VIRAL HEPATITIS Table 1 Characteristics of hepatitis viruses

VIRAL HEPATITIS
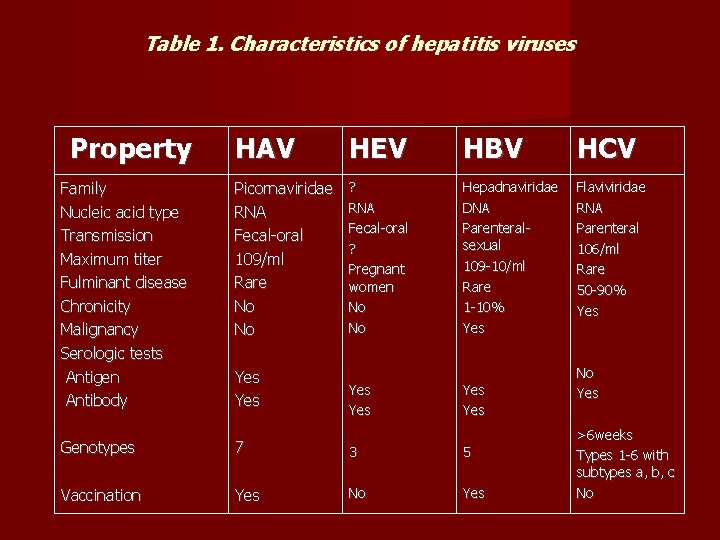
Table 1. Characteristics of hepatitis viruses Property Family Nucleic acid type Transmission Maximum titer Fulminant disease Chronicity Malignancy Serologic tests Antigen Antibody HAV HEV HBV HCV Picornaviridae RNA Fecal-oral 109/ml Rare No No ? RNA Fecal-oral ? Pregnant women No No Hepadnaviridae DNA Parenteralsexual 109 -10/ml Rare 1 -10% Yes Flaviviridae RNA Parenteral 106/ml Rare 50 -90% Yes Yes Genotypes 7 3 5 Vaccination Yes No Yes >6 weeks Types 1 -6 with subtypes a, b, c No

HEPATITIS A Hepatitis A virus (HAV) is a member of picornaviridae family (which includes poliovirus and rhinoviruses, agents of the common cold). The virus particle - is of 27 nm in diameter - is not enveloped - has an outer capsid which surrounds a single-stranded RNA

Risk factors for acquisition: contact with acutely ill individuals travel to endemic areas homosexual activity contact with children attending day care centers illicit drug use Among the persons under 18 years old, 50% in France, 90% in Portugal and >95% in Romania present antibodies against HAV.

Pathogenesis During acute HAV infection, there is an initial viremia and fecal shedding of viruses; viremia persists for up to 3 weeks. Histopathological aspects include the existence of: hepatocellular necrosis centrilobular cholestasis periportal infiltration of the liver with mononuclear inflammatory cells

Clinical manifestations Only 10% of A-hepatitis are clinically manifested. The incubation period: the symptoms of HA usually appear about 4 weeks after exposure, but the interval may range from 2 -6 wks. The clinical manifestations are different in adults and young children (children of less than 2 years old experience asymptomatic infection or develop other symptoms than those suggestive for hepatic inflammation.

A. Prodromal (preicteric) phase lasts for 4 -10 days Gastrointestinal symptoms: anorexia, nausea and vomiting, right upper-quadrant pain, diarrhea (in children – 20% of cases ), distaste for cigarettes (in smokers only); Influenza-like - syndrome: fever /chills, muscle pain, headache; Rheumatic syndrome: arthralgias (usually disappear after onset of jaundice); Eruptions: petechiae, measles/scarlet fever-like exanthema; Fatigability; Pruritus; Jaundice Acute abdomen (mimicking an appendicitis) could be very variable:

B. Icteric phase – is ushered by the appearance of dark, golden-brown urine due to bilirubinuria, followed one to several days later by pale stools yellowish coloration of the mucous membranes, conjunctivae, sclerae and skin. The physical examination 2 special forms of evolution may be encountered: Cholestatic hepatitis A - >12 weeks of jaundice Relapsing hepatitis A

