Viral Diagnostics Lab Beating the Next Pandemic Viral

Viral Diagnostics Lab: Beating the Next Pandemic
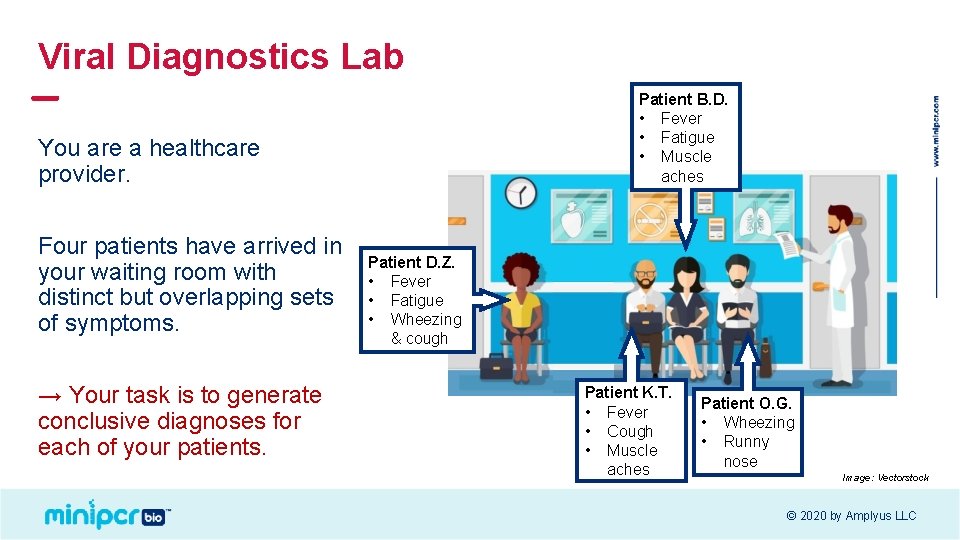
Viral Diagnostics Lab Patient B. D. • Fever • Fatigue • Muscle aches You are a healthcare provider. Four patients have arrived in your waiting room with distinct but overlapping sets of symptoms. → Your task is to generate conclusive diagnoses for each of your patients. Patient D. Z. • Fever • Fatigue • Wheezing & cough Patient K. T. • Fever • Cough • Muscle aches Patient O. G. • Wheezing • Runny nose Image: Vectorstock © 2020 by Amplyus LLC

Viral Diagnostics Lab We will test for two viruses that may have infected your patients: Seasonal influenza Novel influenza Q virus (n. IQV) • Vaccine available • Emergent (and fictional) virus • Fatal to 0. 1% of sufferers • No vaccine available • Fatal to 4 -8% of sufferers Discussion question: What are some potential consequences of an incorrect diagnosis? © 2020 by Amplyus LLC

Background: What is a virus? A virus is an infectious agent with a simple structure: • DNA or RNA genome • Enclosed in a shell Because they lack organelles and other cellular machinery, viruses cannot reproduce independently of a host. Since they are unable to carry out some of the critical functions of life – including independent reproduction – viruses are considered nonliving. © 2020 by Amplyus LLC
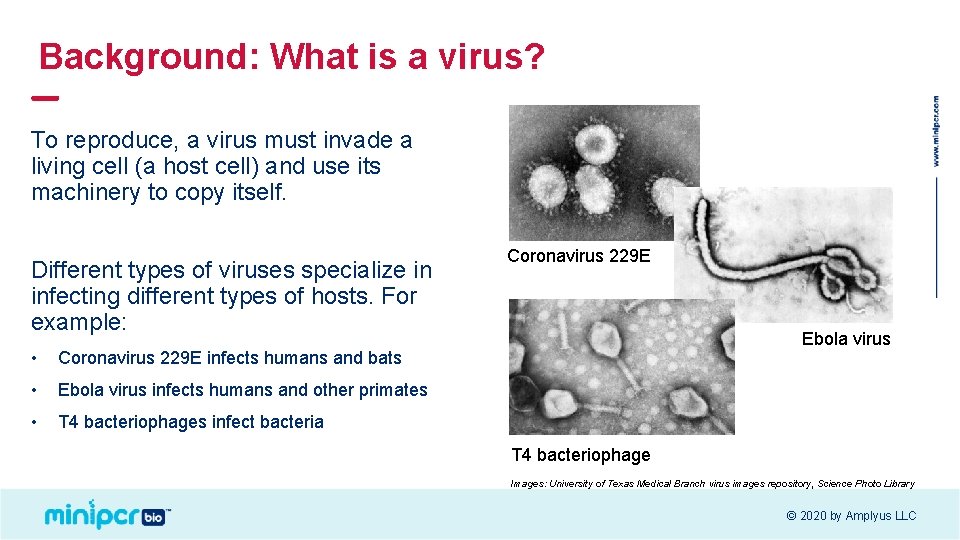
Background: What is a virus? To reproduce, a virus must invade a living cell (a host cell) and use its machinery to copy itself. Different types of viruses specialize in infecting different types of hosts. For example: • Coronavirus 229 E infects humans and bats • Ebola virus infects humans and other primates • T 4 bacteriophages infect bacteria Coronavirus 229 E Ebola virus T 4 bacteriophage Images: University of Texas Medical Branch virus images repository, Science Photo Library © 2020 by Amplyus LLC

