Viral Latency in Bacteriophage and HIV1 Leor Weinberger

Viral Latency in Bacteriophage and HIV-1 Leor Weinberger

Outline 1. Review phage lysis/lysogeny • phage lysis/lysogeny decision as a stochastic switch 2. HIV-1 proviral latency • • • Review of HIV-1 and T cell lifecycles Eric Verdin’s work HIV-1 proviral latency as a bi-stable stochastic switch 3. Therapeutic implications (harnessing latency) • Designing a gene therapy to prevent AIDS but maintain HIV-1

Phage Lambda Life Cycle infection E coli lytic path lysogenic path lysis/lysogeny decision induction lysogenic cell Percent lysogens depends on • Multiplicity of infection • Cell nutrition lysis
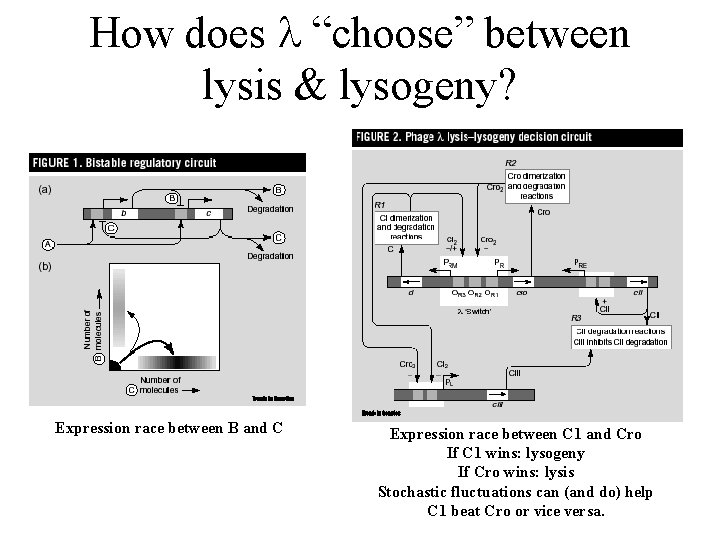
How does “choose” between lysis & lysogeny? Expression race between B and C Expression race between C 1 and Cro If C 1 wins: lysogeny If Cro wins: lysis Stochastic fluctuations can (and do) help C 1 beat Cro or vice versa.

HIV-1 latency • 2 things are called latency in HIV-1 infection • Clincial latency (aka asymptomatic or Stage II infection) this isn’t really viral latency • Post-integration or proviral latency this is real viral latency • Does post-integration latency in HIV-1 arise inpart due to a stochastic component analogous to lysis/lysogeny in bacteriophage ?

HIV-1 budding from the cell surface
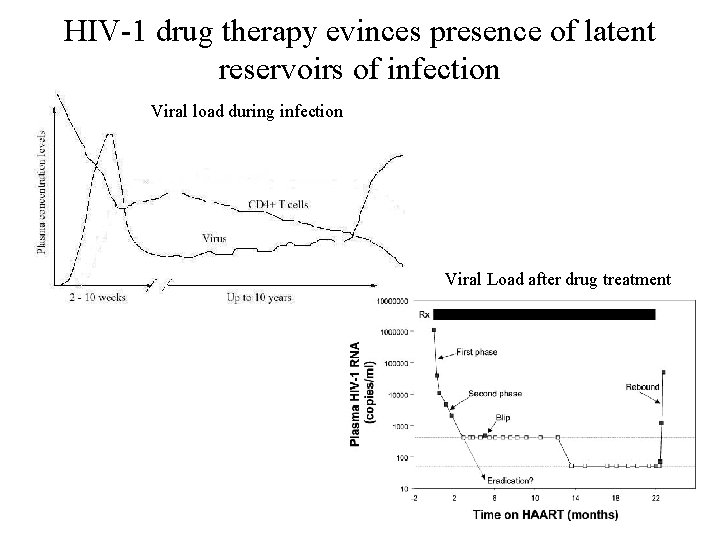
HIV-1 drug therapy evinces presence of latent reservoirs of infection Viral load during infection Viral Load after drug treatment

