Virtual Company Supply Chain Challenges of CMO Based

Virtual Company & Supply Chain – Challenges of CMO Based on “The complexity and challenges of the management of global product release scenarios” - Dissertation submitted to Trinity College Dublin for the degree of Masters in Pharmaceutical Manufacturing Technology By Deirdre Dunne, B. Sc. (Applied Science) QP Forum 12 April 2018 TCD School of Pharmacy & Pharmaceutical Sciences

Committed to improving patients’ lives by identifying, developing and commercializing meaningful products that address unmet medical needs 2
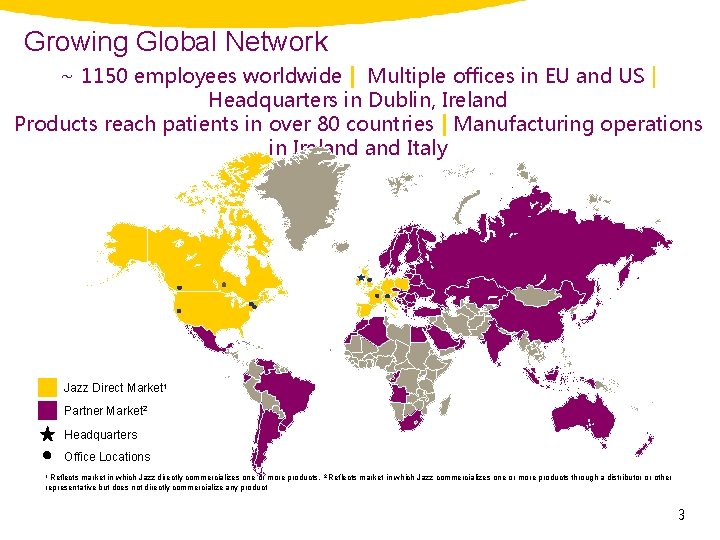
Growing Global Network ~ 1150 employees worldwide | Multiple offices in EU and US | Headquarters in Dublin, Ireland Products reach patients in over 80 countries | Manufacturing operations in Ireland Italy Jazz Direct Market 1 Partner Market 2 Headquarters Office Locations 1 Reflects market in which Jazz directly commercializes one or more products, 2 Reflects market in which Jazz commercializes one or more products through a distributor or other representative but does not directly commercialize any product 3

Focus for 2015 QP Forum “The Challenges with managing CMOs” • Due diligence and assessment of Quality Culture • Supplier qualification and audit program to evaluate robustness of CMO QMS • Risk based approach to developing level of oversight • Quality Technical Agreement and Ongoing monitoring 4

Focus for 2018 QP Forum Virtual Company & Supply Chain – Challenges of CMO oversight • Increased Regulatory focus on Sponsor and CMO Relationship • Challenges in External Manufacturing • Management of significant c. GMP compliance problems (is there a “warning system”? ) • Workshop 5

Increased Regulatory focus on Sponsor and CMO relationship • At the Parenteral Drug Association (PDA)/FDA Regulatory Conference in September 2017, Washington. Francis Goodwin Division Director CDER FDA presented ‘CDER Regulatory Perspective Compliance and Enforcement trends’. He particularly addressed warning letters, import alerts and regulatory discretion activities. Contract Manufacturing Top Concerns Data Integrity Barbara Unger Consulting Inc. Data Integrity & Your Contract Manufacturer – Common Pitfalls to Avoid. Outsourced Pharma, 03 rd November, 2017. 6

Number of warning letters issued by FDA in 2017 The question could be asked by the Regulators why these issues were not highlighted earlier by the Sponsors who contracted the services of these facilities. The requirements are clear in respect to oversight of contract facilities, is this a failure in the Sponsors supplier management program? 13 warning letters from 2017 referenced Contract Operations Reflects 16% of the 69 warning letters issued in 2017 to API and drug product manufacturing sites FDA Data Dashboard Warning letters and Responses 7

Importance of Regulatory Intelligence Evaluation of Health Authority inspections or other enforcement actions • Monitoring warning letters alone is not sufficient. Firms should seek to obtain form 483 s from all FDA Inspections, including sites of the contract manufacturer where they do not have product. Frequently, problems are systematic and evaluation of all inspections conducted at the firm’s sites can provide an early warning that something is amiss. • Firms should monitor FDA import alerts and recalls. • Firms should be informed of all health agency inspections of the contract site and be informed of their findings, outcomes, and recommendations. Barbara Unger Consulting Inc. Best Practices for Data Integrity Oversight at your Contract Manufacturer. Bioprocess Online , 04 th December, 2017. 8