Diagnostic 1. AST (Aspartate Aminotransferase) or SGOT (Serum Glutamic. Oxalacetic Transaminase) (N = 5 – 54 U/L). 2. ALT (Alanine Aminotransferase) or SGPT (Serum Glutamic-Piruvic Transaminase): (N = 0 – 36 U/L). 3. Alkaline Phosphatase (N = 40 – 120 U/L). 4. GGT (Gamma Glutamyltransferase) (N = 3 – 59 U/L). 5. LDH (Lactose De. Hydrogenase) (N = 135 – 225 U/L). . 6. Total bilirubin 7. Direct bilirubin (conjugated) (N = 0 – 5 μmol/L). 8. Albumin (N = 35 – 50 g/L)9. PT (Prothrombin Coagulation Time) (N = 10 – 12 sec. ) or QUICK Index (N≥ 80%). 10. Urine tests: bilirubin appears in the urine in the prodromal phase and may lead to the diagnosis 11. Peripheral blood: 12. Serological confirmation is always required, by the specific detection of Ig. M anti HAV.

Complications Neurological complication: encephalitis, meningoencephalitis, Guillain-Barre syndrome, mononeuritis affecting cranial or peripheral nerves. Myocardial involvement: myocarditis and electrocardiographic changes Pancreatitis Pleural effusions and ascites Aplastic anemia

Differential diagnosis of jaundise I Abnormalities of bilirubin metabolism 1. Increased bilirubin production (unconjugated bilirubinemia): • Hemolysis • Ineffective erythropoiesis: megaloblastic anemias, myeloproliferative diseases, protoporphyria etc • Blood transfusions • Resorbtion of haematomas 2. Decreased hepatic clearence: a. decreased uptake and/or conjugation of bilirubin • Gilbert syndrome, Crigler Najjar syndrome • Rifampin • Physiologic jaundice of neonate b. Decreased canalicular excretion of bilirubin: Dubin-Johnson and Rotor syndromes

II. Hepatocellular jaundice 1. Acute/subacute hepatocellular damage • Acute hepatitis: viral (CMV, Epstein-Barr virus, herpes), bacterial (leptospirosis, brucellosis, staphylococcal, gram-negative bacillary sepsis, tuberculosis, salmonellosis), amoebiasis, giardiasis • Hepato-toxines: alcohol, Amanita, acethaminophen; • Drugs: isoniazide, macrolides, sulfonamides, metildopa, valproic acid, paracetamol, non steroidal anti-inflammatory drugs, halothane and derivatives, antidepressants, anti-HIV, ketoconazole • Chemical agents: carbon tetrachloride, tetrachloroethylene • Metabolic disorders: Wilson’s disease, Reye’s syndrome • During pregnancy: acute liver steatosis 2. Chronic hepatocellular diseases □Chronic active hepatitis □ Cirrhosis □ Neoplasia □ Neonatal hepatitis □ Autoimmune hepatitis □ Alfa 1 -antitripsin deficiency

III. Biliary obstruction Extrahepatic • Gallstones • Neoplasia • Sclerosing colangitis • Biliary stricture • Pancreatitis • Parasites Intrahepatic • Cholestatic viral hepatitis • Postoperative cholestasis • Benign recurrent cholestasis • Primary biliary cirrhosis • Chronic inflammatory bowel disease • Biliary atresia

Treatment The aim of the treatment is to maintain comfort and nutritional balance. Bed rest is indicated. A high-calorie, high-protein diet has been shown to reduce the length of time in hospital. The adequate calories must be provide: 30 -35 kcal/kg, mostly from carbohydrates. Low-fat diet is frequently advised. Avoidance of alcohol is recommended for some months after acute infection. Patients with severe anorexia, nausea or vomiting occasionally need nutritional support. Meat can be introduced when urine exam for bilirubin and urobilinogen are back to normal. In cholestatic hepatitis hypertonic glucose is used.

Prevention Now, there is available a double vaccine against A and B hepatitis viruses (Twinrix)

HEPATITIS B The hepatitis B virus (HBV), discovered in 1966, infects more than 400 million people world-wide.

Epidemiology Areas with high prevalence Areas with low level of endemicity The distribution of hepatitis B infection varies greatly throughout the world.