Why does viral load rebound after prolonged HAART therapy?
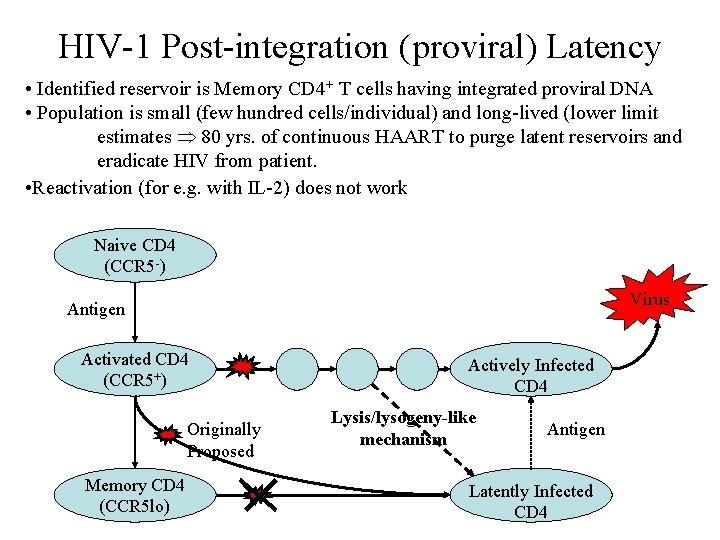
HIV-1 Post-integration (proviral) Latency • Identified reservoir is Memory CD 4+ T cells having integrated proviral DNA • Population is small (few hundred cells/individual) and long-lived (lower limit estimates 80 yrs. of continuous HAART to purge latent reservoirs and eradicate HIV from patient. • Reactivation (for e. g. with IL-2) does not work Naive CD 4 (CCR 5 -) Virus Antigen Activated CD 4 (CCR 5+) Originally Proposed Memory CD 4 (CCR 5 lo) Actively Infected CD 4 Lysis/lysogeny-like mechanism Antigen Latently Infected CD 4