Background: Viral replication cycle Virus particle Host cell Viruses infect their hosts via the following steps. (*Though note that the details of this process will vary from virus to virus. ) Receptors © 2020 by Amplyus LLC
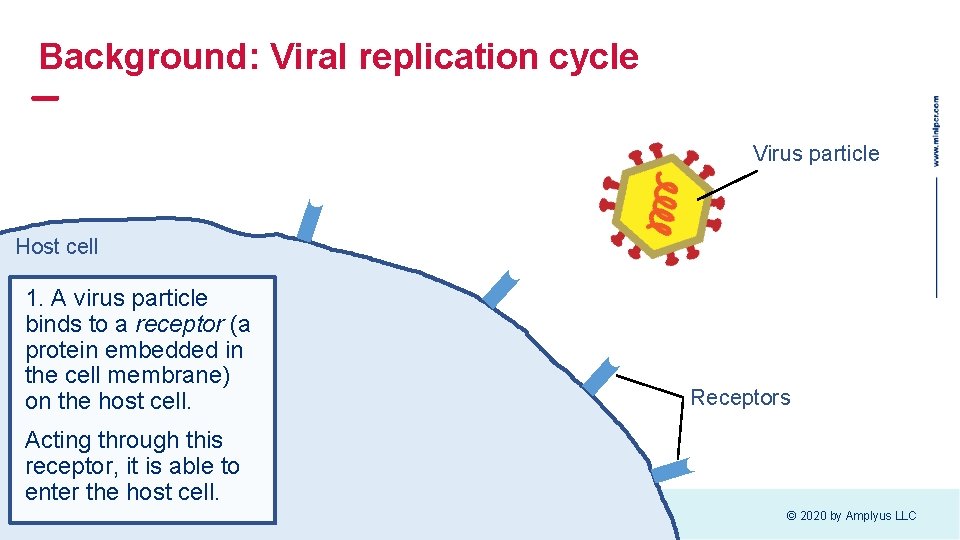
Background: Viral replication cycle Virus particle Host cell 1. A virus particle binds to a receptor (a protein embedded in the cell membrane) on the host cell. Receptors Acting through this receptor, it is able to enter the host cell. © 2020 by Amplyus LLC
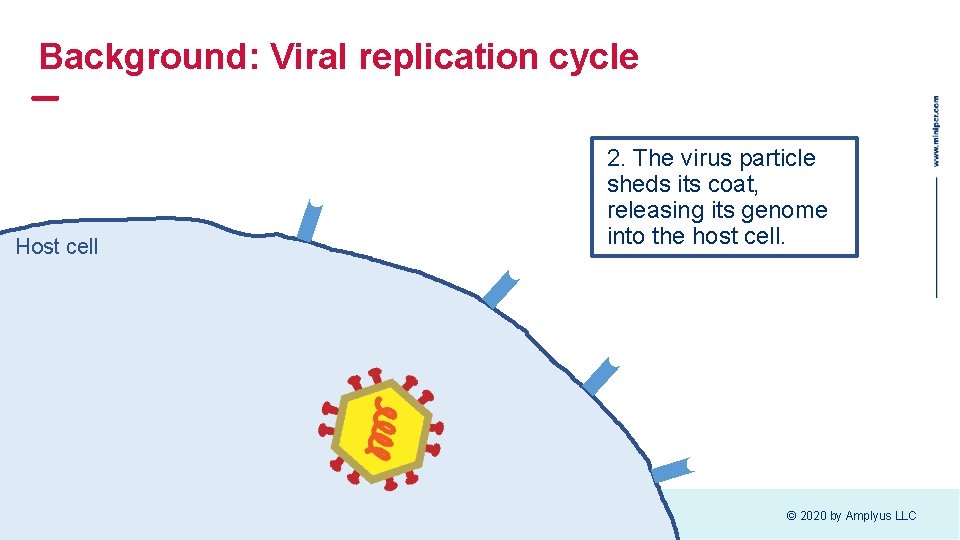
Background: Viral replication cycle Host cell 2. The virus particle sheds its coat, releasing its genome into the host cell. © 2020 by Amplyus LLC
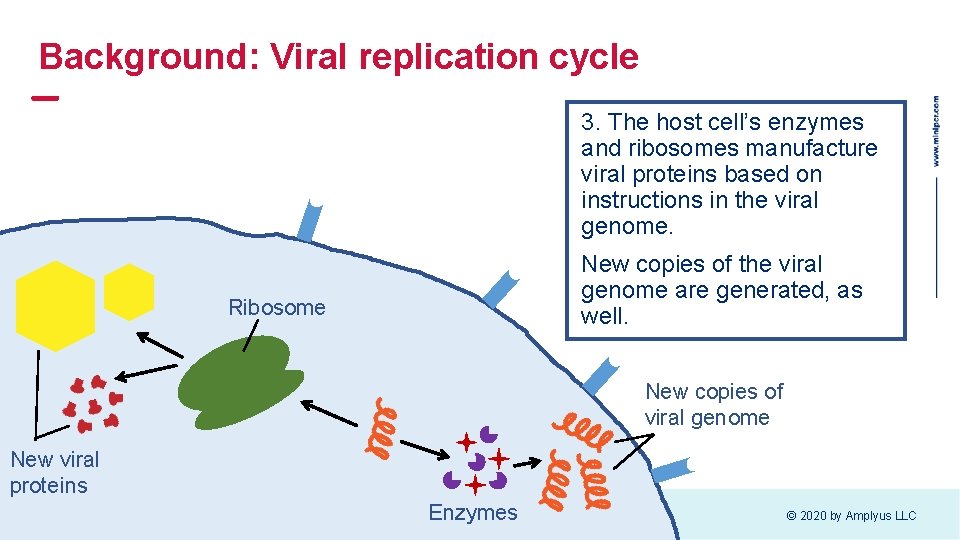
Background: Viral replication cycle 3. The host cell’s enzymes and ribosomes manufacture viral proteins based on instructions in the viral genome. New copies of the viral genome are generated, as well. Ribosome New copies of viral genome New viral proteins Enzymes © 2020 by Amplyus LLC
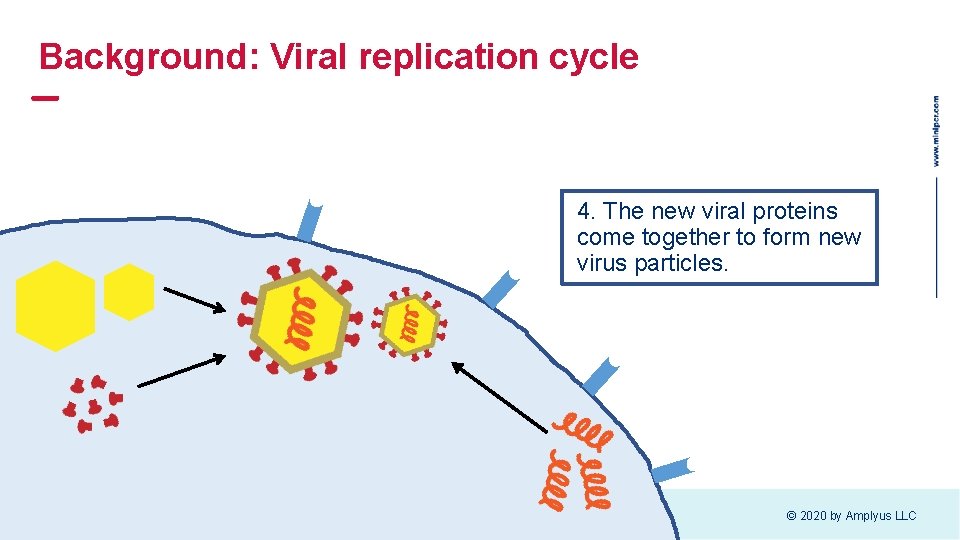
Background: Viral replication cycle 4. The new viral proteins come together to form new virus particles. © 2020 by Amplyus LLC