Data Integrity EU GMP Chapter 7 / EMA guidance • Data Integrity requirements should be incorporated into the company’s contractor/vendor qualification/assurance program and associated procedures • In addition to having their own data governance systems, companies outsourcing activities should verify the adequacy of comparable systems at the contract acceptor. The contract acceptor should apply equivalent levels of control to those applied by the contract giver. • Formal assessment of the contract acceptor’s competency and compliance in this regard should be conducted in the first instance prior to the approval of a contractor, and therefore verified on a periodic basis at an appropriate frequency based on risk. Barbara Unger Consulting Inc. Best Practices for Data Integrity Oversight at your Contract Manufacturer. Bioprocess Online , 04 th December, 2017. 9

Data Integrity How can the sponsor make lot release decisions with confidence based on data from their contracted partner? • Effective on-site evaluation of contract manufacture (initial and ongoing) • On site auditing by auditors with significant expertise or training and experience of how health authorities are currently evaluating data governance within the GMP system • Identify and justify information required to support a lot release decision – which data and why • Create secure means of data transmission between CMO and Sponsor • QTA Barbara Unger Consulting Inc. Best Practices for Data Integrity Oversight at your Contract Manufacturer. Bioprocess Online , 04 th December, 2017. . 10

Management of change is critical to ensure MA compliance in a virtual company like Jazz Both parties must ensure they are aware of their responsibilities Jazz must ensure compliance with the Marketing Authorisation is maintained at the contract facility 11

Jazz Quality Oversight of CMOs Change control Supplier Qualification CMO is qualified as per Jazz Pharmaceuticals’ supplier management program Quality Agreement Quality Technical Agreement defines the management of changes Written Procedures CMO is required to have established written procedures for control of changes that include Jazz notification, Jazz approval of changes, follow-up, and closure of changes as they relate to manufacture, testing, storage, assembly, labelling and distribution of the Product Notification of Changes Jazz Review CMO notifies Jazz in accordance with QTA and written procedures Jazz has the opportunity to acknowledge and/or approve changes made to procedures, processes, materials, including the specifications thereof, used in the execution of Services which may impact upon the quality of the Product prior to the execution of those changes The CMO reviews and/or approves changes made to any procedures that impact on the assembly, labelling or distribution of the Product

Challenges in External Manufacturing 13

Challenges for Jazz We produce small volume biopharmaceutical products • As a small volume biopharmaceutical company we compete with big pharma companies for slots in the production schedule Many of our products have Orphan status • Due to this status many of our products are single sourced and moving the process to an alternative manufacturer if there are performance issues could bring many complex challenges e. g. Transferability of the manufacturing process We contract out manufacturing • We do not have direct access to production • We are competing with other customers 14

Challenges for Jazz Mergers and acquisitions • Jazz like many pharmaceutical companies acquires products and their associated manufacturers through company mergers and acquisitions. Therefore these manufacturers need to be integrated into the Jazz supplier management system using quality risk management CMOs spread globally • Jazz manufacturers are spread across the globe, the majority of CMOs are based in the US. The remainder are located across Europe and Japan. This requires management of different quality cultures and time-zones based on geographic locations Complex Supply chains • Jazz has complex supply chains. We have several suppliers involved in the various stages of manufacture, testing, storage and distribution. 15

Communication can be a challenge for a virtual Company • Can be hard to maintain cohesion in virtual teams • Co-located teams working in multiple time zones • Need to establish clear processes for communication • On how people react during virtual meetings – rules for good virtual meeting habits • Dealing with different quality cultures and time-zones • Jazz has clearly defined team support roles – ‘virtual plant teams’ for each product that ‘mirrors’ the structure of teams in a manufacturing plant QA QC Supply Chain Technical Support Project Manager Tips for virtual teams Chapter 7. Build your Team ‘The First 90 Days’ by Michael 16 D. Watkins
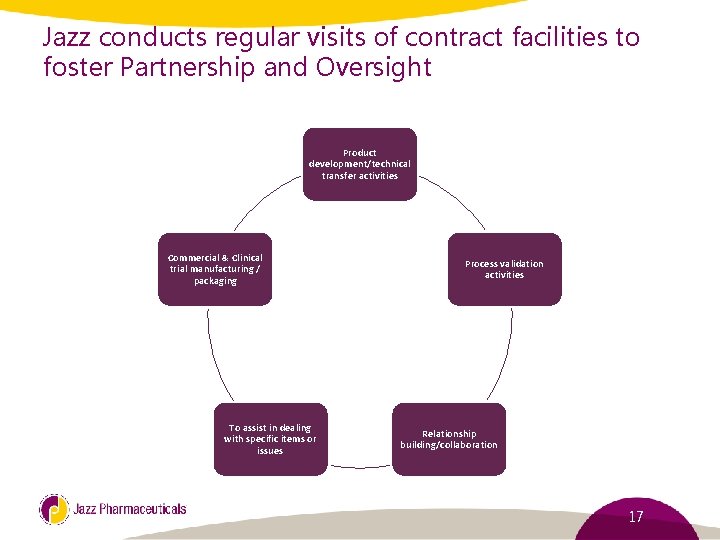
Jazz conducts regular visits of contract facilities to foster Partnership and Oversight Product development/technical transfer activities Commercial & Clinical trial manufacturing / packaging To assist in dealing with specific items or issues Process validation activities Relationship building/collaboration 17

