Ventilatory support in asthma and COPD By HP

Ventilatory support in asthma and COPD By HP Shum 3 Mar 2006
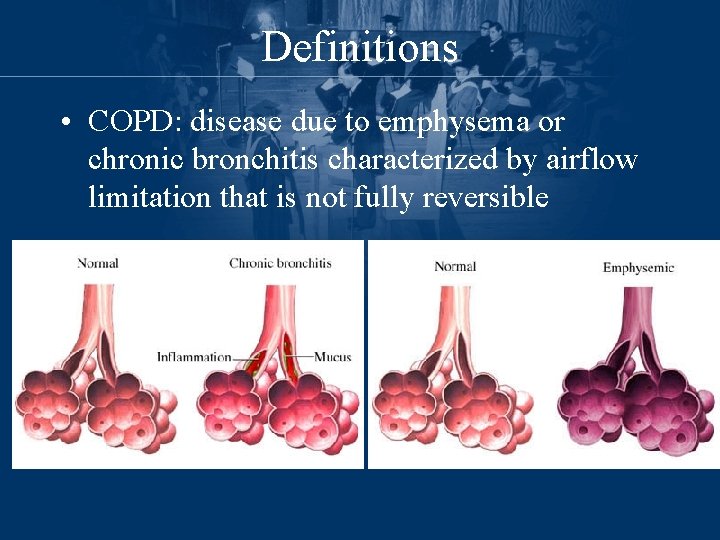
Definitions • COPD: disease due to emphysema or chronic bronchitis characterized by airflow limitation that is not fully reversible

Definitions • Asthma: an inflammatory condition in which complex cellular, chemical, and nervous system (sympathetic, cholinergic and nonadrenergic-noncholinergic) mediators lead to heightened bronchial responsiveness and episodic, variable and reversible airflow obstruction
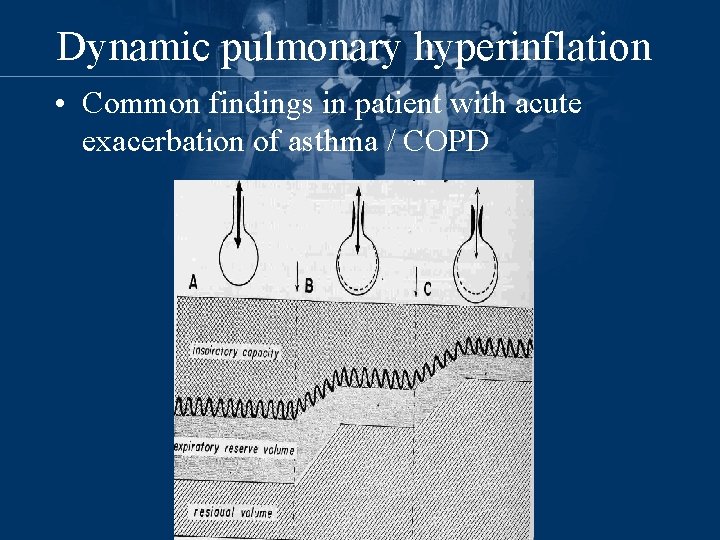
Dynamic pulmonary hyperinflation • Common findings in patient with acute exacerbation of asthma / COPD

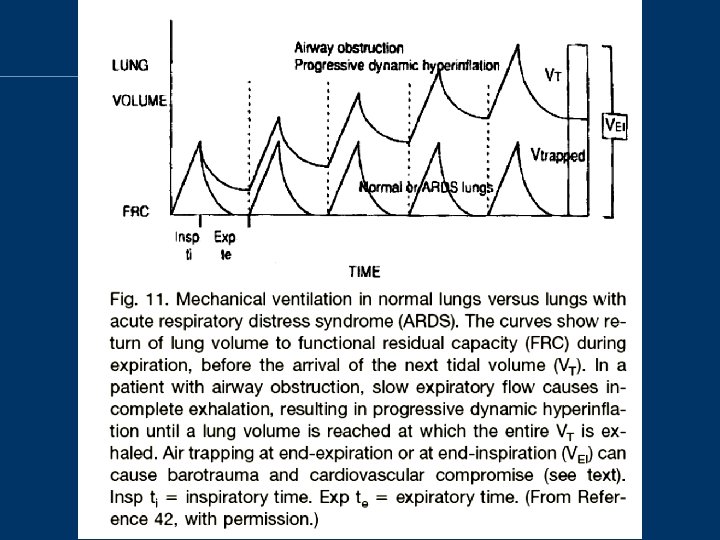
Dynamic pulmonary hyperinflation • Why this occur? – Increased ventilatory requirement – Prolong expiratory time constant due to increased airway obstruction by inflammation, mucus plug, bronchospasm, with or without reduced elastic recoil – Shortening of expiratory time Am Rev Respir Dis 1989, 139: 242– 246 Eur Respir J 1997, 10: 1663– 1674