Background: Viral replication cycle 5. Newly assembled virus particles emerge from the cell, ready to infect new host cells. Sometimes new viruses are slowly leaked from the host cell, and sometimes they burst out of the host cell in a massive swarm. But this process almost always results in the death of the host cell. *Note: This example was simplified, but one virus particle typically gives rise to hundreds or thousands of new virus particles. © 2020 by Amplyus LLC

Background: About receptors Any given virus uses surface proteins on its coat to bind only to a limited range of receptors. A virus can only infect a cell if that cell bears specific receptors on its surface. This is why you can’t typically catch a cold from your dog – the receptors on your cells differ too much The green virus particles are only equipped to bind to the green receptors. They will not be able to infect cells lacking green receptors. © 2020 by Amplyus LLC

Background: Viral mutations • If the surface proteins on a virus change – i. e. , via a genetic mutation – the virus may gain the ability to bind to new types of receptors and therefore infect new hosts. • Mutations to the viral genome happen randomly, just like in living things. • Many have no effect on how the virus functions, and some make it harder for the virus to replicate, but others may help the virus infect new species. • An event like this is thought to have triggered the COVID-19 pandemic; scientists believe a mutation in a strain of coronavirus carried by bats allowed the virus to jump species and eventually infect human cells. © 2020 by Amplyus LLC

Background: Viral evolution Discussion question: Why is it that we rarely witness the emergence of new animal or plant species, but over the past few decades we’ve seen Zika, MERS, swine flu, and COVID-19 emerge? • Compared to plants and animals, viruses evolve rapidly. This is because: • Viruses are abundant. Scientists have estimated there are 10^31 individual virus particles on Earth. More virus particles = more opportunities for mutations. • Viruses are prolific, meaning they reproduce in great numbers. A single virus particle produces thousands of “offspring” in a single replication cycle, so mutations can be spread through a population rapidly. • Viruses – especially those with RNA genomes – are error-prone. That is, the enzymes that copy their genetic material are more likely to introduce mutations. © 2020 by Amplyus LLC
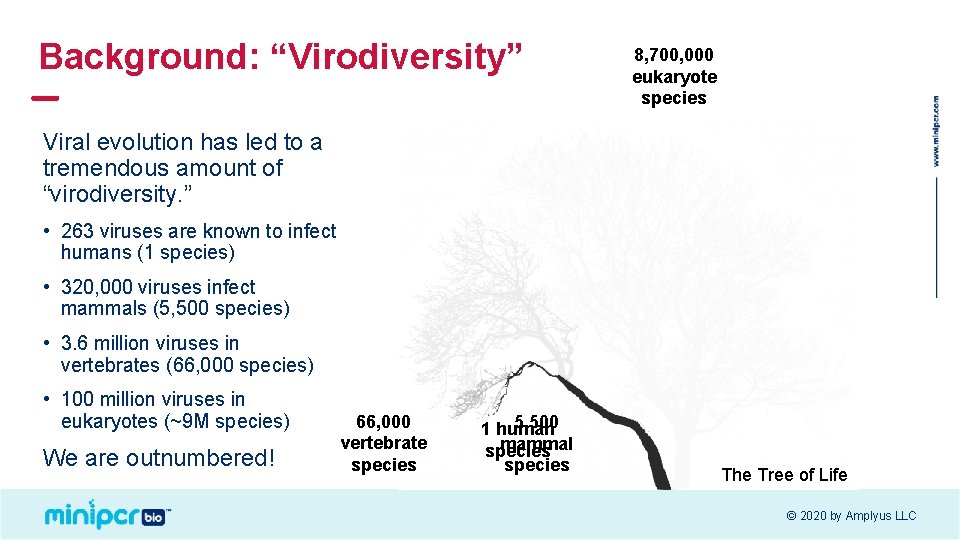
Background: “Virodiversity” 8, 700, 000 eukaryote species Viral evolution has led to a tremendous amount of “virodiversity. ” • 263 viruses are known to infect humans (1 species) • 320, 000 viruses infect mammals (5, 500 species) • 3. 6 million viruses in vertebrates (66, 000 species) • 100 million viruses in eukaryotes (~9 M species) We are outnumbered! 66, 000 vertebrate species 5, 500 1 human mammal species The Tree of Life © 2020 by Amplyus LLC