HIV-1 latency • 2 things are called latency in HIV-1 infection • Clincial latency (aka asymptomatic or Stage II infection) this isn’t really viral latency • Post-integration or proviral latency this is real viral latency • Does post-integration latency in HIV-1 arise in-part due to a stochastic component analogous to lysis/lysogeny in bacteriophage ?
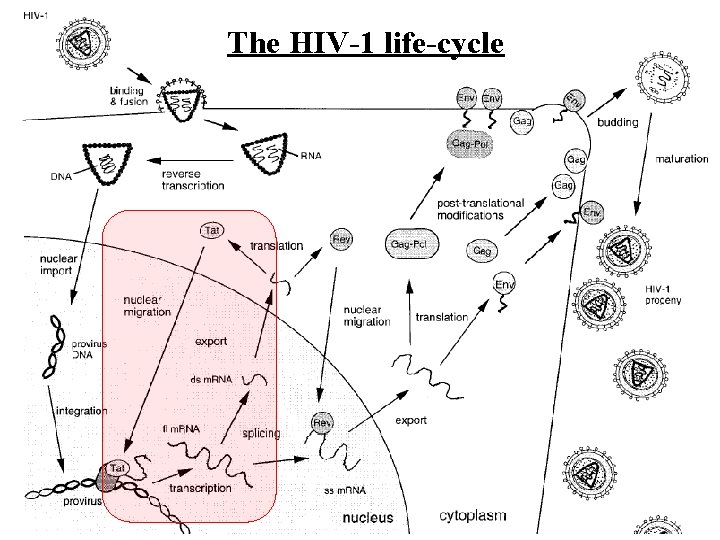
The HIV-1 life-cycle
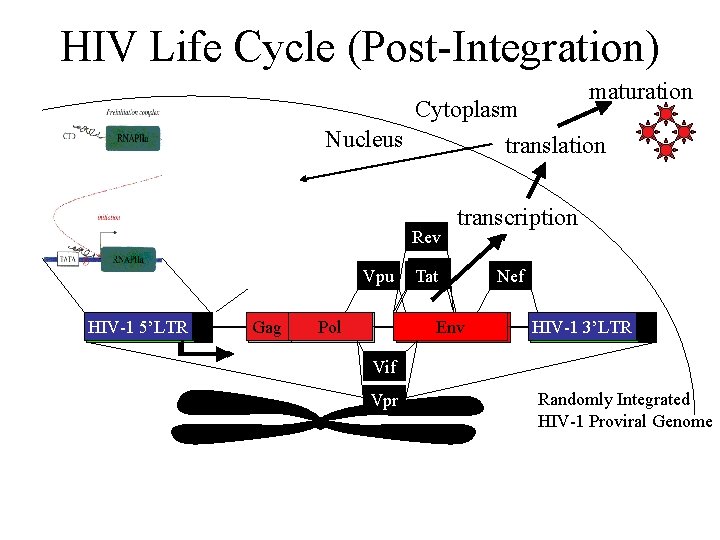
HIV Life Cycle (Post-Integration) maturation Cytoplasm Nucleus translation Rev Vpu HIV-1 5’LTR Gag Pol transcription Tat Env Nef HIV-1 3’LTR Vif Vpr Randomly Integrated HIV-1 Proviral Genome
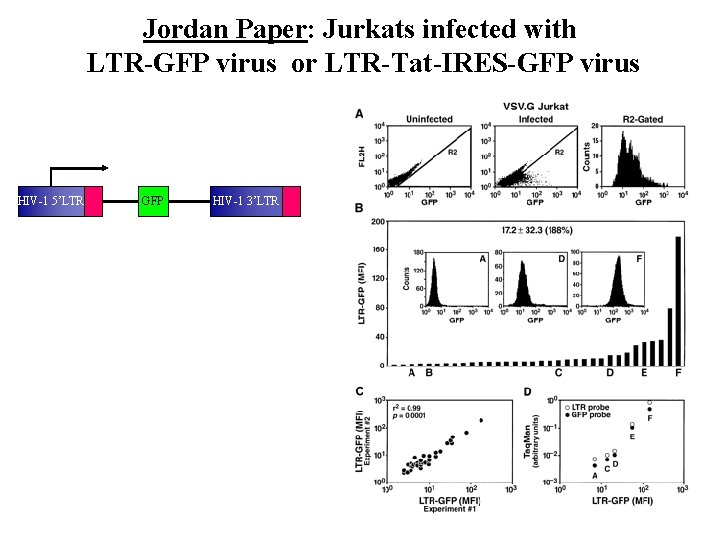
Jordan Paper: Jurkats infected with LTR-GFP virus or LTR-Tat-IRES-GFP virus HIV-1 5’LTR GFP HIV-1 3’LTR
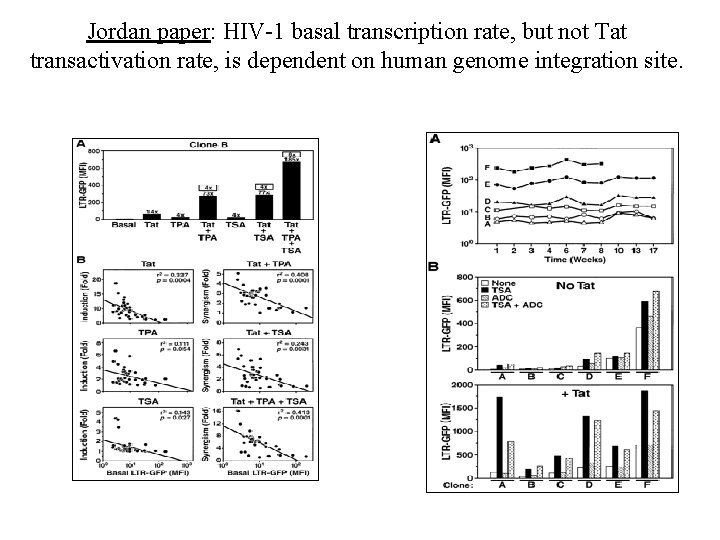
Jordan paper: HIV-1 basal transcription rate, but not Tat transactivation rate, is dependent on human genome integration site.
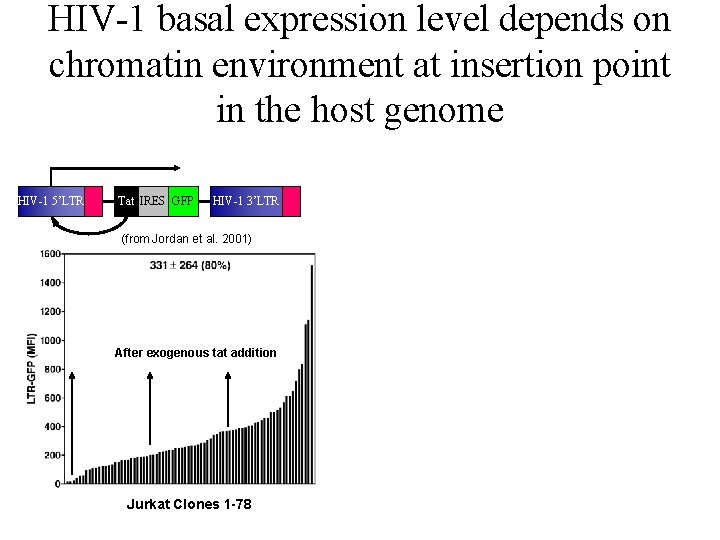
