Vigi Base Magnus Wallberg Senior Systems Architect M

Vigi. Base Magnus Wallberg Senior Systems Architect M Sc Engineering Physics November 24 th 2009 Dar Es Salaam, Tanzania magnus. wallberg@who-umc. org

Agenda of this block • Who is that guy standing in front of you • • Vigi. Base WHO-ART WHO Drug dictionary Vigi. Search • Live Vigi. Search demo Magnus Wallberg, UMC 09: 00 09: 40 10: 45 11: 30 - 09: 40 10: 15 11: 30 12: 00

Vigi. Base (WHO ICSR database) Magnus Wallberg, UMC

Agenda of this module • Generally about Vigi. Base • Different formats for reporting of ICSRs • Who reports to the UMC? • What to report to the UMC? Magnus Wallberg, UMC
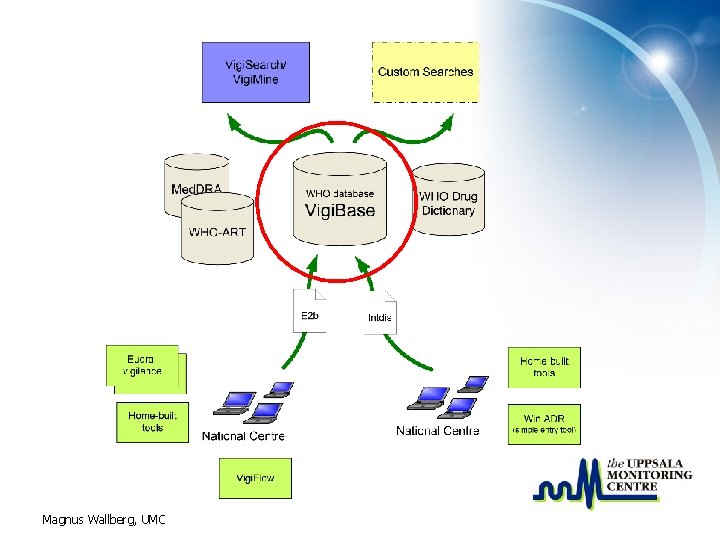
Magnus Wallberg, UMC

Vigi. Base – the database Core of the data collection done within the WHO Drug Monitoring Programme • in use for more than 30 years – Located in Uppsala since 1978 • For spontaneous reports • maintained by the UMC • Relational database with tools for import, maintenance and analysis Magnus Wallberg, UMC
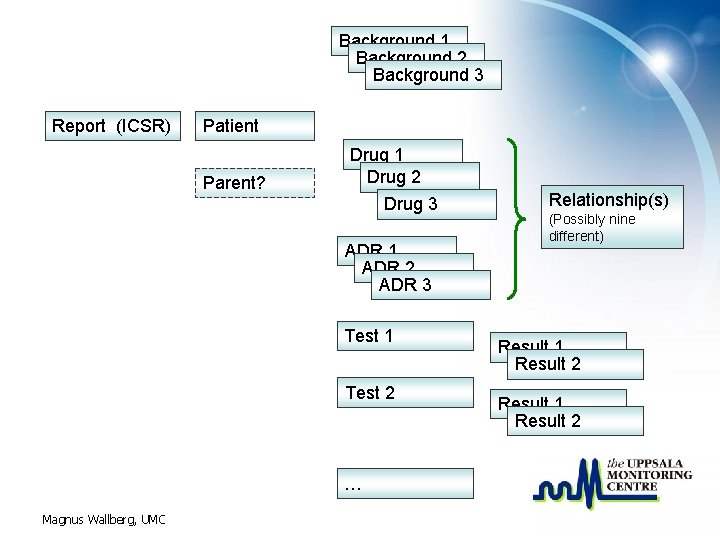
Background 1 Background 2 Background 3 Report (ICSR) Patient Parent? Drug 1 Drug 2 Drug 3 ADR 1 ADR 2 ADR 3 Test 1 Test 2 … Magnus Wallberg, UMC Relationship(s) (Possibly nine different) Result 1 Result 2
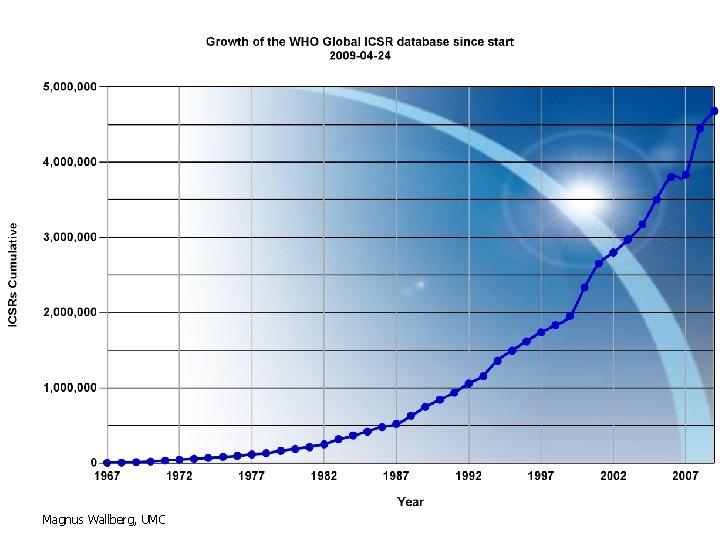
Cumulative number of correct reports processed per year Magnus Wallberg, UMC

