Veterinary Biological Products 4 H Veterinary Science Extension

Veterinary Biological Products 4 -H Veterinary Science Extension Veterinary Medicine Texas Agri. Life Extension Service College of Veterinary Medicine and Biomedical Science Texas A&M System http: //aevm. tamu. edu

Objectives n Describe modified live vaccines n Describe killed vaccines of bacterins n Discuss proper handling of vaccines n Discuss proper vaccination procedures

Infectious Vaccines (Antigens) n Types n Modified live vaccines – intramuscular, subcutaneous, intranasal n Modified live vaccines (body temperature sensitive) – intranasal

n Immunity duration n Life-time (infection established, cellular and humoral immunity) n One dose required n Annual boosters may not be required

n Caution n Label restrictions (may be unapproved for pregnant cows/nursing calves) n Ineffectiveness (following non-infectious vaccine)

Non-infectious Vaccines (Antigens) n Types n Killed vaccines and bacterins – intramuscular, subcutaneous n Toxoids and leukotoxoids – intramuscular, subcutaneous n Modified live vaccines (body temperature sensitive) – intramuscular

n Immunity duration n 4 to 12 months (infection not established, humoral immunity) n Primary dose required (priming) n Secondary dose required – 3 to 6 weeks later (within 4 months) n Annual or semiannual boosters required

Antisera /Antiserum (Antibodies) n Types n Antitoxins – subcutaneous n Monoclonal antibody – oral
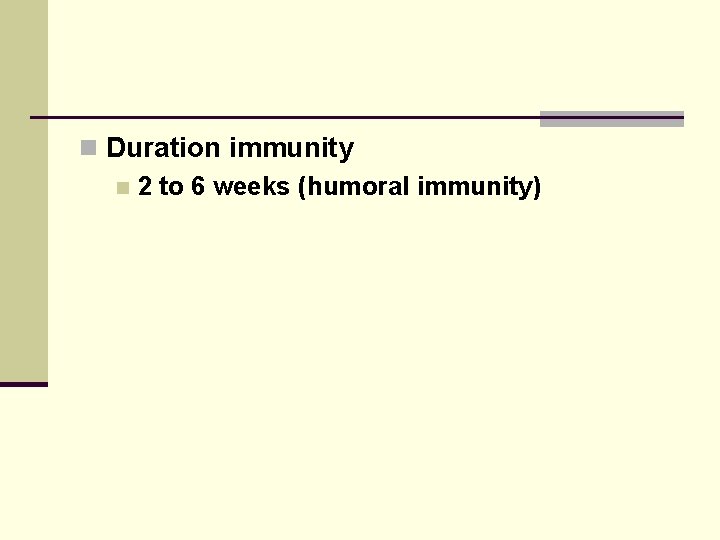
n Duration immunity n 2 to 6 weeks (humoral immunity)

Vaccine Label Handling Instructions n Follow the label n Refrigerate in use and storage n Use sanitary practices in use n Keep out of sunlight in use n Properly dispose of opened vials after use n Abide expiration date of unopened vials

Vaccine Label Use Directions n Follow the label n Dose n Route(s) n Withdrawal time for slaughter (21 to 60 days)

Vaccine Failures n No response (efficacy, stress, malnutrition) n Improper dose n Improper route

n Mishandled (non-refrigerated, sunlight exposure, unsanitary) n Stored opened vials n Expired vaccine n Poor quality vaccine

n Primary dose only (non-infectious vaccine) n Immunity duration expired n Vaccination after exposure (incubation period or latent period)
- Slides: 14