HIV-1 basal expression level depends on chromatin environment at insertion point in the host genome HIV-1 5’LTR Tat IRES GFP HIV-1 3’LTR (from Jordan et al. 2001) After exogenous tat addition Jurkat Clones 1 -78
![Possible HIV-1 transcriptional states RNAPII [1] – Basal transcription HIV-1 5’LTR Tat GFP IRES Possible HIV-1 transcriptional states RNAPII [1] – Basal transcription HIV-1 5’LTR Tat GFP IRES](http://slidetodoc.com/presentation_image_h2/4728e8ae9a329fe04847a544c6473953/image-17.jpg)
Possible HIV-1 transcriptional states RNAPII [1] – Basal transcription HIV-1 5’LTR Tat GFP IRES “closed” chromatin HIV-1 3’LTR degradation [2] - Transactivation HIV-1 5’LTR RNAPII GFP IRES Tat HIV-1 3’LTR “open” chromatin
HIV-1 basal expression level depends on chromatin environment at insertion point in the host genome HIV-1 5’LTR Tat IRES GFP HIV-1 3’LTR (from Jordan et al. 2001) After exogenous tat addition Jurkat Clones 1 -78 HIV-1 5’LTR GFP IRES Tat HIV-1 3’LTR
HIV-1 5’LTR GFP IRES Tat HIV-1 3’LTR
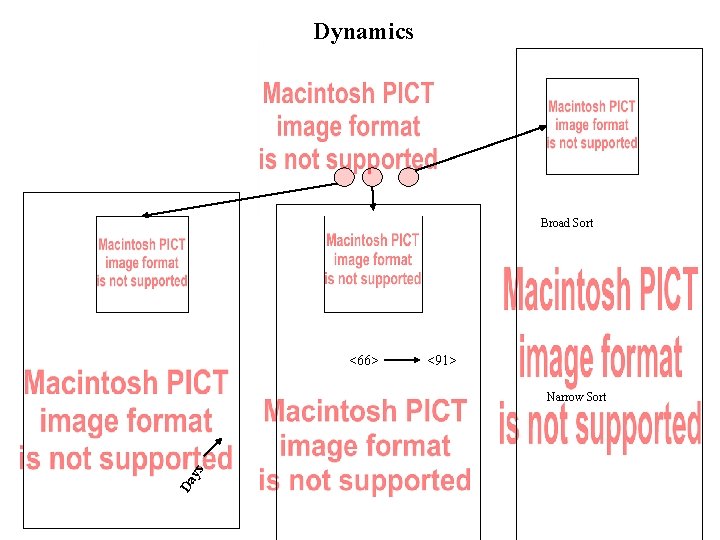
Dynamics Broad Sort <66> <91> Da ys Narrow Sort
Bulk Sort Clonal Sorts:

