Veterinary Medicines Directorate Presentation 14 November 2011 Veterinary

Veterinary Medicines Directorate Presentation 14 November 2011

Veterinary Medicines Directorate n VMD became an executive agency of Defra in 2001. n Prior to that it was an agency of MAFF from 1989. n About 160 staff (a mix of specialists and administrators).

Veterinary Medicines Directorate Core business is to protect public health, animal health and the environment by the: n assessment and authorisation of veterinary medicinal products n controls on their manufacture, distribution and use n post-authorisation surveillance schemes n

Veterinary Medicines Directorate n Enforcement unit focusing on illegal substances. n Policy (residues, antimicrobial resistance, OPs, hormonal substances). n Legislative changes (Veterinary Medicines Regulations). n R&D programme. n Finance/IT.

This Talk § Authorisations of Veterinary Medicines § Legislation/Regulation of Veterinary Medicines § Residues Surveillance Programme

AUTHORISATIONS AT THE VMD THERE ARE 2 ASSESSOR TEAMS: § Pharmaceuticals and Feed Additives § Biologicals (mainly assess applications for vaccines)

1. CRITERIA FOR AUTHORISATION There are 3 evaluation criteria: n QUALITY n SAFETY Ø to the user Ø to the consumer (if relevant) Ø to the environment Ø to the target species n EFFICACY These criteria apply to both Pharmaceutical and Biological products

§ There is one difference between the Pharmaceuticals and the Biologicals teams: how they are structured § The Pharmaceuticals team has 4 sub-teams who assess different parts of the dossier: Ø quality (pharmacists & chemists) Ø user safety and consumer safety (toxicologists) Ø environmental safety (ecotoxicologists) Ø efficacy and target species safety (veterinarians) § Each has a senior assessor at the head of the team

§ In the Biologicals team one assessor assesses all parts of the dossier § The assessors are: Ø Microbiologists Ø Biochemists Ø Immunologists Ø Veterinarians

ANY QUESTIONS?

§ A veterinary medicinal product must be authorised before it can be placed on the UK market § It needs a Marketing Authorisation

2. ROUTES OF AUTHORISATION TO OBTAIN A MARKETING AUTHORISATION § § The national procedure European procedures Ø The Centralised Procedure Ø The Mutual Recognition Procedure Ø The Decentralised Procedure

2. 1 THE NATIONAL PROCEDURE (UK ONLY) § Marketing Authorisation issued by the national licensing authority and valid in that Member State only § In the UK the VMD issues national Marketing Authorisations for veterinary medicines

2. 1 THE NATIONAL PROCEDURE (UK ONLY) § Peer review of conclusions at Ø the Scientific Secretariat for pharmaceuticals Ø the Biologicals Committee for biological products § Advice is available (if needed) from the Veterinary Products Committee (independent experts). § Other government agencies also have an input to these committees, for example: Ø The Food Standards Agency, the Health Protection Agency, the Environment Agency etc.

EU AUTHORISATION ROUTES § The VMD plays a full and active part in the following 3 European authorisation routes

2. 2. THE CENTRALISED PROCEDURE (EU INCLUDING THE UK) § Obligatory for products derived from biotechnology § Can also be used for innovatory veterinary medicinal products (e. g. a new active ingredient or a new indication) but does not have to be § Coordinated by the European Medicines Agency (EMA) who are based in London § Assessment is done by experts in Member States § The members of the Committee for Veterinary Medicines Products (CVMP) give opinions § Each Member State has one CVMP member

2. 3 THE MUTUAL RECOGNITION PROCEDURE (EU INCLUDING THE UK) § Mutual recognition of existing national Marketing Authorisations by other Member States § A Marketing Authorisation has been issued by one EU Member State – the Reference Member State (RMS) § The applicant can then apply to other Member States for Mutual Recognition of that Marketing Authorisation § The Committee for Mutual Recognition and Decentralised Procedures (veterinary) [CMD (v)] meeting is where applications are discussed

2. 4 THE DECENTRALISED PROCEDURE (EU INCLUDING THE UK) § This is a relatively new procedure which was introduced in October 2005 § The ‘new’ Decentralised procedure has proved to be very popular with companies and it is rapidly becoming the EU procedure of choice § It is a fast track system to obtain authorisation in wider markets (12 -18 months) § Therefore, the dossier must be of a high standard as there isn’t enough time to answer serious questions/resolve major issues

2. 4 THE DECENTRALISED PROCEDURE (EU INCLUDING THE UK) § The applicant asks one Member State to do the assessment. They are the Reference Member State § All the other Member States involved with the procedure contribute to the evaluation process. They are Concerned Member States § The Committee for Mutual Recognition and Decentralised Procedures (veterinary) [CMD (v)] meeting is where applications are discussed

ANY QUESTIONS?

