Viral Detection Methodologies Group8 Introduction To Laboratory Medicine

Viral Detection Methodologies Group-8 Introduction To Laboratory Medicine. Ug-3 , 6 th Semester

Viral Diagnosis Proudly Presented By: Sheroze Ameen Kinza Waqar Sayeda Sarah Saleem Sameen Ruqia Sayeda Kashmala Zahra Uzma Batool
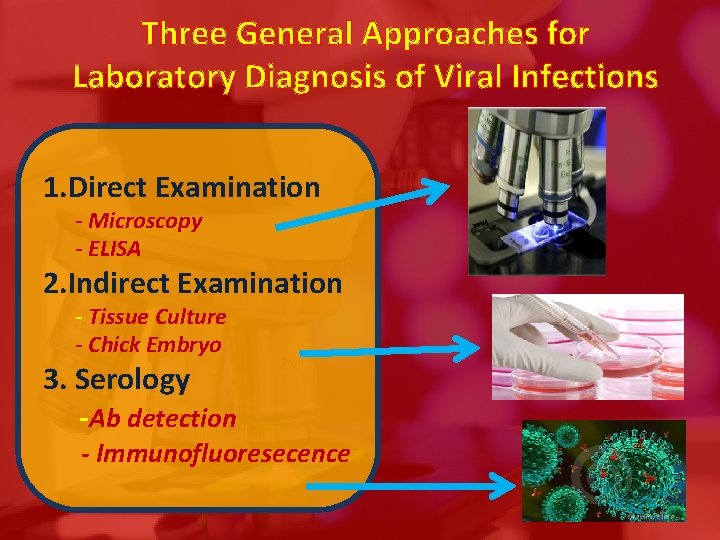
Three General Approaches for Laboratory Diagnosis of Viral Infections 1. Direct Examination - Microscopy - ELISA 2. Indirect Examination - Tissue Culture - Chick Embryo 3. Serology -Ab detection - Immunofluoresecence

Collection of Viral Specimens Nasal Tract Infection Type Of Specimen Virus Type G. l Tract Infection Blood /Vesicular Infection Swabs , Sputum from Throat, Nasal Stool, rectal swab. Urine. Skin scraps, Blood RSV, Influenza A, B Diarrheal and Entero Viruses HSV, Rubella, Pox virus, Rabies, HBV HCV, HIV, CMV

Viral Specimen Storage & Bio-safety Personal protective equipment (PPE) Viral transport medium (VTM) collection vials Store specimen at : 4 °C to 8 °C for short periods of time -20 °C to - 40 °C for long term storage Polyester Fiber-Tipped Applicator