DIM clones can be activated to express high levels of GFP LGIT clone After 17 hr incubation with TNF- Why do cells turn off ? ? ?

7 SK (sn. RNA) introduction leads to increased transactivation not decreased!!! Cyclin T 1 CDK 9 Tat p. TEF-b complex 7 SK sn. RNA Proposed “inhibitor” • Bulk Bright sort (LGIT infection) • PIII-7 SK-CMV-Neo infection of LGIT infected cells. (Neo selected) (infection with control virus PIII-GFP-CMV-Neo, and Neo selection produced no observable change in bright cells)

Control: Extracellular Tat is not responsible for switching Tat Tat HIV-1 5’LTR Tat Heparan Sulfate Tat GFP IRES Tat HIV-1 3’LTR
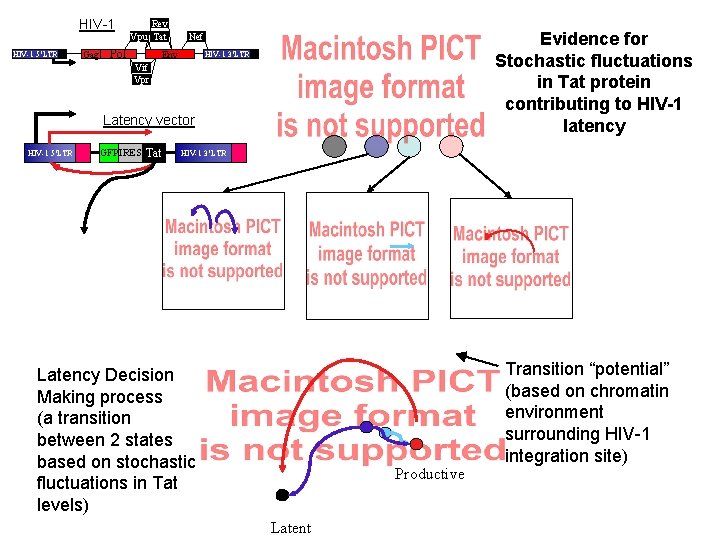
HIV-1 5’LTR Gag Rev Vpu Tat Pol Evidence for Stochastic fluctuations in Tat protein contributing to HIV-1 latency Nef Env HIV-1 3’LTR Vif Vpr Latency vector HIV-1 5’LTR GFP IRES Tat HIV-1 3’LTR Latency Decision Making process (a transition between 2 states based on stochastic fluctuations in Tat levels) Productive Latent Transition “potential” (based on chromatin environment surrounding HIV-1 integration site)

Control: LTR-GFP clones are stable (No “switching” in absence of Tat) Uninfected Jurkats LTR-GFP infection 9 different LTR GFP clones

Implications • HIV-1 may use this “stochastic decision” to help it evade an aggressive immune response. • It may possible to harness this “decision making process” and nudge the virus one way or the other.

Conclusions • The arisal of HIV-1 proviral latency appears to have a stochastic component dependent upon integration site in the human genome and independent of cell state (I. e. cellular factors) • 2 examples of viral latency mediated at the transcriptional level (HIV-1 and phage )

Computational Design of an HIV trafficked Gene Therapy Vector to Prevent AIDS but maintain HIV-1 Infection Leor Weinberger UNIV. OF CALIFORNIA BERKELEY LABS OF ADAM ARKIN (Bioengineering) AND DAVE SCHAFFER (Chemical Engineering)

BACKGROUND • What is AIDS? • Actual clinical syndrome when immune system breaks down • CD 4+ T cell count < 200 cells L & Opp. Infections present • How does HIV cause AIDS? • CD 4+ T cell killing and eventual exhaustion of T cell replenishment machinery (currently most accepted of 3 theors. ) • What is the treatment? • HAART (Highly Active Anti-Retroviral Therapy) a. k. a. tripledrug cocktails reduce viral production and increase CD 4 count. • HAART decreases the rate of CD 4+ cell turnover. • HAART delays progression to AIDS.