Background: Nucleic acid detection tests Viruses that infect the same body systems can produce very similar symptoms. • E. g. , a cold and a case of seasonal flu may both cause coughing, sneezing, and runny nose Healthcare providers can use nucleic acid detection tests to determine what virus is making a patient sick. • Nucleic acid tests look for viral DNA or RNA in patients’ bodies. If viral DNA or RNA is found in a patient sample, the patient is likely infected with that virus. Collect patient sample Extract DNA & RNA Convert RNA to c. DNA Copy target gene from virus Visualize © 2020 by Amplyus LLC

Background: Nucleic acid detection tests Step 1: Collect tissue from patient. For a respiratory virus like the flu, tissue from respiratory tract is sampled. Collect patient sample Extract DNA & RNA Convert RNA to c. DNA Copy target gene from virus Visualize © 2020 by Amplyus LLC
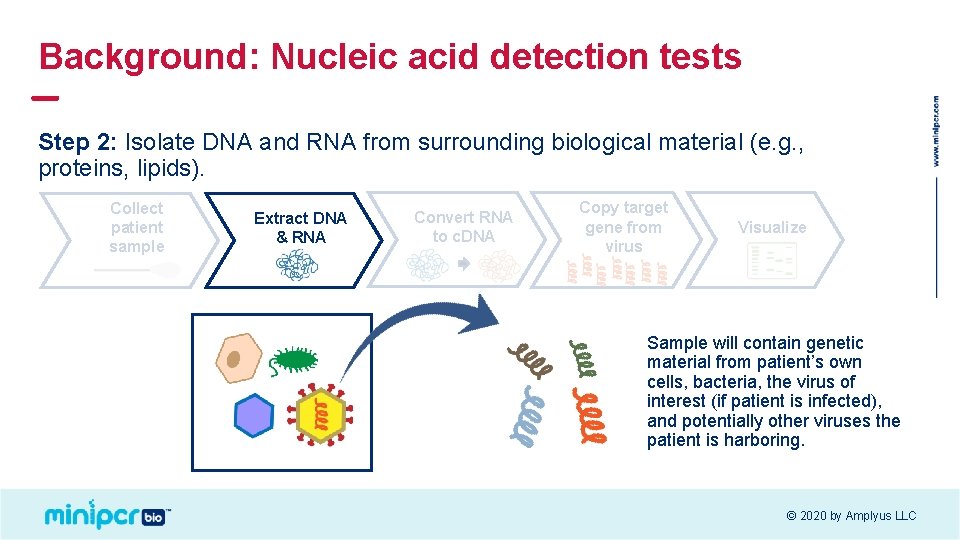
Background: Nucleic acid detection tests Step 2: Isolate DNA and RNA from surrounding biological material (e. g. , proteins, lipids). Collect patient sample Extract DNA & RNA Convert RNA to c. DNA Copy target gene from virus Visualize Sample will contain genetic material from patient’s own cells, bacteria, the virus of interest (if patient is infected), and potentially other viruses the patient is harboring. © 2020 by Amplyus LLC

Background: Nucleic acid detection tests Step 3: Use polymerase chain reaction (PCR) to amplify (make many copies of) a gene segment specific to the virus of interest. Collect patient sample Extract DNA & RNA Convert RNA to c. DNA Copy target gene from virus Visualize © 2020 by Amplyus LLC
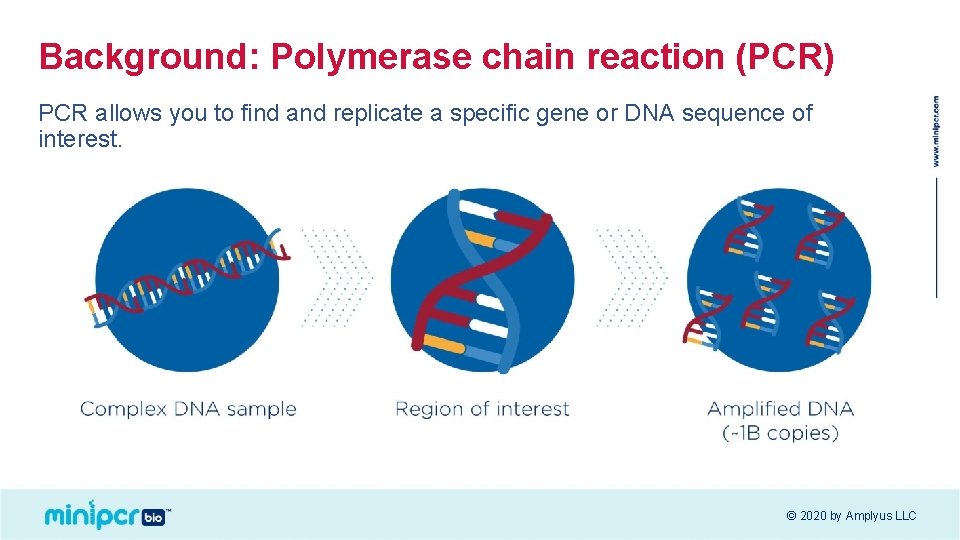
Background: Polymerase chain reaction (PCR) PCR allows you to find and replicate a specific gene or DNA sequence of interest. © 2020 by Amplyus LLC