Management of significant c. GMP compliance problems (is there a “warning system”? ) 18

Jazz monitors Suppliers for compliance by three main activities Quality Agreement Auditing Communication • Establishing a formal Quality Technical Agreement (QTA) that delineates GMP responsibilities, including quality activities between contractor giver and acceptor • By routinely auditing the contractor’s facilities against Jazz and international GMP standards • Communication is critical and is achieved by way of regular quality to quality meetings, business review meetings and partnership meetings 19

Establishing a formal Quality Technical Agreement (QTA) One of the most important documents used in contract manufacturing between the MAH and the contract facility This agreement is even more important in the virtual contract manufacturing world that Jazz operates in Ensure a standard Jazz QTA template is approach and used for establishing consistency is taken to quality agreements with delineate quality new suppliers responsibilities between both parties 20

Establishing a formal Quality Technical Agreement (QTA) • Training and awareness of a new quality agreement or changes to a quality agreement is a key activity as part of the supplier oversight program. • • QTAs are stored in an central electronic location for cross functional access; to ensure deliverables within the agreement are understood. Non conformance to the Quality Agreement between Jazz and contract facility is monitored and trended in the deviation management system. • A specific drop down code is selected in the deviation system for an event related to the quality agreement. This is used as a tool used for trending conformance to the QTA within the PQS. 21

Limitations of the QTA? Limits Priorities • There are limitations of what can be covered in a quality agreement • Focus should be made on other parts of the relationship that is not covered in a quality agreement • Understanding requirements, good communication, collaboration and open/honesty, timeliness are some of the common priorities found between both parties • Having a good working relationship is key to contract manufacturing arrangements Relationships • Building relationships through regular face to face meetings supports building trust and collaboration between Jazz and our suppliers Deirdre Dunne (2016). The Complexity and Challenges of Management of Global Product Release Scenarios – Quality Overs 5 Dec 2016 22

Auditing the contractor’s facilities Auditing of Jazz suppliers is key to the supplier oversight program The extent and frequency of the audit for a particular supplier is determined based on the criticality of the material/service Jazz routinely enlists the support of external, ex. Inspectorate auditors and SMEs to perform audits of our critical suppliers Having extensive GMP expertise or training in how Inspectorates are currently evaluating data within the PQS of our contract facilities e. g. Data Integrity reviews It is acknowledged by the Regulators that Audits cannot cover all products and processes and provides a limited subset of data FDA, US Food and Drug Administration (2015) Request for Quality Metrics, Guidance for the Industry draft, July 2015. 23

Assessing the Quality Culture of the CMO PDA Quality Culture Survey report suggested that certain characteristics are driving good behaviours This is a useful tool that can be incorporated in audit programs Top five quality attributes that can serve as substitutes for quality culture Management communication that quality is everyone’s responsibility Site has formal quality improvement objectives and targets Clear performance criteria for feedback and coaching Quality topics included in at least half of all-hands meetings Collecting error prevention metrics Pritesh Patel, Denyse Baker, Rick Burdick, et al. (2015) Quality Culture Survey Report. PDA Journal of Pharmaceutical Science and Technology 2015, 69 631 -642. September-October 2015. 24