Dynamic pulmonary hyperinflation • Resulting in: – Increased inspiratory threshold load for breath initiation – Increased work of breathing Intensive Care Med 2001, 27: 166– 178

B A C

Other factors • The capacity of the respiratory muscles to generate inspiratory pressure is limited by decreased operating length and impaired geometric arrangement Clin Chest Med 2000, 21: 679– 692 • Long-term steroid use and/or malnutrition contributes to strength impairment in many patients with severe chronic disease Am Rev Respir Dis 1996; 153: 610– 615 • Blunted respiratory drive decrease responsiveness to hypoxia and hypercapnia N Engl J Med 1994, 330: 1329– 1334

Pharmacological Management • Bronchodilator – 2 -adrenergic receptor agonists causes bronchodilation via c. AMP-mediated relaxation of bronchial smooth muscle (Albuterol, Terbutaline) – Anticholinergics mediated by blocking formation of c. GMP (Ipratropium bromide, tiotropium)

Pharmacological Management • Corticosteroids – Reduce airway inflammation, airway responsiveness, mucus secretion and airway edema. Also enhance -adrenergic responsiveness and relax bronchial smooth muscle (beclomethasone, budesonide, hydrocortisone, methylprednisolone)

Pharmacological Management • Methylxanthines – Improve respiratory drive and muscle function, weak bronchodilator. Act by non-specific inhibition of phosphodiesterase to increase intracellular c. AMP, blockage of adenosine and release of endogenous catecholamines (theophylline, aminophylline)

Pharmacological Management • Antibiotics – COPD • Airway colonization with potential pathogens • High risk of progress to more severe bronchitis / pneumonia which is poorly tolerated in those critically ill COPD • Antibiotic use recommended Expert Rev Anti Infect Ther. 2006 Feb; 4(1): 101 -24 Treat Respir Med. 2005; 4(3): 153 -67 – Asthma • Usually no chronic bacterial colonization • Use of antibiotic should based on clear evidence of infection only

Benefits of mechanical ventilation • • • Assist or support impaired ventilatory function Reduce the work of breathing Allow muscle resting Facilitate sleeping Improve gas exchange Allowing time for restoration of ventilatory function through treatment of either the underlying disease or the precipitating causes of acute decompensation

Ventilation in COPD and Asthma • Even though the pathogenesis and clinical course of COPD and asthma differ, their management is similar in many aspects and this is especially true on the ventilatory support

NIPPV for COPD exacerbation • Consensus statement from the American Association for Respiratory Care Respir Care 1997; 42: 361 • Suggest early use of NIPPV in setting when at least 2 of the following occur: – Respiratory distress with moderate to severe dyspnea – Arterial p. H less than 7. 35 with Pa. CO 2 above 45 mm. Hg (6 k. Pa) – Respiratory rate of 25/minute or greater


NIPPV for COPD exacerbation • Keenan SP et al: Crit Care Med 1997 Oct; 25(10): 1685 -92 – Meta-analysis of use of NIPPV in those with acute resp. failure – 7 RCTs
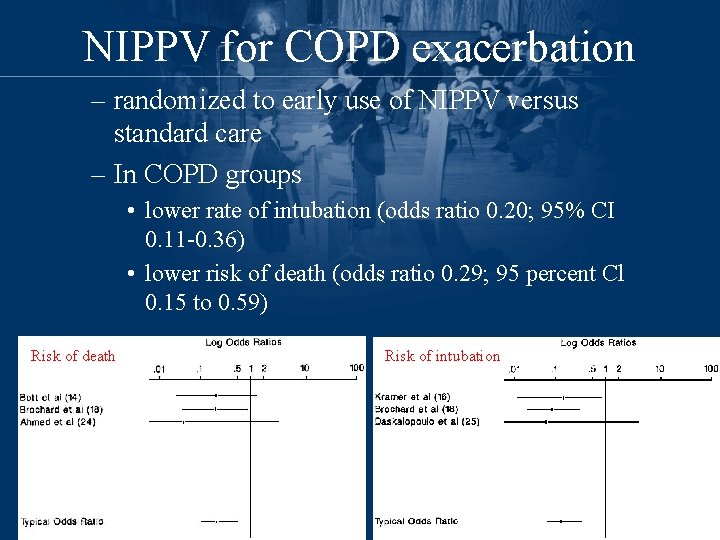
NIPPV for COPD exacerbation – randomized to early use of NIPPV versus standard care – In COPD groups • lower rate of intubation (odds ratio 0. 20; 95% CI 0. 11 -0. 36) • lower risk of death (odds ratio 0. 29; 95 percent Cl 0. 15 to 0. 59) Risk of death Risk of intubation