PROBLEMS WITH HAART • Serious side-effects • • Expensive • • • $15, 000/year for treatment Majority of 40 million infected live in 3 rd world. Viral resistance • • Many patients can’t tolerate many of the 15 available drugs Liver, Spleen, Pancreatic damage Fat redistribution (humpback condition) arises on average after 18 months of therapy Latent reservoirs of infection • • • Exist latent reservoirs of infection, resting cells not actively producing virus and thus not effected by HAART Estimates 50 -60 yrs. of continuous HAART to purge latent reservoirs and eradicate HIV from patient. Reactivation with IL-2 did not work
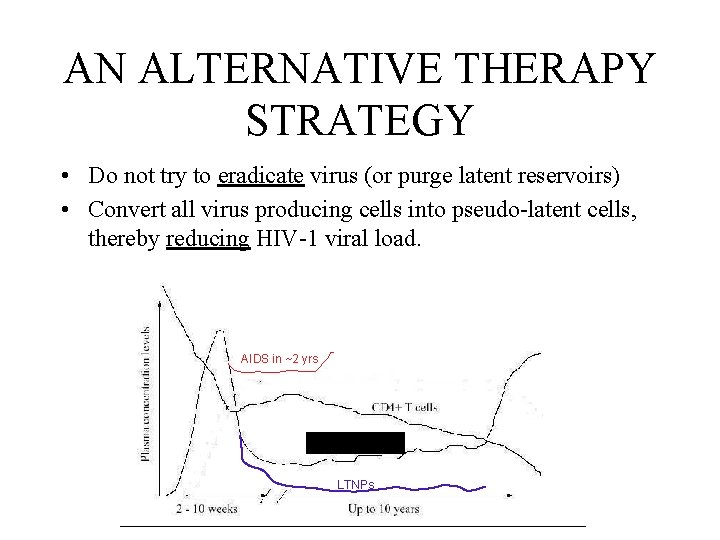
AN ALTERNATIVE THERAPY STRATEGY • Do not try to eradicate virus (or purge latent reservoirs) • Convert all virus producing cells into pseudo-latent cells, thereby reducing HIV-1 viral load. AIDS in ~2 yrs AIDS in ~10 yrs LTNPs
Is it possible to create a therapeutic cr. HIV-1 vector HIV Life HIV-1 Cycle (Post-Integration) cr. HIV-1 Gene Therapy Vector that reduces viral load & persists with maturation HIV-1 in vivo? Cytoplasm Nucleus translation transcription Tat HIV-1 5’LTR Gag Pol Env HIV-1 3’LTR Randomly Integrated HIV-1 Proviral Genome HIV-1 5’LTR D P HIV-1 3’LTR
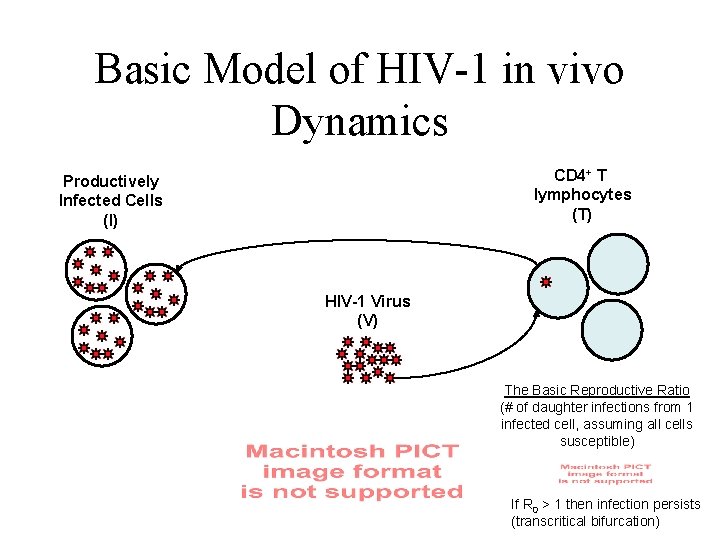
Basic Model of HIV-1 in vivo Dynamics CD 4+ T lymphocytes (T) Productively Infected Cells (I) HIV-1 Virus (V) The Basic Reproductive Ratio (# of daughter infections from 1 infected cell, assuming all cells susceptible) If R 0 > 1 then infection persists (transcritical bifurcation)
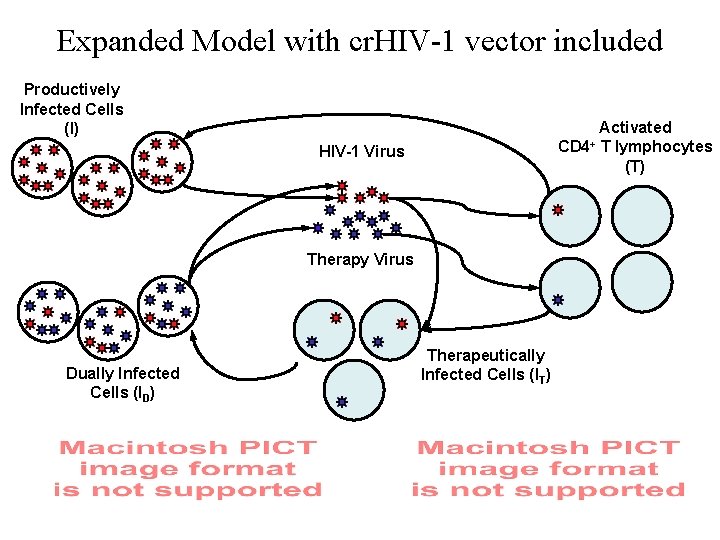
Expanded Model with cr. HIV-1 vector included Productively Infected Cells (I) CD 4+ HIV-1 Virus Therapy Virus Dually Infected Cells (ID) Therapeutically Infected Cells (IT) Activated T lymphocytes (T)