Background: PCR in a nucleic acid detection test In a nucleic acid detection test, we will selectively amplify a gene from our virus of interest. DNA from patient’s cells DNA from another virus the patient carries DNA from bacteria in respiratory tract DNA from virus of interest © 2020 by Amplyus LLC
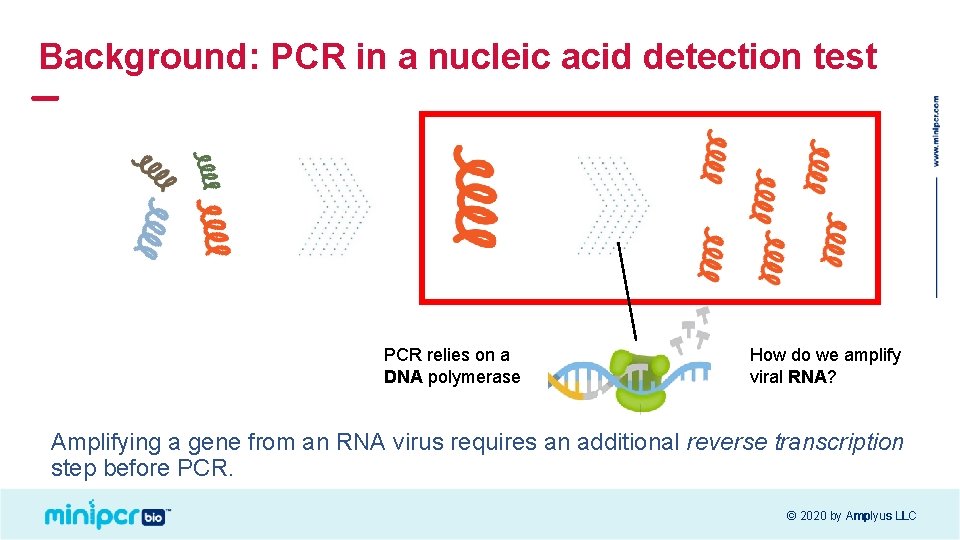
Background: PCR in a nucleic acid detection test PCR relies on a DNA polymerase How do we amplify viral RNA? Amplifying a gene from an RNA virus requires an additional reverse transcription step before PCR. © 2020 by Amplyus LLC

Background: Reverse transcription Remember that in transcription, an RNA transcript is generated from a DNA template. In reverse transcription, the opposite occurs: a DNA transcript is generated from an RNA template. We can use an enzyme called reverse transcriptase to carry out this process. © 2020 by Amplyus LLC
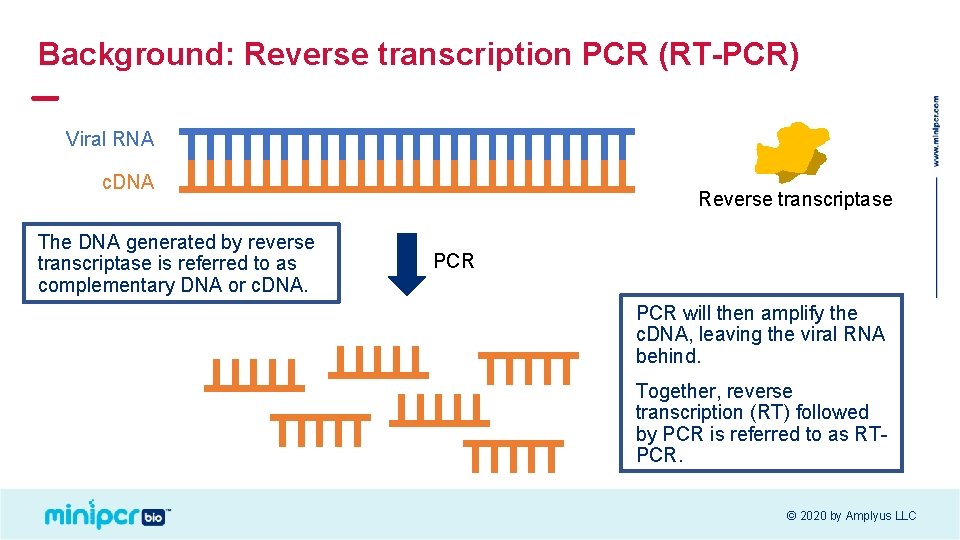
Background: Reverse transcription PCR (RT-PCR) Viral RNA c. DNA The DNA generated by reverse transcriptase is referred to as complementary DNA or c. DNA. Reverse transcriptase PCR will then amplify the c. DNA, leaving the viral RNA behind. Together, reverse transcription (RT) followed by PCR is referred to as RTPCR. © 2020 by Amplyus LLC

Background: Nucleic acid detection tests Step 4: Detect the amplified viral DNA. This is often done by a machine using a technique called quantitative PCR (q. PCR). Today, you will directly visualize the amplified DNA using gel electrophoresis. Collect patient sample Extract DNA & RNA Convert RNA to c. DNA Copy target gene from virus Visualize In this lab, the first 3 steps of a nucleic acid detection test (sample collection, DNA extraction, and RT-PCR) have already been completed. You will only carry out the final (visualization) step. © 2020 by Amplyus LLC

Background: Agarose gel electrophoresis • In gel electrophoresis, an electrical field is used to separate molecules based on their size. • Agarose gels, made from a sugar extracted from seaweed, are typically used for separating DNA segments. • The electrical field pulls negatively charged DNA molecules through microscopic pores in the gel (see image at right) • Small DNA segments move more quickly than large ones, as they can easily move through the pores of the gel Magnified image of an agarose electrophoresis gel © 2020 by Amplyus LLC

Background: Agarose gel electrophoresis + DNA is negatively charged and migrates toward the positive pole of the electrical field. Over time, small DNA segments travel further than large ones. © 2020 by Amplyus LLC