NIPPV in asthma exacerbation • Cochrane Database Syst Rev. 2005 Jul 20; (3): CD 004360 – Only RCT for adult included, those with features of COPD were excluded – Up till 4/2004, only 1 RCT obtained – The application of NPPV in patients suffering from status asthmaticus, despite some interesting and very promising preliminary results, still remains controversial. Large, prospective, randomised controlled trials are therefore needed to determine the role of NPPV in status asthmaticus

NIPPV in asthma exacerbation • Soroksky A et al: Chest 2003; 123: 1018 -1025 – Randomised double-blind, placebo controlled trial conducted in the emergency department of the Asaf Harofe Medical Center, Zerifin, Israel – Inclusion criteria: FEV 1 < 60% of predicted; respiratory rate > 30 breaths/min; Hx of asthma >= 1 year; duration of current asthma attack of > 7 days – Exclusion criteria: smoking history of > 10 years; known chronic pulmonary disease other than asthma; intubation for CPR; haemodynamic instability (HR>=150 bpm / SBP <=90 mm. Hg), decreased GCS, CHF, IHD, pregnancy, facial deformity, lung infiltration due to APO / pneumonia

NIPPV in asthma exacerbation • Soroksky A et al: Chest 2003; 123: 1018 -1025 – NIPPV: control (nebulized salbutamol / ipratropium + IV steroid) = 17: 16 – Duration of tx: 3 – 4 hrs – NPPV was applied through a nasal mask (Bi. PAP model ST; Respironics) – IPAP start at 8 cm. H 2 O, increased gradually by 2 cm. H 2 O q 15 min to max 15 cm. H 2 O, or until RR< 25 breaths/min (mean: 13) – EPAP set at 3 cm. H 2 O, increased gradually by 1 cm. H 2 O q 15 min to max 5 cm. H 2 O (mean: 4) – Primary end point: increase of FEV 1 >50% baseline – Secondary end point: hospitalization rate
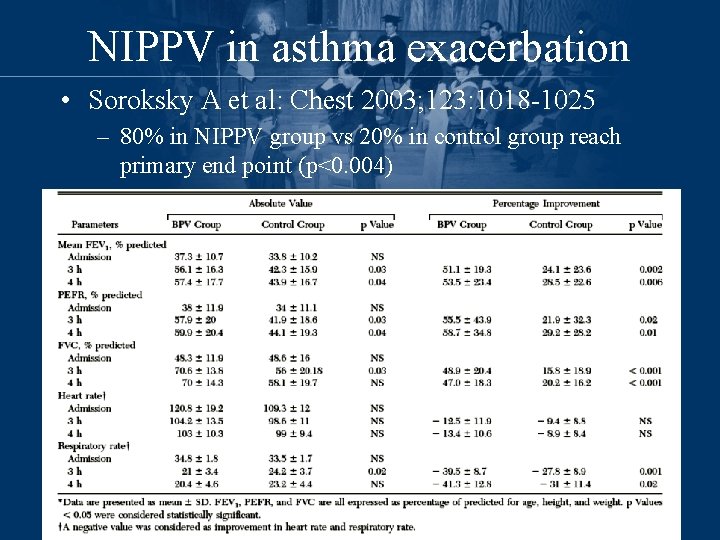
NIPPV in asthma exacerbation • Soroksky A et al: Chest 2003; 123: 1018 -1025 – 80% in NIPPV group vs 20% in control group reach primary end point (p<0. 004)

NIPPV in asthma exacerbation – Hospitalization was required for 3 of 17 patients (17. 6%) in the NIPPV group, as compared with 10 of 16 patients (62. 5%) in the control group (p = 0. 0134). – Concluded that: In selected patients with a severe asthma attack, the addition of NIPPV to conventional treatment can improve lung function, and significantly reduce the need for hospitalization