Direct Methods of Virus Detection Shehroze Ameen Direct Methods of Viral Detection
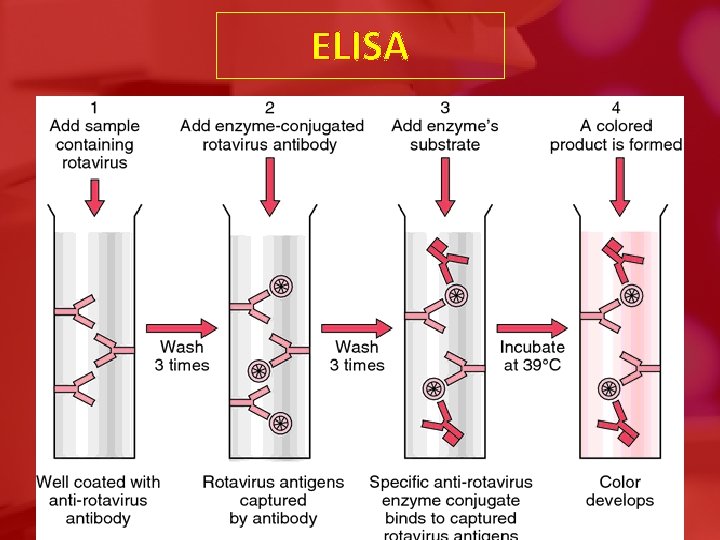
ELISA
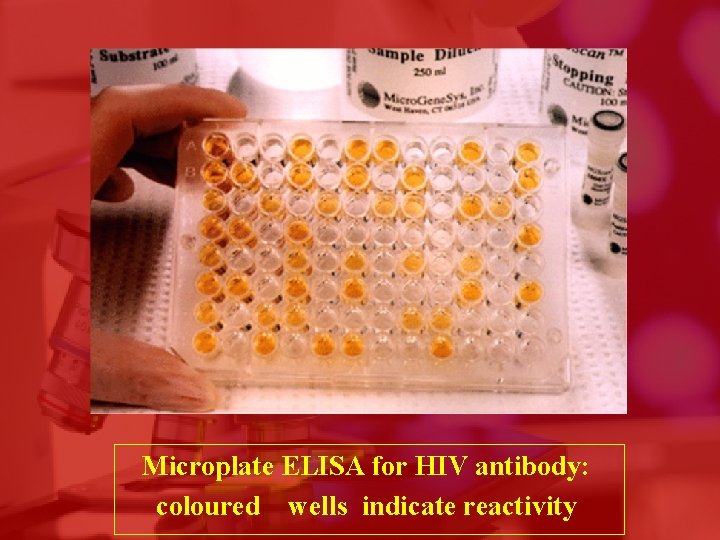
Microplate ELISA for HIV antibody: coloured wells indicate reactivity
Electron Microscopy • immune electron microscopy can increase specificity of EM • disadvantage: expensive, poor sensitivity due to high requirement of viral titres • Virus particles are detected and identified on the basis of morphology
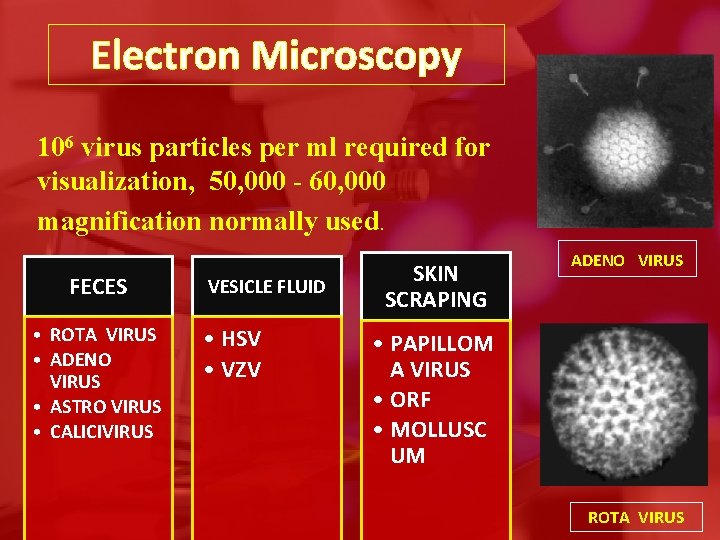
Electron Microscopy 106 virus particles per ml required for visualization, 50, 000 - 60, 000 magnification normally used. FECES • ROTA VIRUS • ADENO VIRUS • ASTRO VIRUS • CALICIVIRUS VESICLE FLUID • HSV • VZV SKIN SCRAPING ADENO VIRUS • PAPILLOM A VIRUS • ORF • MOLLUSC UM ROTA VIRUS

Light Microscope • Replicating virus often produce histological changes in infected cells • Viral inclusion bodies are defined as replicating virus particles either in the nucleus or cytoplasm of infected cells

Light Microscope

RT, Reverse Trancriptase & Allele Specific PCR allows the in vitro amplification of specific target DNA sequences by a factor of 106 and is thus an extremely sensitive technique. It is based on an enzymatic reaction involving the use of synthetic oligonucleotides flanking the target nucleic sequence of interest. These oligonucleotides act as primers for thermostable Taq

PCR Advantages of PCR: 1. Extremely high sensitivity, may detect down to one viral genome per sample volume 2. Easy to set up 3. Fast turnaround time Disadvantages of PCR 1. Extremely liable to contamination 2. High degree of operator skill required 3. Not easy to quantitate results 4. A positive result may be difficult to interpret, especially with latent viruses such as CMV, where any seropositive person will have virus present in their blood irrespective whether