Modes of trasmission Contact transmission through body secretions: semen, vaginal secretions, blood, saliva Maternal-neonatal transmission: 5 – 10 % of neonatal HBV infections result from in utero infection Percutaneous transmission: drug abusers, health care workers Blood transfusions.

Virologic characteristics HBV belongs to Hepadnaviruses (DNA-woodchuck hepatitis virus, the duck hepatitis virus and other avian and mammalian variants). The hole VIRION – the Dane particle is a 42 nm sphere, which contains: □ the core enclosing the DNA and □ an envelope material , which is found abundantly in the circulation, shaped as small spheres and rods with an average width of 22 nm. The viral genome is represented by a double – stranded circular DNA of approximately 3200 pairs of bases that encodes 4 overlapping open reading frames: 1. S, for the surface or envelope gene 2. C, for the core gene 3. X, for the X gene 4. P, for the polymerase gene

HBV replication cycle The replication cycle of HBV begins with attachment of the virion to the hepatocyte. Inside the hepatocyte nucleus the viral genome is converted into a covalently closed circular DNA (ccc. DNA). The ccc. DNA is

Serologic makers of importance in HBV infection Hepatitis B core antigen (HBc. Ag) - represents the nucleocapsid that Hepatitis B ″e″ antigen( HBe. Ag). It is a circulating peptide derived from DNA polymerase (encoded by the P gene) – also serves as a witness of Antibodies against HBs. Ag (anti – HBs) are produced as a response to Antibodies against HBc. Ag (HBc. Ab) encloses the viral DNA. the core gene and then modified and exported off the liver cells and serves as a maker of active viral replication. reserve transcriptase activity. the presence of the envelope (surface) Ag: • are detected virtually in all patients who have ever been exposed to HBV: they are not protective • Ig. M subtype antibodies are associated with acute infection; usually they disappear within 4 -8 months after acute infection. Antibodies against HBe. Ag (Hbe. Ab) appear once the antigen has been cleared

Immune response to acute HBV infection Recovery from acute HBV infection is dependent on: □ B cell response with production of antibodies to pre-S and S antigens □ T cell response. Immune response to chronic HBV infection In patients with chronic HBV infection the HBV-specific CTL response is weak and limited to one or a few epitopes. Immune tolerance in perinatally acquired HBV infection Immune tolerance to HBV is manifested by: □ A high rate of progression to chronic infection □ Lack of disease activity in the presence of very high level of virus □ Very low rate of spontaneous or interferon-induced HBe. Ag seroconversion.

Pathogenesis 1. Immune-mediated liver injury HBV-related liver injury is related to: • CTL-mediated lysis of infected hepatocytes; • Non-cytolytic pathways via the release of cytokines. 2. Direct cytopatic effects HBV is in general not a cytopatic virus. Direct cytopathic liver injury can occur when the viral load is very high (as in fibrosing cholestatic hepatitis). Hepatocarcinogenesis Life cycle of HBV in the human host □ 95% of infected neonates become asymptomatic chronic HBV carriers □ 30% of children of 3 months – 6 years become asymptomatic chronic HBV carriers □ only 3 – 5% of adults become asymptomatic chronic HBV carriers.
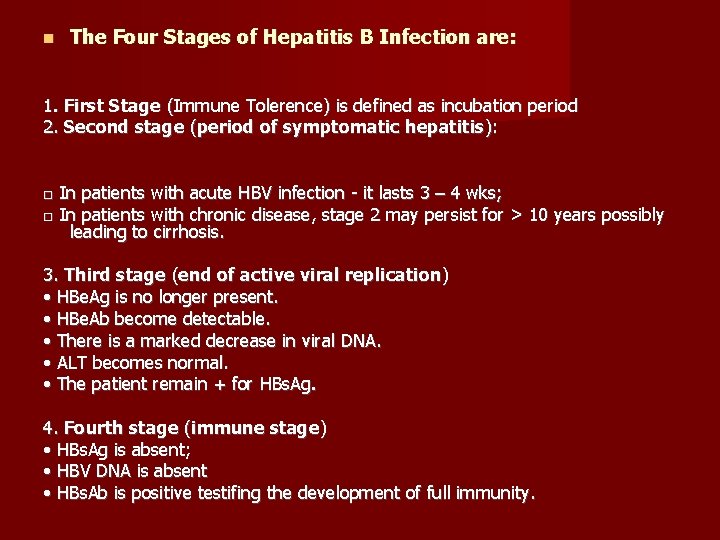
The Four Stages of Hepatitis B Infection are: 1. First Stage (Immune Tolerence) is defined as incubation period 2. Second stage (period of symptomatic hepatitis): □ In patients with acute HBV infection - it lasts 3 – 4 wks; □ In patients with chronic disease, stage 2 may persist for > 10 years possibly leading to cirrhosis. 3. Third stage (end of active viral replication) • HBe. Ag is no longer present. • HBe. Ab become detectable. • There is a marked decrease in viral DNA. • ALT becomes normal. • The patient remain + for HBs. Ag. 4. Fourth stage (immune stage) • HBs. Ag is absent; • HBV DNA is absent • HBs. Ab is positive testifing the development of full immunity.