NIPPV in asthma exacerbation • No clear guideline for use of NIPPV in asthma • Selection criteria may include: – Moderate to severe dyspnea – RR >=25 – Accessary muscle use or paradoxical breathing – Presence of hypercapnic acidosis Am J Respir Crit Care Med 2001; 163: 540 -77
Intubation in asthma • Rapid sequence intubation should be performed by the most experienced operator • Manipulation of the airway in the asthmatic can worsen the bronchospasm or provoke a laryngospasm

Intubation in asthma • Pretreatment with atropine and/or topical anesthetics at the level of the hypopharynx and glottis • The largest endotracheal tube possible should be chosen to lower airflow resistance and permit suctioning of thick mucosal secretions • A cuffed endotracheal tube is sometimes useful even in small children (<5 years) when insufflation pressures become very high

Induce hypotension through a combination of direct vasodilation, histamine release, and vagally-mediated bradycardia.

Post-intubation • Hypotension very common in severe asthmatic after intubation – Sedative use – Hypovolemia (increased sweating and decreased water intake) – Pneumothorax – Use of PEEPe – Dynamic hyperinflation (gas trapping) • delivering of 2– 3 breaths/min of 100% oxygen for few minutes or disconnect from ambu-bag / ventilator • in the absence of pneumothorax, the mean intrathoracic pressure will fall, systemic blood pressure will rise, pulse pressure will increase, and pulse rate will fall Am J Respir Crit Care Med 1995, 151: 1296 -1316

Principles of ventilation • Minimization of dynamic pulmonary hyperinflation – Controlled hypoventilation / permissive hypercapnic ventilation – Prolonged expiratory time – Unload breathing effort – Promote synchrony between patient and ventilator

Controlled hypoventilation • Minute volume, expiratory time fraction, and severity of airway narrowing are primary determinants of dynamic hyperinflation • Decrease minute ventilation – lessens the expiratory flow requirement – allowing the lung to decompress – peak static lung pressures (plateau pressures) to decline

Controlled hypoventilation • Aimed to achieve plateau pressure <=30 cm. H 2 O, moderate hypercapnia (aim Pa. CO 2<=90 mm. Hg / 12 KPa) and acidosis (aim p. H >=7. 2) Curr Opin Pulm Med 1998, 4: 4– 8 Crit Care Clin 1998, 14: 685– 705
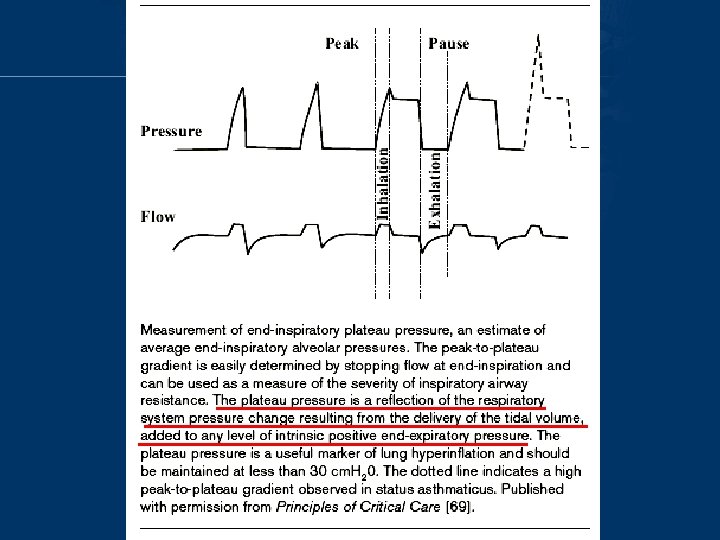

Prolonged expiratory time • Achieved by: – shorter inspiratory time fraction – using a higher peak inspiratory flow setting (70 – 100 L/min) – eliminating inspiratory pause time • However, lengthening of the expiratory period is only modestly effective if minute ventilation remains the same

Unload breath effort • Gas trapped at the end of expiration exerts a positive pressure on the alveoli – PEEPi or auto-PEEP • 41% of inspiratory muscle effort was expended to overcome PEEPi in patients with COPD during spontaneous breathing Am J Respir Crit Care Med 1996, 54: 1301– 1309