Background: Agarose gel electrophoresis At the end of an electrophoresis run, you’ll see bands: groups of DNA segments of the same size. © 2020 by Amplyus LLC

blue. Gel: simultaneous electrophoresis + visualization © 2020 by Amplyus LLC
Meet your patients Patient B. D. • Fever • Fatigue • Muscle aches Patient D. Z. • Fever • Fatigue • Wheezing & cough Patient K. T. • Fever • Cough • Muscle aches Patient O. G. • Wheezing • Runny nose Image: Vectorstock © 2020 by Amplyus LLC

Your patients may be suffering from: • Seasonal influenza • Vaccine available • Fatal to 0. 1% of sufferers • Novel influenza Q virus (n. IQV) • Emergent (and fictional) virus • No vaccine available • Fatal to 4 -8% of sufferers • A common cold • Seasonal allergies • Something else entirely © 2020 by Amplyus LLC

Possible diagnoses cause similar symptoms Based on their case details, can you make a hypothesis about what your patients are suffering from? © 2020 by Amplyus LLC

Patient cases (part 1) Patient K. T. • 66 -year-old male retiree • Presents with muscle aches and a persistent cough • Has 102°F fever • Has asthma and seasonal allergies. Was hospitalized for asthma 2 years ago Patient O. G. • 37 -year-old father of three • Works part-time as a nurse • Presents with wheezing and a runny nose • No fever • Received flu vaccine this year. Is in good health aside from seasonal allergies © 2020 by Amplyus LLC

Patient cases (part 2) Patient B. D. : • 15 -year-old female, sophomore in high school • Presents with fatigue and a persistent cough • Has 104°F fever • Otherwise in good health Patient D. Z. : • 42 -year-old female • Vacationing in the U. S. from Australia • Presents with fatigue, shortness of breath, mild wheezing, and cough • Has 102°F fever • Suffers from lupus, a chronic autoimmune disease • Received flu shot in Australia (may differ from U. S. vaccine) © 2020 by Amplyus LLC

Test your hypotheses We will use a nucleic acid detection test to determine whether either of these viruses might be causing your patients’ symptoms: Seasonal influenza Novel influenza Q virus (n. IQV) • Vaccine available • Emergent (and fictional) virus • Fatal to 0. 1% of sufferers • No vaccine available • Fatal to 4 -8% of sufferers Discussion question: Why is a healthcare provider unlikely to give you a test for a common cold? © 2020 by Amplyus LLC
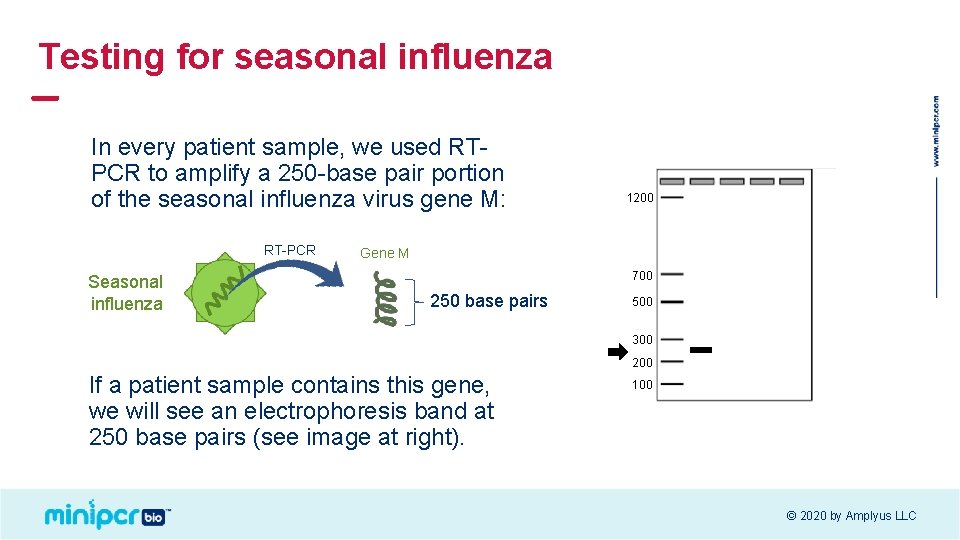
Testing for seasonal influenza In every patient sample, we used RTPCR to amplify a 250 -base pair portion of the seasonal influenza virus gene M: RT-PCR Seasonal influenza 1200 Gene M 700 250 base pairs 500 300 200 If a patient sample contains this gene, we will see an electrophoresis band at 250 base pairs (see image at right). 100 © 2020 by Amplyus LLC
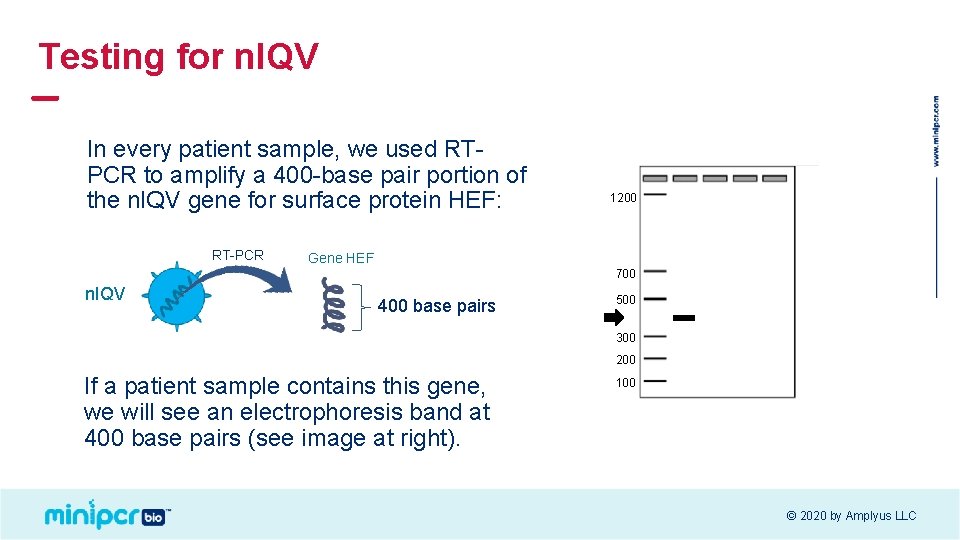
Testing for n. IQV In every patient sample, we used RTPCR to amplify a 400 -base pair portion of the n. IQV gene for surface protein HEF: RT-PCR 1200 Gene HEF 700 n. IQV 400 base pairs 500 300 200 If a patient sample contains this gene, we will see an electrophoresis band at 400 base pairs (see image at right). 100 © 2020 by Amplyus LLC