Reporting formats Magnus Wallberg, UMC

Reporting formats INTDIS – (International Drug Information System) • 41 countries E 2 B – (ICH standard) • 53 countries So – what’s all this about formats? Magnus Wallberg, UMC

WHO Format Paper formats • ’Yellow cards’ • CIOMS forms None of those are accepted by the UMC • Med. Watch Electronic media • INTDIS format (text files)





E 2 B-format • ICH E 2 B standard (xml files) Format for transferring data between databases of different structure • Guidelines (WHO, EMEA, ICH. . . ) Description of content • DTD (Document Type Definition) Description of file format ICH ; International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use
Magnus Wallberg, UMC

WHO- versus E 2 B-format WHO E 2 B Ø Outcome – case level Ø Causality assessment on case level Ø Limited data on a case Ø Ø Ø Ø Ø All ICSRs are converted into E 2 B format during import to Vigi. Base! Outcome – ADR level Causality assessment on ADR/drug level Parent child / fetus reports Linked reports Literature cases Suspected duplicate Report nullification Lab values Free text fields: Patient medical history, case narrative, labelling info, reporter’s comment, sender’s diagnosis, sender’s comment

What to Report to the UMC? • Serious and Non-serious cases • Conventional medicines ü Prescription ü OTC • Traditional medicines • Biologicals including vaccines Do NOT report: • Veterinary medicines • Cosmetic/hygiene products • Foreign cases Brand name!

Reporting of Brand Names Fluoxetine (Eli Lilly) • FDA - USA Prozac® • MPA - Sweden Fontex®
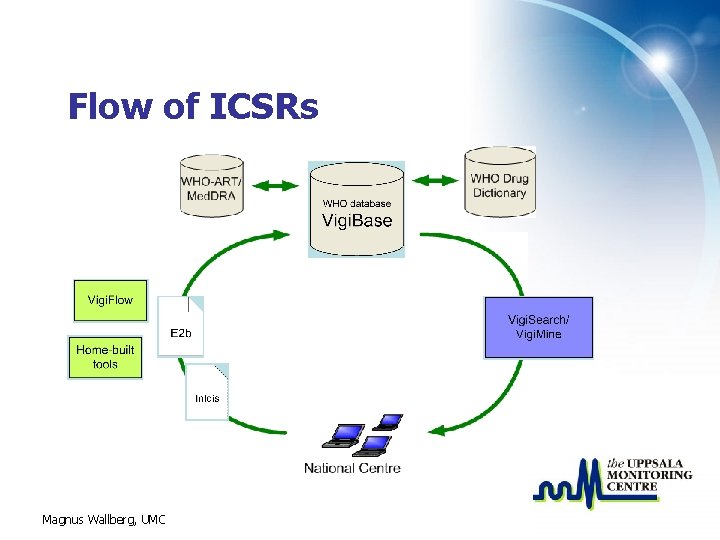
Flow of ICSRs Magnus Wallberg, UMC