Maintenance and monitoring 25
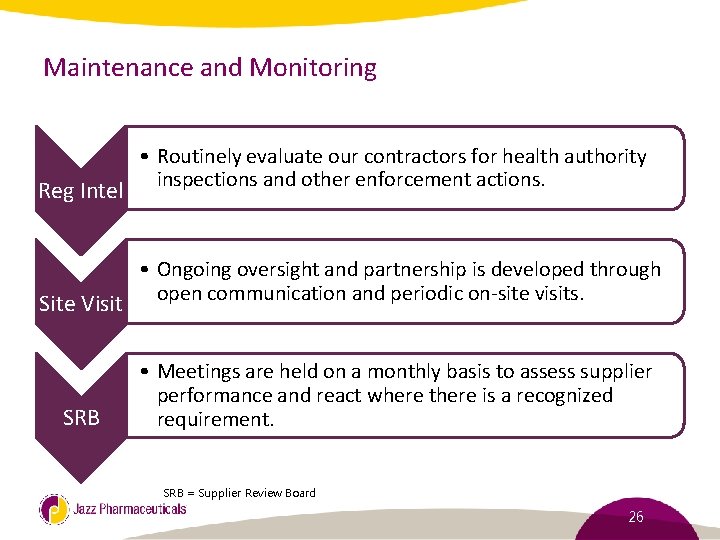
Maintenance and Monitoring Reg Intel Site Visit SRB • Routinely evaluate our contractors for health authority inspections and other enforcement actions. • Ongoing oversight and partnership is developed through open communication and periodic on-site visits. • Meetings are held on a monthly basis to assess supplier performance and react where there is a recognized requirement. SRB = Supplier Review Board 26

Maintenance and Monitoring Metrics • Generation of metrics to review trends and key items relating to the products manufactured by our approved suppliers, which include a CMO scorecard system. Escalation • Any adverse trends noted during the compilation of this report are communicated to Senior Management through the Management Review process and Quality Council. 27

CMO Scorecards • We utilise CMO scorecards as a tool to support supplier performance management. • If the scorecard flags red we know there is an issue. • The score card attributes are built on the requirements set out in the quality agreement e. g. stability reporting on time, notification of changes, batch acceptance rate. 28

Person in Plant • • • • Benefits • Validation campaign presence • Campaign-based manufacturing • Faster visibility to issues • Immediate decision making and approvals • Liaison between sponsor and CMO departments • Can build a strong long term partnership with CMO Risks • May not make sense for low volume products • Does not prevent deviation events (reactive) • On site presence may not result in quicker visibility of issues • Must have authority to make product related decisions • Must involve QA at sponsor Barbara Unger Consulting Inc. Best Practices for Data Integrity Oversight at your Contract Manufacturer. Bioprocess Online , 04 th December, 2017. 29

Workshop Discussion • How big of a challenge is it for the MA holder to keep actual activities in the CMO and Suppliers aligned with registrations? • For virtual company what approaches have others taken to data integrity? • What are key Competencies of Person-In-Plant? • Management of primary packaging component deficiency leading to parenteral finished product inspection action limit excursion • The value of regulatory surveillance as an indicator of CMO performance 30
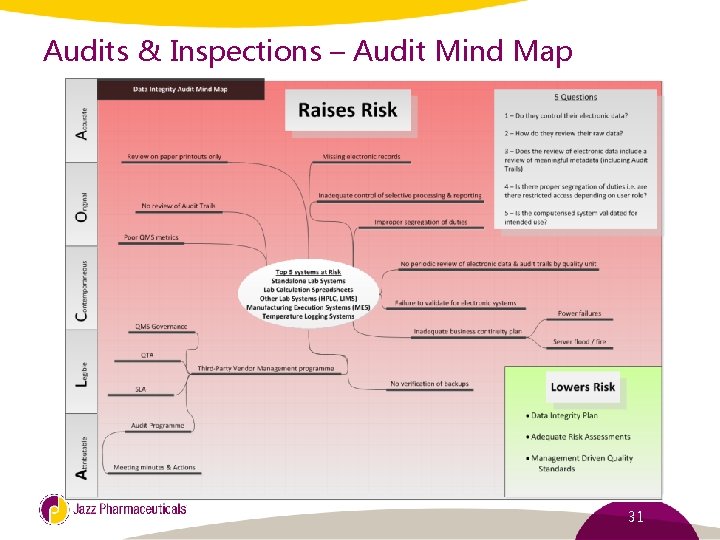
Audits & Inspections – Audit Mind Map 31

Person in Plant – Key Competencies • • • Effectiveness depends on skills and experience of individual –cultural fit with CMO –credentials and technical expertise –ability to influence without authority –trusted by sponsor and CMO –ability to build and balance relationships with sponsor and CMO –ability to facilitate resolution of issues –has authority to make decisions on behalf of sponsor –know which side of the fence they are on Barbara Unger Consulting Inc. Best Practices for Data Integrity Oversight at your Contract Manufacturer. Bioprocess Online , 04 th December, 2017. 32
- Slides: 32