Some factors can affect the evolution through the four stages, like: • the genetic predisposition of the host, • the presence of other viruses, • the treatment with immunosuppressive agents, sex, • infections with HBV mutants.

HBV variants HBV mutant that are detected clinically confer survival advantage over the wild type virus by: • evading host immune response • enhancing virus replication. Mutations in the precore, S and P genes have been most extensively studied. Precore mutants S mutants Mutations in the HBV gene have been reported in infants born to carrier mother who developed HBV infection despite vaccination, and in liver transplant recipients who developed HBV reinfection despite prophylaxis with hepatitis B immune globuline (HBIG).

Clinical manifestations I. Asymptomatic hepatitis A. Subclinical infection: abnormal blood tests, without jaundice and symptoms; B. Inapparent infection: no symptoms or biochemical abnormalities are present, but serologic tests are positive II. Symptomatic hepatitis A. Anicteric hepatitis – B. Icteric hepatitis Icteric phase: - dark urine, lightening of stool color – a few days before onset of jaundice, - as jaundice progress the constitutional symptoms become less severe, duration of jaundice is 1 – 2 months after its onset. The physical findings are similar to those described in HAV, adenopathy, small spider angiomata.

Extrahepatic manifestations of viral hepatitis • transient serum-sickness like syndrome: • polyarteritis nodosa: • glomerulonephritis • mixed cryoglobulinemia • acrodermatitis of childhood (Gianotti’s disease): .

Laboratory features Blood chemistry was shown above. Minor elevation of ALT values may persist for a few months, even if the bilirubin level turned normal. Serologic Tests • HBs. Ag is the first identifiable marker (before the level of ALT start to Increase) by ELISA, RIA. It’s persistence beyond 6 months suggests the development of HBV carrier state. • HBe. Ag it will be detectable in the next days to weeks after the appearance of HBs. Ag and in uncomplicated HBV infections it will disappear priory to the HBs. Ag.

• HBe. Ab become detectable shortly after the disappearance of HBe. Ag. They may persist for long periods. • DNA- HBV: is detectable during the early phase of infection and → undetectable several wks later. • HBc. Ab (anti - HBc) are detectable in serum shortly after the HBs. Ag′s discovery and before the appearance of anti-HBs. • Ig. M anti – HBc peak in several weeks from the onset of infection; is the most sensitive test for acute HBV infection. They persist for 4 to 8 months after their appearance. • Ig. G anti – HBc are synthetised in late convalescence and are found thereafter for years, but levels decline slowly. • Anti – HBs. As the titer of HBs. Ag declines, corresponding antibodies become detectable and reach a peak level in a few months. They are neutralizing,

Evolution of HBV infection 1. Full recovery 2. Fulminant hepatitis 3. Persistence of HBs. Ag 4. Chronic hepatitis B 5. Cirrhosis 6. Hepatocellular carcinoma 1. Full recovery is assumed if the ALT reaches normal or close to normal levels and if HBs. Ag becomes negative in EIA. This takes more than 6 months in some cases. 2. Fulminant hepatic failure (FHF) was defined as a potentially reversible condition, the consequence of severe liver injury, in which encephalopathy developed within 8 weeks of the appearance of the first symptoms, in the absence of pre-existing liver disease

Pathophisiology Pathophisiologically, the terminal event in this illness is injury and/or death of hepatocytes. Although the actual pathogenesis is not well understood, this damage is probably due to liberation of chemical and immunological mediators.