UNITED KINGDOM PUBLIC ASSESSMENT REPORTS (UKPARS) As a result of new legislation in October 2005, the VMD now has to produce UKPARs. § These are a less detailed version of the assessment reports which are written § Any commercially confidential information is taken out § They are drafted in a style that enables the reasonably educated public to read them

SUMMARIES OF PRODUCT CHARACTERISTICS (SPCS) § An SPC is a document which contains all the basic details about a product § It is approved as part of the authorisation process. Companies have to market their products in accordance with the information in the SPC Also as a result of the new legislation: § SPCs are publicly available on the VMD’s website for all UK authorised products

All authorised veterinary products available in the UK are listed on the VMD’s website: www. vmd. defra. gov. uk Look for Product Information Database A-Z list plus search facility + UKPARs (if available) + SPCs

ANY QUESTIONS?

Regulation of veterinary medicines in the UK

Veterinary Medicines Regulations (VMRs) Current legislation n n VMRs 2011 (S. I. 2159/2009) came into force 1 October 2011 Implementing European Directive 2001/82/EC (as amended) The VMR are backed up with 20 Guidance Notes The current legislation and guidance notes are available on our website www. vmd. defra. gov. uk

Veterinary Medicines Regulations background n n n Veterinary medicines sections of Medicines Act 1968 were replaced by VMRs 2005 in October 2005 (regulating veterinary medicines only) VMRs annually updated (except in 2010) – always to produce one consolidated SI Covering manufacture, marketing, retail & wholesale supply, prescription, administration, import, medicated feeds and more

What does the legislation cover? n. Authorisation of the marketing and manufacture of UK veterinary medicinal products (VMPs) § Pharmaceuticals § Immunologicals (for food and non-food animals) n. Application requirements for the authorisation process for VMPs that are to be marketed within both UK and other EU Member States n. Requirements for mandatory batch release information and Suspected Adverse Reaction reports, to provide ongoing monitoring of products on the UK market

What does the legislation cover? (cont. ) n n n Authorisation of clinical trials on animals for the purpose of developing VMPs - Animal Test Certificates Non-food animal blood banks Manufacture of autogenous vaccines Equine stem cell centres Requirements for wholesale distribution of VMPs under Good Distribution Practice (GDP) Requirements for the manufacture and distribution of medicated feed additives and premixtures

What does the legislation cover? (cont. ) n n Criteria for exemption from the need to have a Marketing Authorisation for certain medicines for small pet animals – including reptiles, rodents, ornamental fish Sets out the 4 different legal distribution categories of VMPs - POM-V, POM-VPS, NFA-VPS and AVM-GSL and indicate who can supply each of them Restrictions on the legal possession and administration of VMPs within these categories (including the Cascade) The requirements for the qualification and premises registration of VMP retailers

What does the legislation cover? (cont. ) n Importation schemes n n Requirements for the issue of import certificates to allow the legal importation of unauthorised medicines into the UK Restrictions on advertising n n Prescription medicines cannot be advertised to the general public All advertisements should be consistent with the product’s indications and its approved Summary of Product Characteristics (SPC)

What does the legislation cover? (cont. ) n Enforcement n n n Lack of compliance with the legislation: enforcement action is graduate and proportionate to the risks associated First step: guidance and, if necessary, a written warning If the problem is serious or persistent: Improvement Notice and/or a Seizure Notice, confiscation of goods or suspension or revocation of an authorisation Non-compliance with an Improvement Notice can result in prosecution Penalties for offences are fines or imprisonment

Distribution Categories n § § § POM-V – Prescription Only Medicine - veterinarian POM-VPS - Prescription Only Medicine – Vet, Pharmacist, SQP NFA-VPS – Non-Food Animal – Vet, Pharmacist, SQP AVM-GSL – Authorised Veterinary Medicine – General Sales List Only a Registered Qualified Person (RQP)can supply medicines above AVM-GSL Small Animal Exemption Scheme (SAES) products – not for food producing animals

Certification and Qualification n What is a Registered Qualified Person (RQP)? n Veterinary Surgeon (RCVS) n Pharmacist (GPh. C) n Suitably Qualified Person (AMTRA) § Agricultural merchant § Pet shop § Saddler § Possibility for others (e. g beekeepers)