INDIRECT METHODS FOR
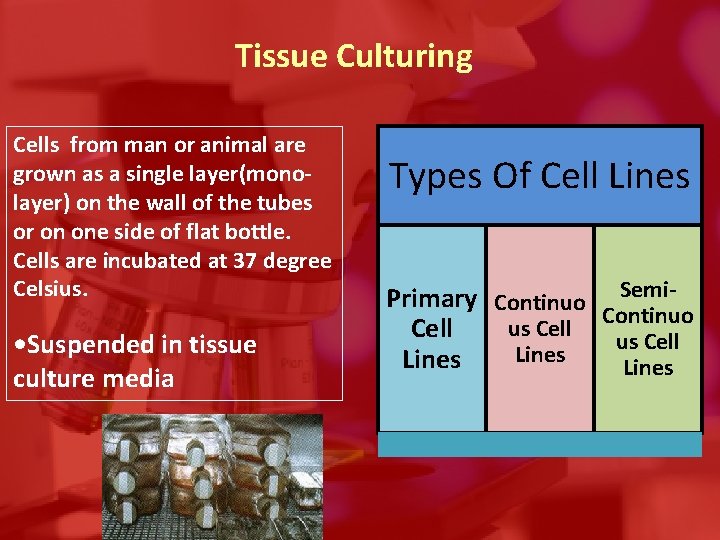
Tissue Culturing Cells from man or animal are grown as a single layer(monolayer) on the wall of the tubes or on one side of flat bottle. Cells are incubated at 37 degree Celsius. • Suspended in tissue culture media Types Of Cell Lines Primary Continuo Semi. Continuo us Cell Lines
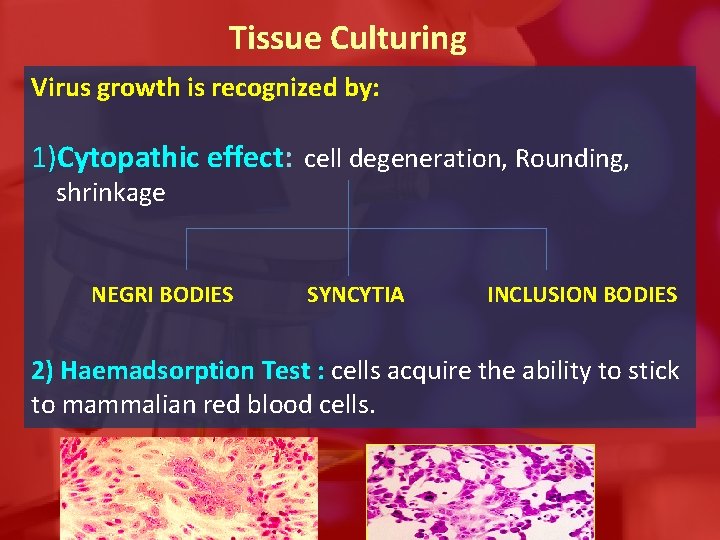
Tissue Culturing Virus growth is recognized by: 1)Cytopathic effect: cell degeneration, Rounding, shrinkage NEGRI BODIES SYNCYTIA INCLUSION BODIES 2) Haemadsorption Test : cells acquire the ability to stick to mammalian red blood cells.
Problems With Tissue Culturing Long period (up to 4 weeks) Poor Sensitivity Susceptible to contamination Not Applicable on all viruses