HIV-1 Dynamics after hypothetical administration of cr. HIV-1 vector (engineered with P = 20 and various D values)
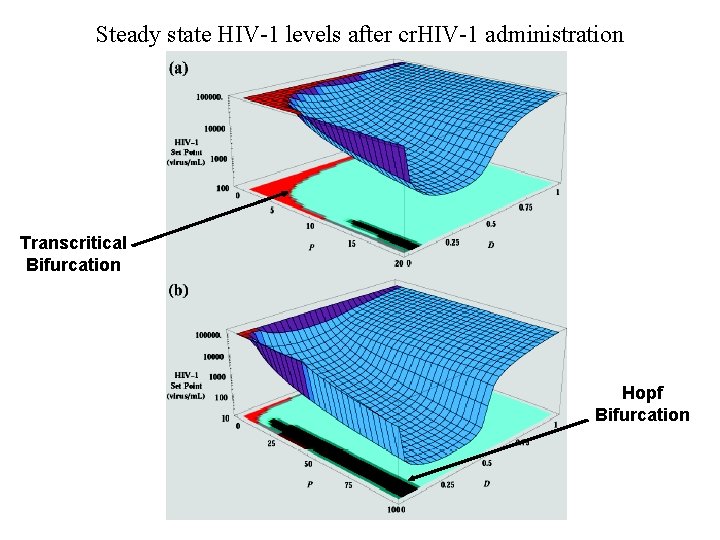
Steady state HIV-1 levels after cr. HIV-1 administration Transcritical Bifurcation Hopf Bifurcation
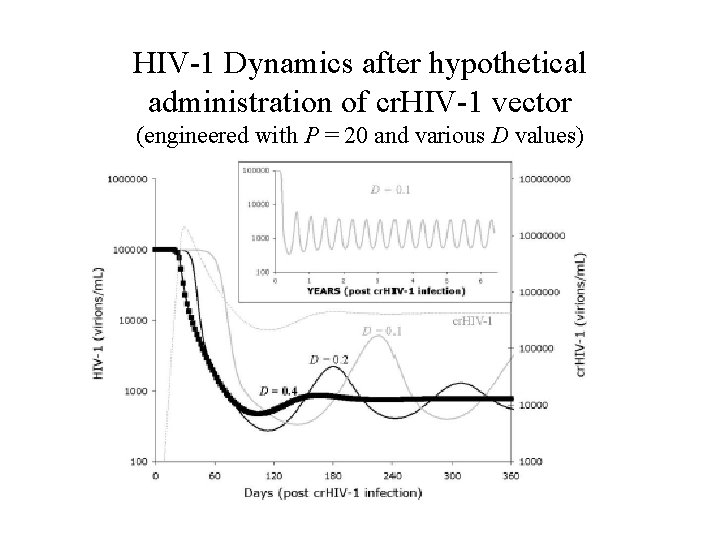
HIV-1 Dynamics after hypothetical administration of cr. HIV-1 vector (engineered with P = 20 and various D values)