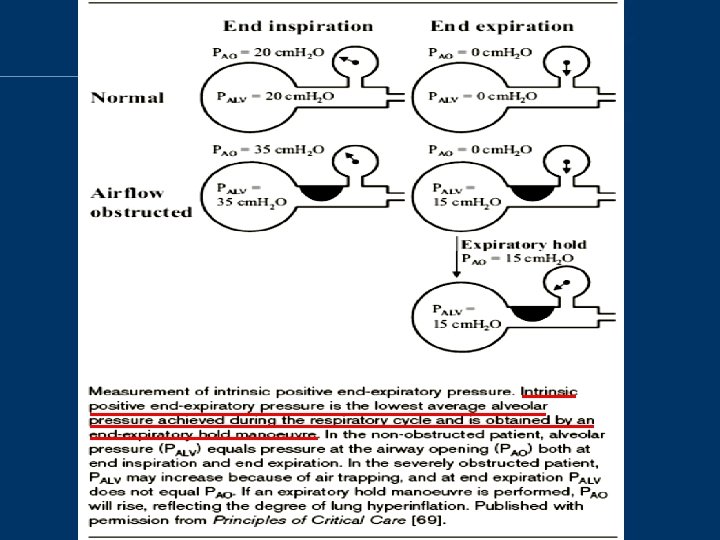

Unload breath effort • Adding PEEPe in COPD may attenuate the inspiratory muscle effort needed to trigger inspiration and improving patient-ventilator interaction • If PEEPe is set higher than PEEPi, it may worsen the dynamic hyperinflation (esp. true in those asthmatics) • Aim 85% PEEPi
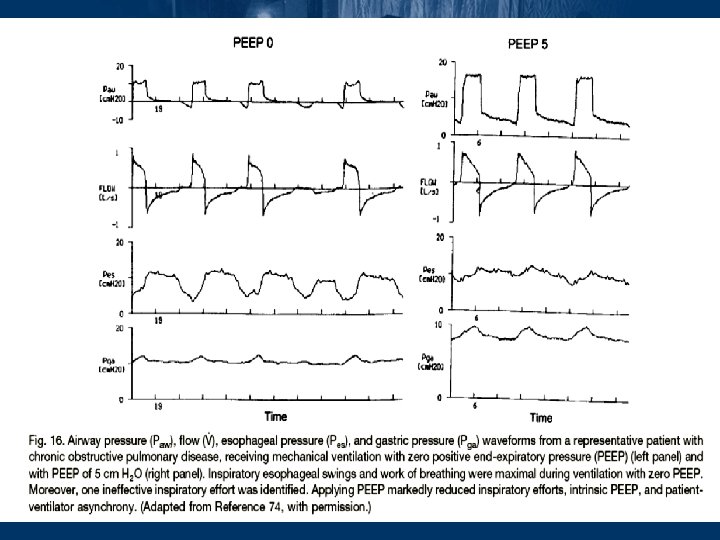

Promote synchrony between patient and ventilator • Most patients take shallow quick breaths and become agitated shortly after intubation because of dyspnea and the discomfort of invasive ventilation Clin Chest Med 1996, 17: 577– 590 • Controlled hypoventilation also required good sedation +/- NMB to achieve • NMB + high dose steroid risk of myopathy • NMB should be used only for short periods (<24 hr) and only as absolutely necessary Chest 1999, 115: 1627– 1631
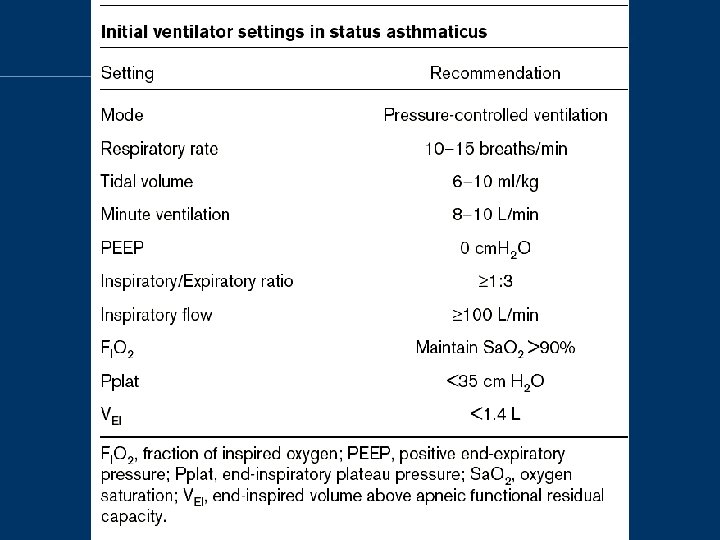