Chick Embryo Inoculation 1. Chorioallantoic Membrane : Variola and Vaccinia Virus 2. Allantoic Cavity : Influenza Virus and Paramyxo virus 3. Amniotic Cavity: Primary isolation of Influenza virus 7 -12 day old chick is used. Costs less, growth of virus is rapid and easy. The Inoculated eggs are kept at 37 degree for 48 hours till the virus replicates.
Serologic Tests
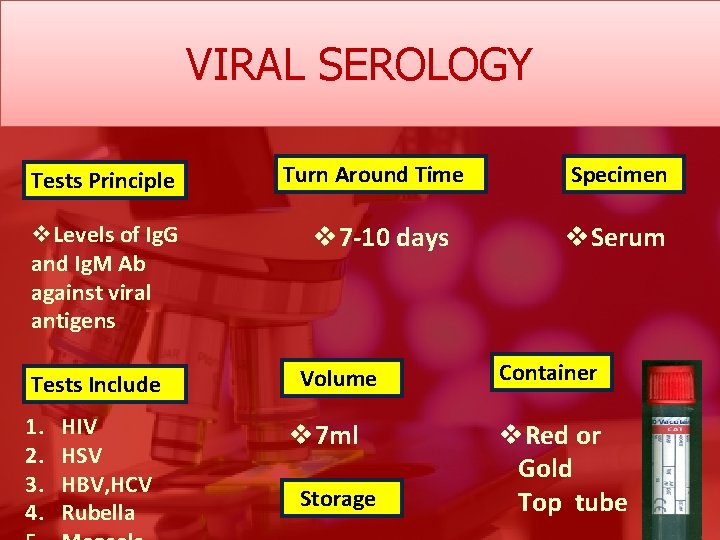
VIRAL SEROLOGY Tests Principle v. Levels of Ig. G and Ig. M Ab against viral antigens Turn Around Time v 7 -10 days Specimen v. Serum Tests Include Volume Container 1. 2. 3. 4. v 7 ml v. Red or Gold Top tube HIV HSV HBV, HCV Rubella Storage
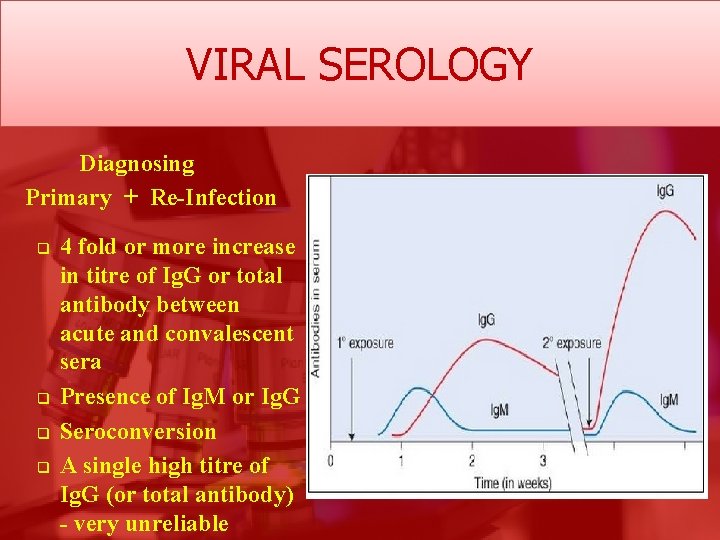
VIRAL SEROLOGY Diagnosing Primary + Re-Infection q q 4 fold or more increase in titre of Ig. G or total antibody between acute and convalescent sera Presence of Ig. M or Ig. G Seroconversion A single high titre of Ig. G (or total antibody) - very unreliable

HEMAGGLUTINATION TEST Purpose : Ø- To quantitate soluble antigens and HI titer in serum samples Ø- Sensitive than CFT, simple, inexpensive, and rapid and is the method of choice for Ø Assaying antibodies to any virus that causes hemagglutination Ø- Commonly used for different strains of influenza viruses, and parainfluenza Ø viruses