Transmitting reports to the UMC is only one of the functions within the Vigi. Flow system Data entry and assessment Search and statistics Submission management But. . . more about that tomorrow!! Report import (E 2 b) Vigi. Flow database Report export (E 2 b) Capability of country wide reporting mechanisms In all a complete management system! Magnus Wallberg, UMC

Some Vigi. Base statistics and comments
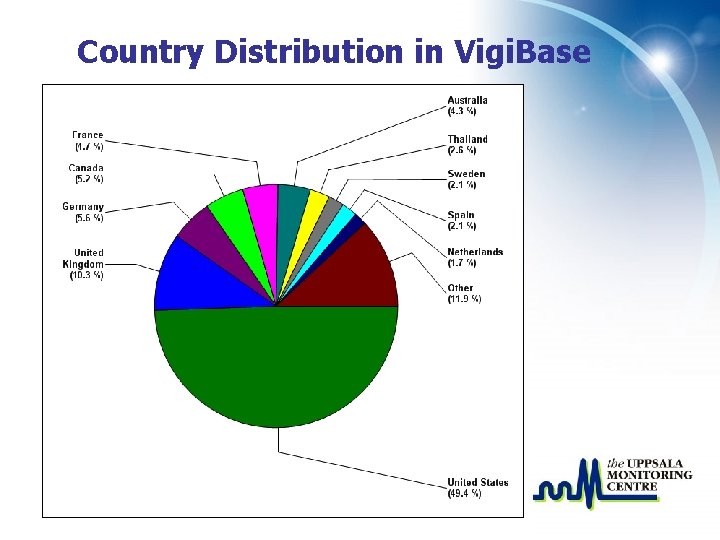
Country Distribution in Vigi. Base
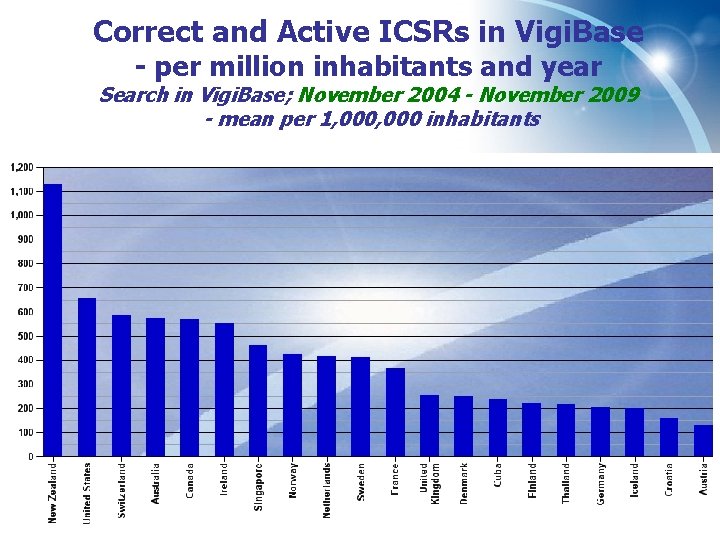
Correct and Active ICSRs in Vigi. Base - per million inhabitants and year Search in Vigi. Base; November 2004 - November 2009 - mean per 1, 000 inhabitants

So why don’t we find all our cases in Vigi. Base when using Vigi. Search? …the report is yet not processed into Vigi. Base …the report is processed but not active in Vigi. Base …the report was rejected due to incorrect format etc.

The UMC Vigis. . . • Vigi. Base – The WHO ICSR database – What was presented here • Vigi. Search – A web based tool to search Vigi. Base data – Separately presented later today • Vigi. Flow – A separate tool for National Centres to manage their ICSRs – Simplifies the process of sending ICSRs to Vigi. Base – Separately presented tomorrow Magnus Wallberg, UMC

Magnus Wallberg, UMC

WHO Collaborating Centre for International Drug Monitoring Box 1051, SE - 751 40 Uppsala Sweden Tel +46 18 65 60 60, Fax +46 18 65 60 88 E-mail: info@who-umc. org Website: www. who-umc. org
- Slides: 29