The importance of controls: Scenario 1 What would it mean to see no bands? 1200 • Not carrying seasonal flu or n. IQV • Or: RT-PCR did not work 700 500 300 200 100 © 2020 by Amplyus LLC
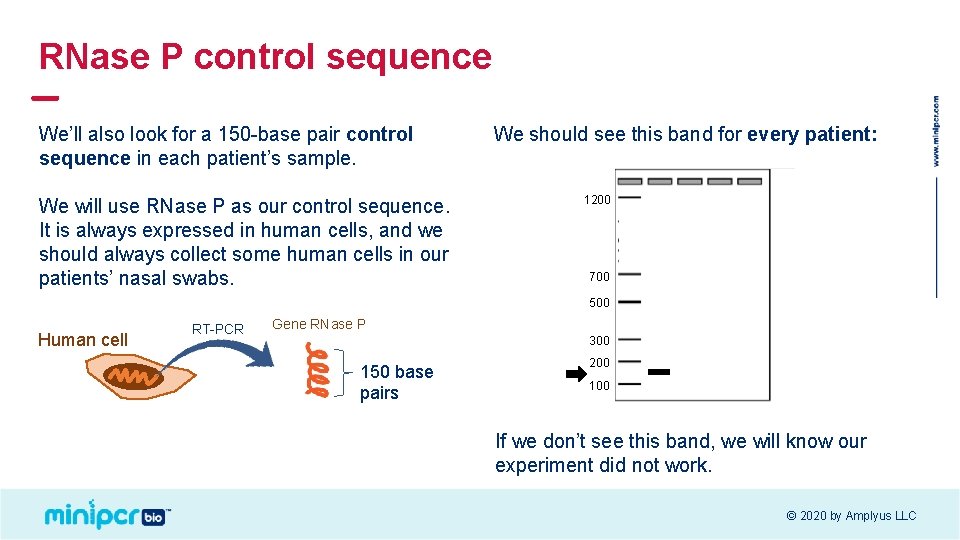
RNase P control sequence We’ll also look for a 150 -base pair control sequence in each patient’s sample. We will use RNase P as our control sequence. It is always expressed in human cells, and we should always collect some human cells in our patients’ nasal swabs. We should see this band for every patient: 1200 700 500 Human cell RT-PCR Gene RNase P 300 150 base pairs 200 100 If we don’t see this band, we will know our experiment did not work. © 2020 by Amplyus LLC
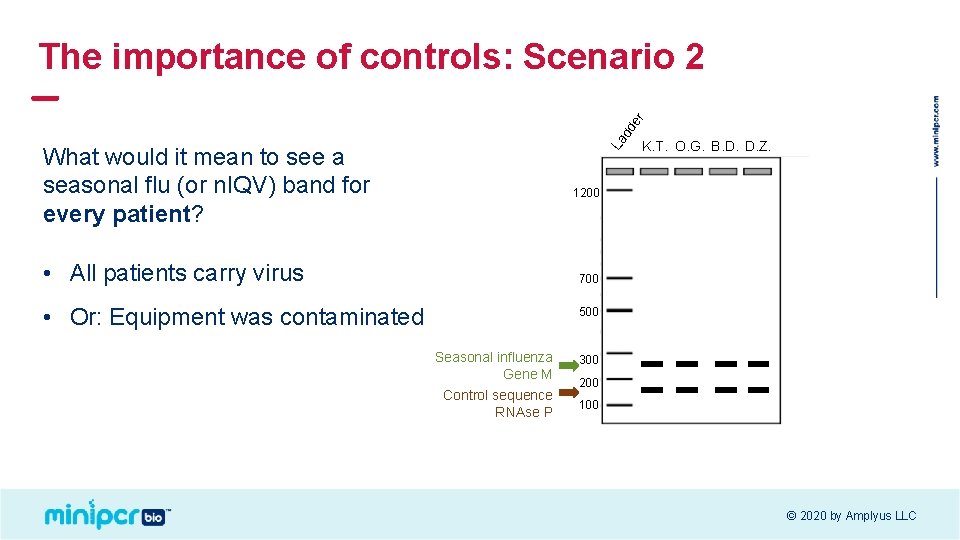
La dd er The importance of controls: Scenario 2 What would it mean to see a seasonal flu (or n. IQV) band for every patient? K. T. O. G. B. D. D. Z. 1200 • All patients carry virus 700 • Or: Equipment was contaminated 500 Seasonal influenza Gene M Control sequence RNAse P 300 200 100 © 2020 by Amplyus LLC