• Correction of hypoxemia is first priority • In status asthmaticus, low V/Q ratio mainly due to mechanical problem and true shunt does not contribute significantly to the hypoxemia • Low Fi. O 2 (0. 3 -0. 5) usually need to raise Pa. O 2 >60 mm. Hg • High Fi. O 2 needs may indicates some kind of complication e. g. mucoid impaction, atelectasis, pneumonia, or pneumothorax • Complete correction of the respiratory acidosis is not an urgent priority

Alternative technique in difficult patients • Severe bronchospasm worsen ventilation, oxygenation and increase DHI • Ketamine – cause bronchodilatation by both sympathomimetic potentiation and direct effect on airway smooth muscle Pediatr Emerg Care 1996; 12: 294 -7 – Some case reports showed beneficial effects Pharmacotherapy. 2001 Sep; 21(9): 1100 -6 J Asthma. 2001 Dec; 38(8): 657 -64 – no clinical benefit in children with moderately severe asthmatic attacks Ann Emerg Med. 2005 Jul; 46(1): 43 -50

Alternative technique in difficult patients • Volatile inhalational agents (isoflurane, halothane): at 0. 5 -2% induce bronchodilation and sedative effect decreased bronchospasm Crit Care Med 1986; 14: 832 -833 Intensive Care Med 1990; 16: 104 -107 Pediatr Crit Care Med. 2000 Jul; 1(1): 55 -9 Arerugi. 2005 Jan; 54(1): 18 -23. Japanese • All are cases reports only and no RCT a/v

Alternative technique in difficult patients • Heliox: inert gas, 1/4 the density of air and mixtures of helium and oxygen (60% / 40%) decrease the work of breathing by decreasing inspiratory and expiratory resistance Crit Care Med 2000, 28: 2721– 2728 • The Cochrane Database of Systematic Reviews 2003, Issue 2. Art. No. : CD 002884 – 6 RCTs, 369 asthmatics – Heliox use did not improve pulmonary functions compared to standard care

Alternative technique in difficult patients • The Cochrane Database of Systematic Reviews 2001, Issue 1. Art. No. : CD 003571 – 4 RCTs, about 200 COPD patients – Heliox vs standard medical care – no significant differences in the change of FEV 1 and FVC between

Alternative technique in difficult patients • Mg. SO 4: block Ca channels thereby mediating smooth muscle relaxation and bronchodilation JAMA 1989; 262: 1210 -3 • The Cochrane Database of Systematic Reviews 2000, Issue 1. Art. No. : CD 001490 – 7 RCTs, 665 patients – Non-significant improvement of PFR – Admission rate decreased in severe subgroup but not in overall asthmatics

Weaning in asthma • When bronchoconstriction improves, dynamic hyperinflation (DHI) and auto-PEEP will decrease • Oxygenation may actually worsen at this point, but should improve with the addition of a small amount of PEEP (3 -5 cm H 2 O) • Weaning process should be brief, sedation off followed by SBT then extubation • Presence of ETT can induce bronchospasm again in stabilized patient ready for extubation

Weaning in COPD • Patients with COPD who require mechanical ventilation generally have greater dependence on ventilators than do those with asthma • 60% of total ventilator time for COPD patients was spent trying to wean Chest 2001; 119(6): 1840– 1849 • SBT can be tried in those with clinical improvement and haemodynamically stable patients • Patients who fail SBT may require a slower method of progressive withdrawal, by daily Tpiece SBTs of increasing duration or daily reductions in PSV

Weaning with help of NPPV • Cochrane Database Syst Rev. 2003; (4): CD 004127 – Randomized and quasi-randomized studies comparing early extubation with immediate application of NPPV to IPPV weaning in intubated adults with respiratory failure – Five studies included, involving 171 participants with predominantly COPD

Weaning with help of NPPV • Compared to the IPPV strategy, the NPPV strategy decreased mortality (RR 0. 41, 95% CI 0. 22 to 0. 76), the incidence of ventilator associated pneumonia (RR 0. 28, 95% CI 0. 09 to 0. 85), intensive care unit length of stay (WMD -6. 88 days, 95% CI -12. 60 to -1. 15), hospital length of stay (WMD -7. 33 days, 95%CI -14. 05 to -0. 61), total duration of mechanical support (WMD -7. 33 days, 95% CI -11. 45 to -3. 22) and the duration of endotracheal mechanical ventilation (WMD 6. 79 days, 95% CI -11. 70 to -1. 87)

The End Questions and comments
- Slides: 55