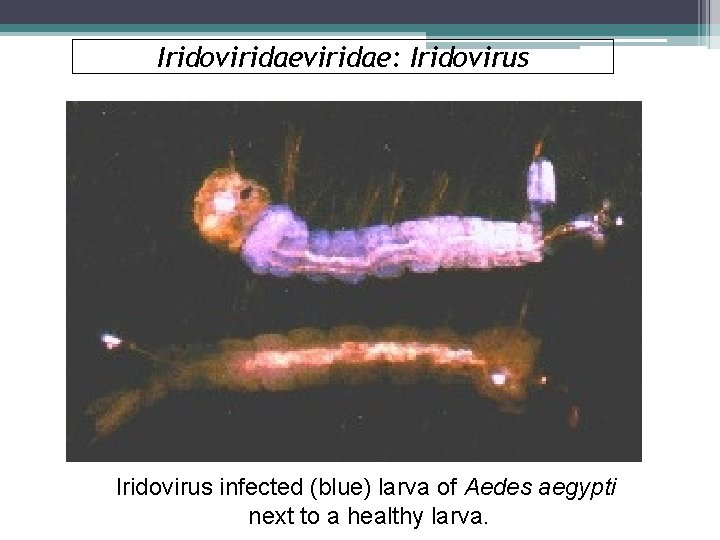
Iridoviridae: Iridovirus infected (blue) larva of Aedes aegypti next to a healthy larva.

Polydnaviridae § Polydnaviridae only infects endoparasitic Hymenoptera. § Member of this family show non-occluded, ovoid virions, containing multipartite ds. DNA § ICTV recognizes two genera within this family, including Ichneovirus and Bracovirus.

Polydnaviruses are symbiotic proviruses of some ichneumonid and braconid wasps that modify the physiology, growth and development of host lepidopteran larvae. Polydnavirus infection targets neuroendocrine and immune systems, altering behavior, stunting growth, and immobilizing immune responses to wasp eggs and larvae. Polydnavirus-mediated disruption of cellular and humoral immunity renders parasitized lepidopteran larvae suitable for development of wasp larvae as well as more susceptible to opportunistic infections. Evidence from the Campoletis sonorensis polydnavirus system indicates that the unique genomic organization of polydnaviruses may have evolved to amplify the synthesis of immunosuppressive viral proteins. Immunosuppressive viruses have been essential to elucidating vertebrate immunity. Polydnaviruses have similar potential to clarify insect immune responses and may also provide novel insights into the role of insect immunity in shaping polydnavirus genomes.
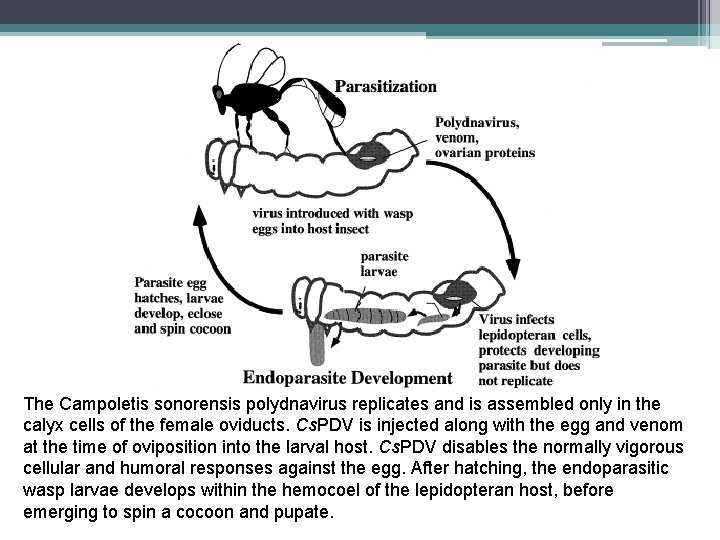
The Campoletis sonorensis polydnavirus replicates and is assembled only in the calyx cells of the female oviducts. Cs. PDV is injected along with the egg and venom at the time of oviposition into the larval host. Cs. PDV disables the normally vigorous cellular and humoral responses against the egg. After hatching, the endoparasitic wasp larvae develops within the hemocoel of the lepidopteran host, before emerging to spin a cocoon and pupate.
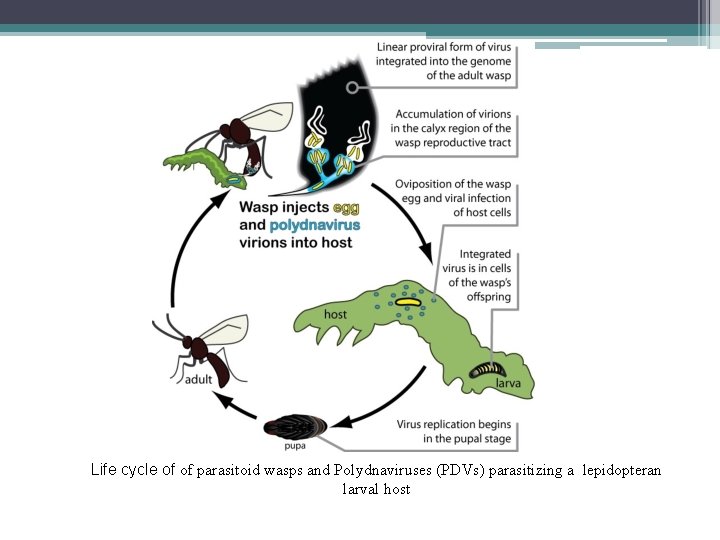
Life cycle of of parasitoid wasps and Polydnaviruses (PDVs) parasitizing a lepidopteran larval host
TERIMA KASIH
- Slides: 65