A simple equation for designing cr. HIV-1 vectors that persist in vivo The Basic Reproductive Ratio : R 0 T Definition: The # of secondary cr. HIV-1 infections, obtained from a single initial cr. HIV-1 infection, assuming • virtually all cells are uninfected by cr. HIV-1. • The basic model system (T, I, V) is at steady state. If R 0 T > 1, then cr. HIV-1 infection persists.

Discussion 1. The model gives a quantitative analysis of diverse gene therapy approaches; the analysis is independent of the specific of anti-viral (downregulating) gene used. 2. There does exist a parameter regime where a cr. HIV-1 therapeutic vector can persist along with HIV-1. A simple function R 0 T can be used to design cr. HIV-1 vectors that persist in vivo. 3. cr. HIV-1 can achieve a 3 log decrease in HIV-1 set point when packaging efficiency is 20 fold greater than HIV-1. 4. Not necessarily “better” to have a very efficient inhibitor of HIV-1 production.

Mutational Escape HIV-1 has a very high mutation rate (1 in 3 103 -104) because reverse transcriptase has no proofreading mechanism (recent evidence suggests HIV-1 is also able to utilize hyper-mutation). HIV-1 mutates towards highest fitness (I. e. fastest replication rate, thus highest viral load) Since cr. HIV-1 is a virus, it will also mutate in order to increase its fitness (virl load) Two types of viral escape mutants exist: • HIV-1 can mutate so that it is no longer susceptible to cr. HIV-1 • cr. HIV-1 can mutate so that it is no longer therapeutic Any mutation HIV-1 makes to decrease cr. HIV-1 fitness (I. e. viral load) will cause cr. HIV-1 to make a reciprocating mutation to increase its own fitness. We can attempt to minimize the HIV-1 mutational escape from cr. HIV-1 therapy by designing the cr. HIV-1 therapeutic gene so that it targets a cell factor (that HIV-1 depends on) rather than targetting HIV-1 directly. Examples include targetting CDK 9/Cyclin. T 1 or cellular furin.
Fitness landscapes Where cr. HIV-1 mutate towards? -Highest viral load, D = 1. 0 Since this corresponds to HIV -1 rebound, cr. HIV-1 will tend to mutate away from being therapeutic.

Fitness Landscapes If we modify the Basic Model to consider logistic growth of T cells: Then, even though cr. HIV-1 still mutates towards D = 1. 0 HIV-1 viral load never reaches its pre-therapy set point, And some therapeutic effect will always be retained.

Conspirators • • • Adam Arkin Dave Schaffer (& the entire Schaffer Lab) John Burnett (Grad student, Chem. Eng. ) Jared Toettcher (undergrad, Bioenginnering) Nory Cabanilla (summer student, 2002) Collaborators • Eric Verdin (UCSF, Gladstone) • Jasper Rine (MCB) • Qiang Zhou (MCB)
- Slides: 44