FHF is announced by some signs, such as: • severe jaundice (> 15 mg% in adults or > 7 mg% in infants); • mild jaundice does not excluded the fulminant hepatitis; • persistence of vomiting and anorexia after the jaundice have installed; • presence of bleedings • persistence of fever in the presence of jaundice • quickly decreased in size of liver; • tachicardia instead of bradicardia in an icteric and afebrile patient • foetor hepaticus

In stage I coma, mild mental changes are present, but asterixis is minimal or absent. Stage II coma is marked by worsening of mental changes and definite asterixis. In stage III coma, the patient develops stupor and semicoma, but can still be roused. With stage IV coma, the patient no longer is arousable, and there may or may not be a response to deep pain stimuli. Patients with hepatic failure, may demonstrate other neurologic sign - flapping of the tongue, involuntary movements, long-tract signs, and decerebrate posturing.

Biological data that announced the FHF are: • decreased of Prothrombin - time (Quick index < 30%) • decreased of plasminogen (<20%) • raised blood ammonia (>120 mg%) • leukocytosis with increased level of neutrophils • sudden decreased of ALT.

Table 2. Complications of FHF and their treatment: (1) Cerebral oedema Renal failure *Monitor intracranial pressure (extradural monitor) if encephalopathy reaches grade III or IV *Avoid manoeuvres that increase intracranial pressure *Control restlessness *If intracranial pressure > 20 -30 mm Hg, apply: 1. Hyperventilation (Pa. CO 2 25 -3 - mm Hg) then 2. Mannitol 0, 5 g/kg in bolus then 3. Haemodialysis or haemofiltration then 4. Barbiturate coma *Avoid arterial hypotension, nephrotoxic drugs, control infection*Dopamine 2 - 4 microgm/min*Haemodialysis or haemofiltration*Careful hygienic measures*High index of suspicion of infection*Daily cultures of blood, urine, and other biological fluids*Start empiric antibiotic therapy*Possible selective intestinal decontamination*Possible parenteral antibiotic prophylaxis

Table 2. Complications of FHF and their treatment: (2) Bacterial infection *Careful hygienic measures *High index of suspicion of infection *Daily cultures of blood, urine, and other biological fluids *Start empiric antibiotic therapy *Possible selective intestinal decontamination *Possible parenteral antibiotic prophylaxis Circulatory dysfunction *Insert pulmonary-artery catheter and measure indicators of tissue hypoxia frequently *Avoid vasoconstrictors *Possibly treat with N-acetylcysteine Haemorrhagic complications *Upper gastrointestinal bleeding; ranitidine or sucralfate *Fresh frozen plasma only in cases of bleeding and before invazive procedures Hypoglicemia *Measure blood glucose concentration every 1 hour *Intravenopus hypertonic glucose Other complications *Pulmonary oedema, respiratory alkalosis, hyponatremia, hypophosphatemia, pancreatitis, aplastic anaemia

Persistence of HBs. Ag for more than 6 months after onset in high concentrations (>100 PEI units/ml) means that: 1. the acute hepatitis B evolved to chronicity; 2. a pre-existing chronic HBV infection was superimposed by an acute hepatitis of another etiology; or 3. the acute disease may actualy be an exacerbation of chronic hepatitis B. Full recovery is assumed if the ALT reaches normal or close to normal levels and if HBs. Ag becomes negative in EIA.

Chronic hepatitis B Clinical manifestations of chronic hepatitis are mild and non-specific : lack of energy, malaise, ease fatigability, myalgias, arthralgias, and skin rash. The evolution is usually intermittent with acute exacerbations consisting on: abdominal discomfort, nausea, weight loss, dark urine, jaundice, loss of appetite.

Cirrhosis The above mentioned symptoms are more important, associated or not with signs of hepatic encephalopathy, blood coagulation is decreased, serum albumin is decreased; the ALT levels are only moderately enhanced, and often fluctuate. The liver status can be assessed by histology, liver function tests, and determination of serum proteins with liver metabolism. Hepatocellular carcinoma In highly epidemic areas, persons positive for Hbs. Ag are 100 times more likely to develop HCC compared to those who have anti-HBs. Abdominal imaging by ultrasound of Hbs. Ag-positive patients with liver cirrhosis is advisable at intervals of 3 -4 months.