PROCEDURE 1. Label plates (8 or 12 wells/row) 2. Add 50 ul PBS to all Wells 3. Add 50 ul AAF to 1 st well 4. Mix & dilute (50 ul) – 1 st through last well 5. Add 50 ul 0. 5% washed rbcs to all wells 7. Mix by shaking plates 8. Read at 20 – 30 min
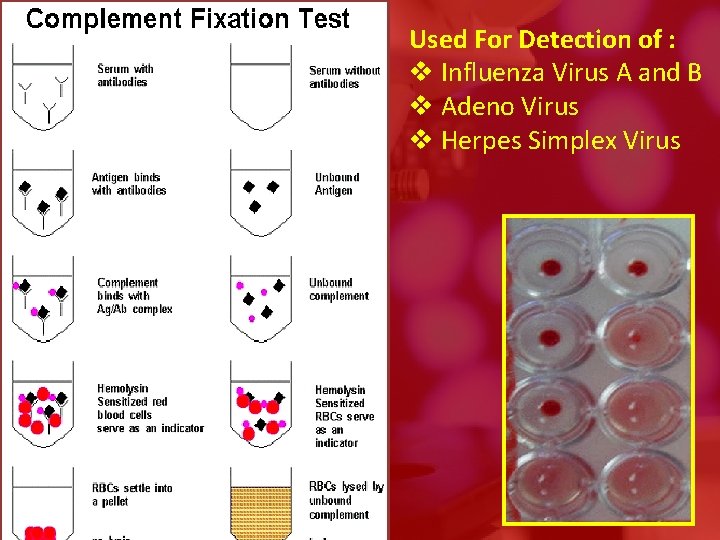
Used For Detection of : v Influenza Virus A and B v Adeno Virus v Herpes Simplex Virus
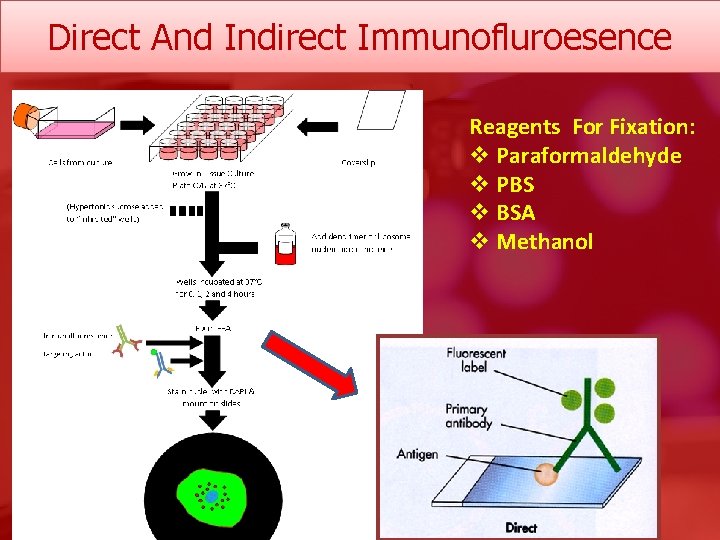
Direct And Indirect Immunofluroesence Reagents For Fixation: v Paraformaldehyde v PBS v BSA v Methanol
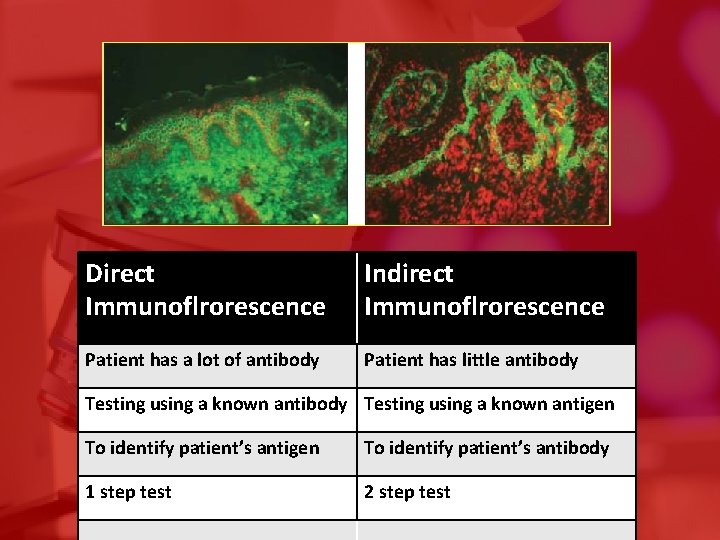
Direct Immunoflrorescence Indirect Immunoflrorescence Patient has a lot of antibody Patient has little antibody Testing using a known antigen To identify patient’s antibody 1 step test 2 step test

Limitations of serological Diagnosis 1. Long length of time required for diagnosis 2. Mild local infections such as HSV genitalis may not produce a detectable humoral immune response 3. Extensive antigenic cross-reactivity between related viruses e. g. HSV and VZV. 4. Immunocompromised patients often give a reduced or absent humoral immune response. 5. Patients given blood or blood products may give a

THANK YOU !
- Slides: 29