Very weak monohydroxide bases These bases Ka values

Very weak monohydroxide bases: These bases Ka values are lower than 10 -8 M. For this reason, it hasn’t any strong OH- that titratable in water media. But, it will be titratable with some strengthening compound. Anilin and pyridine are examples to these bases. This kind of bases should be titratable in non-water media. For calculation of p. OH, OH- from water has not omitted. Relations between Ka and Kb Ka x Kb = Kw

In Diprotic acids: Ka 1 x Kb 2 = Kw Ka 2 x Kb 1 = Kw p. H Calculations in Salt Solutions p. H of salts are calculated related to form of salts. Hyrolysis is significant in calculations. Generally salts are divided to 4 group. a) Established from strong acids + strong bases (For example; Na. Cl: HCl + Na. OH) b) Established from strong acids + weak bases (For example; NH 4 Cl: HCl + NH 4 OH)

c) Established from weak acids + strong bases (For example; CH 3 COONa: Na. OH + CH 3 COOH) d) Established from weak acids + weak bases (For example; CH 3 COONH 4: NH 4 OH + CH 3 COOH) Established from strong acids + strong bases (For example; Na. Cl: HCl + Na. OH) When these types of salts are hydrolysis, equal amount and equal strength of [OH-] and [H+] are occurred. Na. Cl + H 2 O → Na+ + Cl- +OH- + H+ Consequently, the media is neutral and p. H = 7. p. H is not changed with concentration.
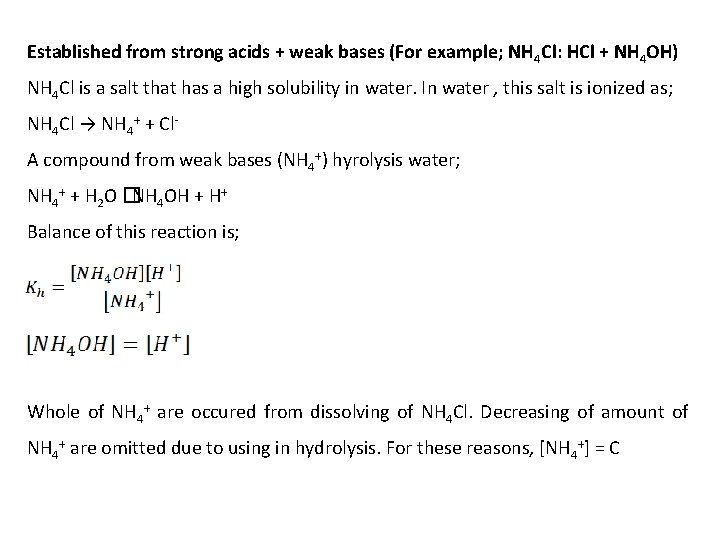
Established from strong acids + weak bases (For example; NH 4 Cl: HCl + NH 4 OH) NH 4 Cl is a salt that has a high solubility in water. In water , this salt is ionized as; NH 4 Cl → NH 4+ + Cl. A compound from weak bases (NH 4+) hyrolysis water; NH 4+ + H 2 O � NH 4 OH + H+ Balance of this reaction is; Whole of NH 4+ are occured from dissolving of NH 4 Cl. Decreasing of amount of NH 4+ are omitted due to using in hydrolysis. For these reasons, [NH 4+] = C

Established from weak acids + strong bases (For example; CH 3 COONa: Na. OH + CH 3 COOH) CH 3 COONa is a salt that has a high solubility in water. In water, this salt is ionized as; CH 3 COONa CH 3 COO– + Na+ A compound from weak acid (CH 3 COO–) hyrolysis water; CH 3 COO– + H 2 O � CH 3 COOH + OH-
![Balance of this reaction is; [CH 3 COOH] = [OH-] Whole of CH 3 Balance of this reaction is; [CH 3 COOH] = [OH-] Whole of CH 3](http://slidetodoc.com/presentation_image/55c4a7917e736e06bf8192455e659fd7/image-6.jpg)
Balance of this reaction is; [CH 3 COOH] = [OH-] Whole of CH 3 COO– are occured from dissolving of CH 3 COONa. Decreasing of amount of CH 3 COO– are omitted due to using in hydrolysis. For these reasons, [CH 3 COO–] = C

Established from weak acids + weak bases (For example; CH 3 COONH 4: NH 4 OH + CH 3 COOH) CH 3 COONH 4 is ionized in water as % 100. CH 3 COONH 4 CH 3 COO– + NH 4+ As we know that CH 3 COO– from weak acid and NH 4+ from weak base, these two compounds hydrolyse water. After necessary omitting; If the strength of a weak acid and a weak base are same, than p. H is equal to 7. But, if this is not, p. H is in area that which one is strong.

p. H CALCULATIONS IN PROTONATED SALTS Protoned salts still have a proton for releasing to experimental media. Na 2 HPO 4, Na. H 2 PO 4, Na. HCO 3 are examples to them. Generally, they are shown as Na. HA and this is a salt of an acid H 2 A is ionized in two steps. H 2 A � HA– + H+ Ka 1 HA– � A– + H+ Ka 2 Na. HA in water; HA– + H 2 O � H 2 A + HA– + H 2 O � H 3 O+ + A 2– Balance of these, the upper and bottom parts of fractions is multiply and divided by [H+] [OH-] and after necessary omitting;
![C = [H 2 A] + [HA–] + [A 2–] [H+] + [N C = [H 2 A] + [HA–] + [A 2–] [H+] + [N](http://slidetodoc.com/presentation_image/55c4a7917e736e06bf8192455e659fd7/image-9.jpg)
C = [H 2 A] + [HA–] + [A 2–] [H+] + [N ] = [HA–] + 2[A 2–] + [OH-] After necessary omitting and shortinings; For Salts derived from a weak acid structure as H 3 A (for example: H 3 PO 4) Ionization balance of H 3 A; H 3 A � H 2 A– + H+ H 2 A– � HA 2– + H+ HA 2– � A 3– + H+

After necessary shortinings, For Na 2 HA (Because; HA 2– was existed in balances of 2 nd and 3 rd) For Na. H 2 A (Because; H 2 A– was existed in balances of 1 st and 2 nd) If there is not omitting, starting concentration should be add to calculations and for Na 2 HA (for example; Na 2 HPO 4) For Na. H 2 A (for example: Na. H 2 PO 4;
- Slides: 10