Complications Neurological complication: encephalitis, meningoencephalitis, Guillain-Barre syndrome, mononeuritis affecting cranial or peripheral nerves. Myocardial involvement: myocarditis and electrocardiographic changes Pancreatitis Pleural effusions and ascites Aplastic anemia Polyarteritis nodosa Membranoprolipherative glomerulonephritis Leucocytoclastic vasculitis

Treatment The main aims of treatment of chronic HBV infection are: • Viral suppression (as shown by loss of HBe. Ag with or without seroconversion to anti-HBe) and a decrease in HBV DNA; • Reduction in liver damage (return to normal values of serum aminotransferases, histological improvement on liver biopsy samples; • Complete eradication of the virus (as shown by loss of HBs. Ag, with seroconversion to anti-HBs) and the absence of HBV DNA detection.

Agents for the treatment of chronic hepatitis B Interferon alfa – it is given parenterally either 5 MU daily or 10 MU three times per week with loss of HBe. Ag and HBV DNA in only 20% of cases, and loss of HBs. Ag in only 6% of cases. Lamivudine Entecavir Telbivudina Adefovir Tenofovir

Hepatitis B vaccines Long-term immunity against HBV infection is conferred by the presence in the blood of adequate levels of anti. BHs, the antibody to the surface antigen. It is generaly accepted that the threshold anti-HBs titre for protection is 10 m. IU/ml. It is significant to note that a 3 -dose schedule of Engerix. B (10 microg/dose) alone gives protective efficacy rates of 94, 8%- not significantly different from the efficacy rates of 97, 6% and 97, 4% for vaccinees who also received HBIG.

Passive immunization A special immunoglobulin is produced from donors who are naturally immune and boosted with plasma-derived vaccine. Passive immunization is recommended together with active immunization to prevent HBV infection in newborns from HBs. Ag and HBe. Ag mothers, and after proven accidental exposure of a non-immune person.

HEPATITIS D Hepatitis D virus is a defective RNA virus that uses HBs. Ag to produce delta hepatitis.

Transmission is similar to that of HBV. A high prevalence of HDV infection is seen in South America, Central Africa, southern Italy and Eastern European countries. Acute HDV infection occurs in two forms depending on the state of HBV infection: Co-infection-acute delta hepatitis occurs simultaneously with acute hepatitis B. Most patients in this situation recover. The rate of fulminant infection is higher than in superinfection. Superinfection- acute delta hepatitis developed on a chronic hepatitis B.

Laboratory data Delta antigen can sometimes be detected in serum during the early phase of acute delta hepatitis. HDV RNA can be detected in the serum and liver using molecular hybridization technology. Anti-HDV arises with the disappearance of the Delta antigen. HBs. Ag is present concomitantly with Ig. M anti-HBc in early phases of co -infection and with Ig. G anti HBc in superinfection. Diagnosis of chronic HDV infection involves: High titers of anti HDV (>1: 100 by radioimmunoassay) Detection of HDV antigen Persistence of Ig. M anti HDV in serum.

Evolution Chronic delta hepatitis is more severely than chronic hepatitis B alone or chronic hepatitis C, and 60 -70% of patients develops cirrhosis.

HEPATITIS C It is a small single-stranded RNA virus that is distantly related to members of the Flaviviridae family (e. g. , Dengue viruses, Yellow fever virus, and the Japanese encephalitis virus). According to WHO data, it is estimated that 3% of the world population has been infected with hepatitis C virus. Therefore, there may be more than 170 million chronic carriers in the world

1. Blood products 2. Intravenous drug users 3. Donor organs (4. The importance of sexual transmission is controversial. The frequency of HCV transmission to female sexual partners was 5 times higher when HIV was also transmitted, possibly because co-infection with HIV leads to higher levels of HCV in serum. 5. The risk of developing hepatitis following needle-stick exposure is 6. Vertical transmission of this virus –the risk of transmission is much lower than for hepatitis B infection. Nosocomial risk is a major point of concern, as documented in oncology units