Prescribing, Dispensing and Supply Who can prescribe, dispense and supply? § Veterinary Surgeons n Prescribe and supply all categories of veterinary medicine n Prescribe medicines to be used under the cascade n Can dispense prescriptions written by another veterinary surgeon § Pharmacists § SQPs § Any retailer n Prescribe POM-VPS; Supply NFA-VPS; AVM-GSL, SAES n Can dispense prescriptions written by a veterinary surgeon n Prescribe POM-VPS; Supply NFA-VPS; AVM-GSL, SAES n Supply AVM-GSL, SAES products

RQP Responsibilities at the time of supply: to ensure n n n The medicine is appropriate for the animal/s and condition to be treated – (e. g. if POM-VPS, ask questions about last treatment, animal weight etc. ) The client understands how to use the product safely (e. g. read warnings, wear gloves if directed, etc) The medicine is prepared correctly The medicine is labelled correctly if not in the original authorised packaging The minimum amount required for treatment is given (except where packs can’t be split)

Prescribing Cascade If there is no authorised veterinary medicinal product in the MS for a condition affecting a (food/non food) species, by way of exception, the veterinarian responsible may, under his/her direct personal responsibility and in particular to avoid causing unacceptable suffering treat the animal concerned with:

Why is the cascade needed? n n Very good reasons to use authorised veterinary medicines Recognition of availability problem Cascade permits a controlled freedom of clinical judgement Cascade is a risk-based model for using unauthorised medicines

Level 1 VMP authorised in the UK for another species, or another condition in the same species Level 2 n Level 3 Extemporaneous preparations, made on a one off basis by an authorised person (and third country imports – STC for import and use) n Human medicine authorised in the UK VMP authorised in another member state (SIC required for import and use)

Key Points on the Cascade n n n Use of imported products: vets are allowed to import unauthorised veterinary medicines from within the EU and for Third countries for use under the Cascade in ‘exceptional circumstances’ An import certificate issued by the VMD granting permission for the import is required in each case Veterinary Medicines Guidance Note 7 contains more detail on Import Certificates

Review of EU Directive n n n In May 2010 the Commission published a questionnaire to gather feedback on the current Directive The next step will be the publication of an Impact Assessment looking at specific areas of EU legislation on veterinary medicines and how they can be improved We held a workshop with stakeholders in January 2010 to discuss this review and hear their views.

Review of EU Directive (cont’d) n The main topics covered by the review are: 1. Data exclusivity 2. Authorisation procedure 3. Packaging and labelling 4. Pharmacovigilance 5. The distribution channel 6. The use of drugs not in accordance with the summary of the product characteristics (off-label use) 7. Harmonisation of already authorised veterinary products 8. New needs, new challenges Full details on VMD website: www. vmd. gov. uk n

Veterinary Medicines Guidance Notes (VMGN) www. vmd. defra. gov. uk/public/vmr_vmgn

VMD Residues Surveillance Eric Crutcher e-mail: e. crutcher@vmd. defra. gsi. gov. uk #282822 April 2010

The Key Messages 1 Good planning processes based on risk analysis to reflect your country’s production. 2 Effectiveness of the programme relies on a well defined sampling strategy. 3 Robust audit trail so action can be taken on non-compliant samples to stop future problems.

Aims of the Control Plan n To detect use of illegal substances as set out in Annex I of Council Directive 96/23/EC. n To ensure that medicines are used in accordance with their authorisation and residues are below the MRL. n Identifying causes of residues in foods.

Structure of NRCP n Council Directive 96/23/EC requires statutory national surveillance for: n n n n Red meat Poultry meat Aquaculture Milk Eggs Wild and farmed game Rabbit Honey

Statutory Surveillance for Veterinary Residues Annexes I and II of 96/23/EC set out the groups veterinary residues that Member States are obliged to look for: Group A n n n stilbenes thyrostats steroids zeranol beta agonists prohibited substances Group B n n n n n antimicrobials coccidiostats anthelmintic agents carbamates and pyrethroids NSAIDs organochlorines and PCBs organophosphates heavy metals mycotoxins dyes (including malachite green)

Commission Decision 97/747/EC n Fixes the levels and frequencies of sampling required by 96/23; n Sets out number of samples required according to national production (e. g. 1 sample per 15, 000 tonnes of milk production, with minimum of 300 samples); n It must be possible to trace sample back to the farm of origin.

Commission Decision 98/179 n Sets out rules on official sampling; n Sample must be targeted, based on criteria provided; n Suspect samples should be taken from farm of origin when routine sample analysed under Council Directive 96/23 is non-compliant.