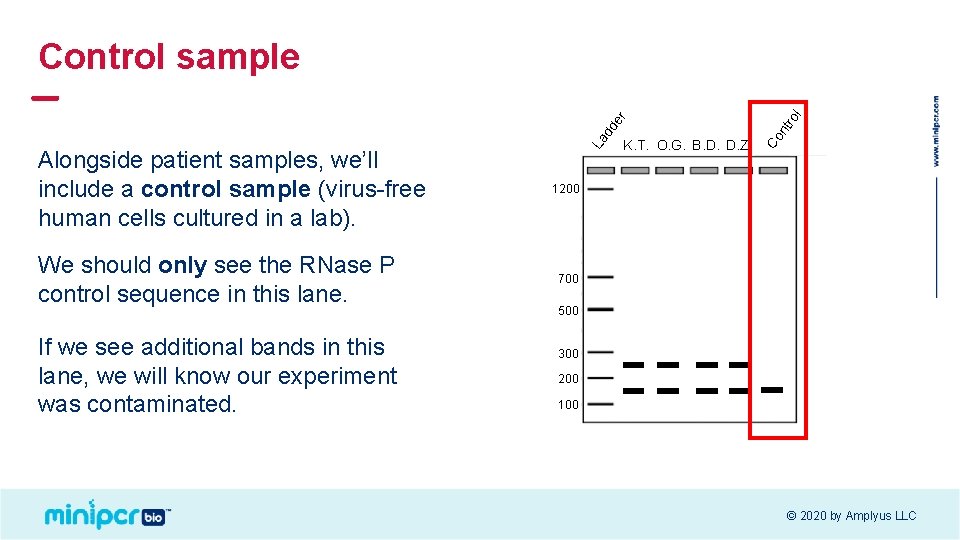
We should only see the RNase P control sequence in this lane. If we see additional bands in this lane, we will know our experiment was contaminated. K. T. O. G. B. D. D. Z. ro l nt Co Alongside patient samples, we’ll include a control sample (virus-free human cells cultured in a lab). La dd er Control sample 1200 700 500 300 200 100 © 2020 by Amplyus LLC
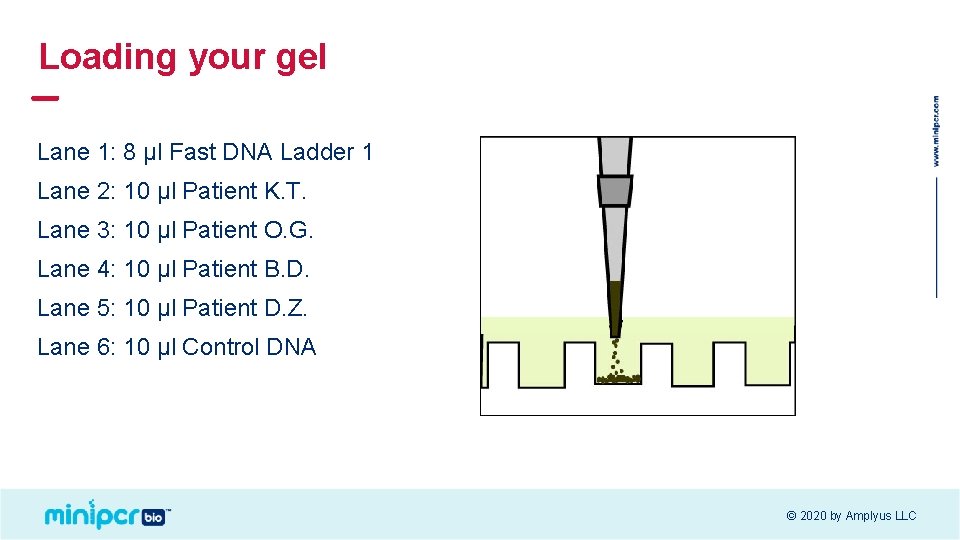
Loading your gel Lane 1: 8 μl Fast DNA Ladder 1 Lane 2: 10 μl Patient K. T. Lane 3: 10 μl Patient O. G. Lane 4: 10 μl Patient B. D. Lane 5: 10 μl Patient D. Z. Lane 6: 10 μl Control DNA © 2020 by Amplyus LLC
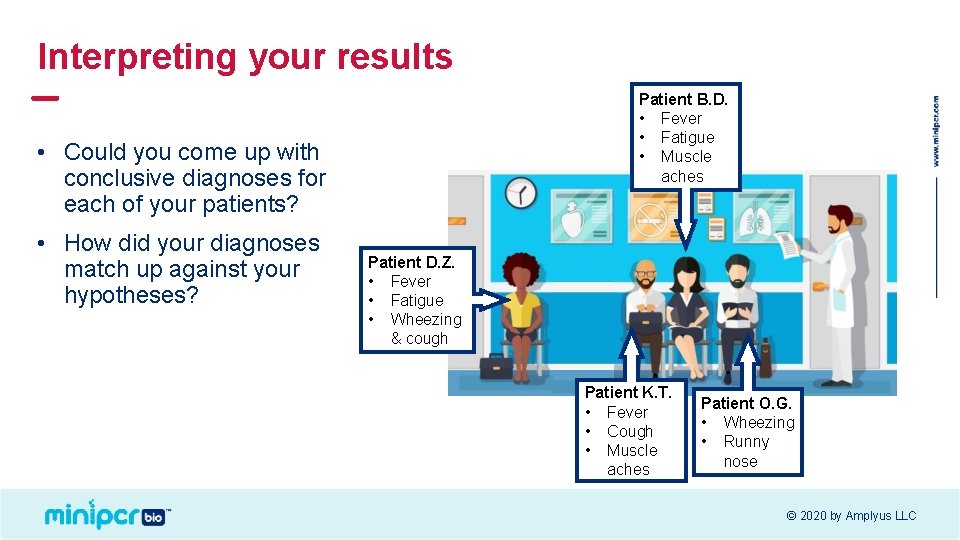
Interpreting your results Patient B. D. • Fever • Fatigue • Muscle aches • Could you come up with conclusive diagnoses for each of your patients? • How did your diagnoses match up against your hypotheses? Patient D. Z. • Fever • Fatigue • Wheezing & cough Patient K. T. • Fever • Cough • Muscle aches Patient O. G. • Wheezing • Runny nose © 2020 by Amplyus LLC

© 2020 by Amplyus LLC
- Slides: 44