Pathogenesis Hepatitis C virus is a member of the Flaviviridae family, which includes the flaviviruses and pestiviruses. that encodes a large single polyprotein of 3000 amino-acids: □ the N-terminal one third harbors the structural proteins; □ the C-terminal two thirds contains the nonstructural proteins:

Antiviral immune response Th 1: interferon-gamma and interleukin-2, which are important stimuli for the development of the host antiviral immune responses, including cytotoxic Tlymphocyte (CTL) generation and NK-cell activation Th 2: interleukin-4, interleukin-5 and interleukin-10, which enhance antibody production and downregulate the Th 1 response. The imbalance between the Th 1 and the Th 2 responses is implicated in disease progression and the inability to clear the infection.

Humoral immune response Cellular immune response The cellular immune response has the ability to recognize and eliminate

Clinical manifestations HCV infection is infrequently diagnosed during the acute phase of infection. Incubation period is usually 7 to 8 weeks (range 2 to 26). The majority of persons have either no symptoms or only mild symptoms. Symptoms of acute hepatitis usually consist of: jaundice, malaise and nausea. A characteristic feature of all these patients is marked fluctuation of aminotransferase level. Although the aminotransferase level (ALT) may occasionally peak as high as 1000 to 2000 IU/L, Extrahepatic manifestations of HCV infection are associated with autoimmune or lymphoproliferative states Lymphoproliferative disorders 1. Mixed cryoglobulinemia. . 2. Lymphoma.

Other extrahepatic diseases Autoimmune thyroiditis Dermatological manifestations- apart from the palpable purpura due to leukocytoclasic vasculitis (the most frequently seen dermatological manifestation of HCVrelated EMC) other cutaneous disorders are: sporadic porphyria cutanea tarda and cutaneous/mucosal lichen planus Sjogren’s syndrome Idiopatic pulmonary fibrosis Mooren corneal ulcers

Tests for HCV There are still no tests for antigens of hepatitis C virus in serum. Infection is usually diagnosed by detecting hepatitis C virus antibodies based on immunoassay detection. 1. HCV antibody testing 2. Detection of serum HCV RNA by PCR is the best test for early diagnosis of acute HCV infection (it is positive as soon as 1 week after exposure). 3. HCV genotypes. HCV was classified into 6 types (1 to 6) each including subtypes (a, b, c. . . ) 4. Quasispecies There is a correlation between a high quasispecies heterogeneity an a more severe liver disease and a poor response to interferon therapy.

Evolution The infection becomes chronic in most cases (80%), and chronic infection is typically characterized by a prolonged period in which there are no symptoms. At the cirrhotic stage, about 3 -5% of patients per year develops hepatocarcinoma, which may develop in up to 50% within 10 years. Chronically infected individuals generally have fluctuating or continuously abnormal levels of ALT and are viraemic.

Treatment of Hepatitis C infection Prior to treatment, the investigation should: ascertain that chronic hepatitis C virus infection (HCV) is present; exclude contraindication of treatment; evaluate histological findings determine the viral genotype, and for genotype 1 patients, also, the viral load.

Exclude contraindication for treatment Contraindications for interferon (IFN) and IFN/ribavirin combination therapy are: Pregnancy or no optimal contraception(ribavirin) Severe heart disease CNS dysfunction/epilepsy Decompensated liver disease(interferon) Organ transplant, except liver transplant Severe renal failure(ribavirin) Untreated severe anemia(ribavirin) Hemoglobinopathies(ribavirin) Autoimmune hepatitis(interferon) Ongoing or recent drug or alcohol abuse Leukopenia(WBC<1500/mm 3, neutrophols<750/mm 3) Thrombocytopenia(<50000/mm 3)

Relative contraindications: -Psychiatric disease/depression -Not well controlled thyroid disease or diabetes melitus -Autoimmune disease -Mild bone marrow suppression.

Evaluation of the histological damage Patients with mild histological inflammation without fibrosis will probably not develop serious liver disease, not even with prolonged follow-up. A second histological evaluation after 2 -5 years is therefore recommended for this patient cathegory. Cirrhotic patients, with manifest disease, should only be treated if they are in compensated phase, while those with decompensated cirrhosis should be evaluated and considered for possible liver transplantation.