Planning the National Residue Control Plan n Work starts on drafting the plan in July for following year; n Draft plan considered by planning group in September; n Aim to have plan agreed by end of October, to enable sampling to start in January; n Samples taken throughout the year, unless sensible to target a particular time of year.
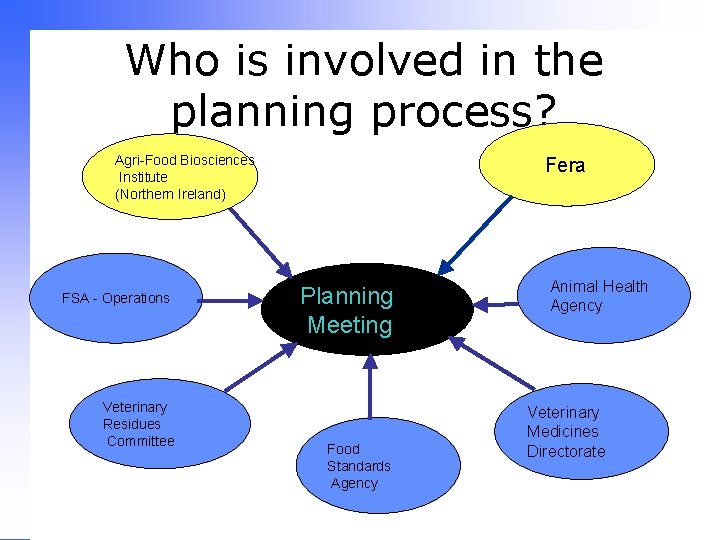
Who is involved in the planning process? Agri-Food Biosciences Institute (Northern Ireland) FSA - Operations Veterinary Residues Committee Fera Planning Meeting Food Standards Agency Animal Health Agency Veterinary Medicines Directorate

What information do we use? n n n UK surveillance results New MRLs New marketing authorisations Which veterinary medicines are used Possible use of illegal products Use of banned substances in Europe

Sample Allocation n National Resides Control Plan (NRCP) shows the number of samples required for all analytes; n plan is entered on database; number of samples allocated to abattoirs according to throughput; n unique sample number on RIM form and label for bag.
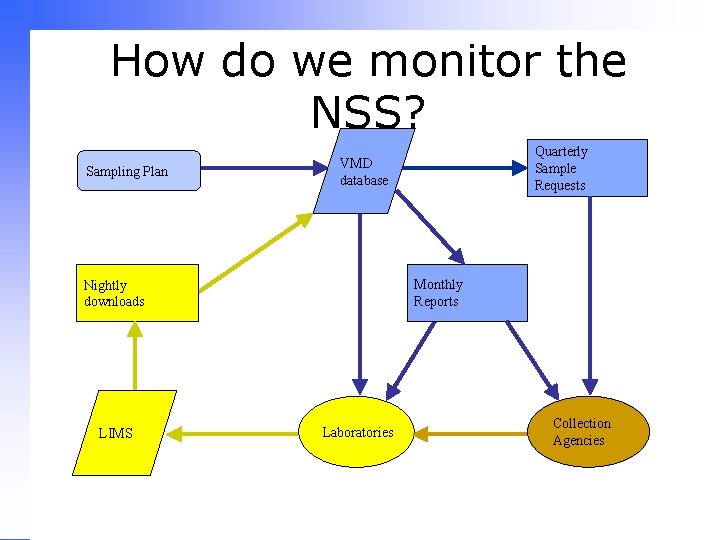
How do we monitor the NSS? Sampling Plan Monthly Reports Nightly downloads LIMS Quarterly Sample Requests VMD database Laboratories Collection Agencies

Who undertakes the analyses? n Laboratories providing analytical services to the VMD operate to internationally accepted standards, use validated methods and participate in routine proficiency testing; n Laboratories are included in the inspections carried out by the European Commission Food and Veterinary Office (FVO); n Laboratories are designated as National Reference Laboratories (NRL) for certain substances/residue groups.

Traceability n n VMD issues sampling request form for every sample (RIM 1); Requests type of animal, sex of animal, analyte; Sampling officer fills in middle section; Laboratory does not know origin of sample.

Reporting of results to Competent Authority n Laboratory contract requires screening analysis within 28 days; n Laboratory informs VMD within 24 hours of non-compliant screening result; n Non-compliant screening results confirmed within next 10 days; n Nightly electronic download of results from LGC to VMD.

What action do we take on non-compliant samples? We take action where we find residues of: n n Authorised substances above the MRL; Unauthorised substances. We can: n n n Ask the Animal Health Agency / Fisheries agencies / National Bee Unit to advise the farmer; Stop animals being slaughtered; Prosecute in serious cases.

ANY QUESTIONS?
- Slides: 60