Recommend treatment if: HCV-RNA is detectable ALT above the upper normal reference level No contraindications for treatment Liver biopsy showing fibrosis stage >=2 and ongoing inflammation or/ fibrosis stage 1 and inflammation grade >2 Treatment with: -alfa-IFN 3 MU s. c. , 3 times a week + ribavirin 1000 -1200 mg /day. Non-responders to previous IFN monotherapy: -Lack of sufficient information - no recommendation given.

Pegylated Interferon Indication of pegylated interferon (with ribavirin): first intention therapy of patients with: fibrosis-stage 3 (Knodell or Metavir score), necroinflamatory score>9, hemophilia/thalasemia, age<55 years recurrences /absence of response after the standard therapy with interferon and ribavirin, coinfections. Inhibition of viral replication might also be achieved by: - Antisense oligonucleotides that bind to specific sequences in the HCV RNA. -Ribozymes that catalyze cleavage of HCV RNA. - Gene transfer of interfering proteins, that specifically interrupt virion assembly in the hepatocyte.

HEPATITIS E Hepatitis E virus was identified by Balayan et al. (1983) in human fecal samples. In 1995 this virus has been classified within the Caliciviridae. HEV is a non-enveloped, RNA virus with single-stranded genome

Epidemiology Hepatitis E could be a sporadic disease or may evolve in outbreaks with contaminated drinking water as the source of infection. The most frequently involved regions are: India, China, Pakistan, Egypt, Algeria, Sudan, Ethiopia, Mexico. HEV infections generally occurred after the age of 16 years

Pathogenesis Pathological changes in the liver include hepatocyte necrosis and inflammatory cell infiltration

Clinical manifestations Incubation period has an average of 40 days. HEV produces an acute, self-limited disease without progression through chronic hepatitis. The prodromal symptoms include: malaise, anorexia, abdominal discomfort. In women infected during the third trimester of pregnancy was noted a high rate of fulminant disease and a 15 -20% case-fatality rate.

Diagnosis Detection of HEV-RNA by PCR Detection of HEV-Ag by electronic immunomicroscopy Detection of HEV-antibody: • immunofluorescence inhibition • ELISA (Ig. M, Ig. G) • Immunotransfert (Ig. M, Ig. G)

GB VIRUS-C/HEPATITIS G VIRUS Virology and tissue tropism GBV-C/HGV is an enveloped RNA virus which belongs to the Flaviviridae family. The envelope 2 (E 2) region encodes a glycoprotein which is located on the outer surface of the virion. At least 4 major genotypes exists. GBV-C/HGV may replicate in human peripheral blood mononuclear cells and endotelial cells, in liver tissue, spleen and bone marrow.

Prevalence and transmission There is a high prevalence of viremia in healthy persons in Europe and North America (1 -4%) and a higher prevalence rate in South America and Africa (10 -33%). GBV-C/HGV may be transmitted via blood/blood products, by sexual contacts, by close social contact, and from mother to infant.

Clinical features The majority of adults have a transient infection with clearence of viremia and development of E 2 antibodies. GBV-C/HGV viremia after vertical or perinatal transmission may persist for at least 36 months.

Diagnostic Reverse transcription polymerase chain reaction (RT-PCR) – PCR is used to diagnose ongoing infections and does not give any information on a resolved infection (may underestimate the prevalence/occurrence of these infections). EIA In situ hybridization to detect GBV-C RNA Immunofluorescence. Liver biopsy

Infection with TT Virus TTV is a unenveloped, single stranded, circular DNA virus that was isolated from a Japanese patient with post-transfusion hepatitis of unknown etiology. Although TTV DNA titers have been shown to be closely correlated with aminotransferase levels in the sera of some patients during post-transfusion hepatitis, no clear association between TTV infection and human liver disease has been establish at this time. TTV is a parenterally transmissible virus TTV DNA was detected in 10% of normal population in England, in 34% of voluntary US blood-donors and in 47% of patients with acute and chronic non A-G hepatitis. Very high prevalences of viremia (>50%), observed in healthy populations in different countries, indicate that persistent infection is common and that routes of transmission other than the parenteral route do exist. However, the main mode of TTV transmission remains to be determined.
- Slides: 74